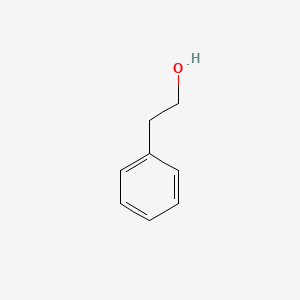
| The Content Variation of Natural Product Induced by Different Factor(s) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Species Name: phaseolorum sp. PR4 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor Name: PDB medium | [1] | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Experiment Detail |
PR4 was isolated as an endophyte from the rhizome of Picrorhiza kurroa. Picrorhiza kurroa Royle ex. Benth (Plantaginaceae) is a perennial herb endemic to the north western alpine Himalayas. The endophyte PR4 was grown on PDA and in PDB at 26 ℃ for 15 days with constant shaking at 200 rpm in the latter case.
Click to Show/Hide
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Mechanism |
The two candidate NR-PKSs (PKS_3671 and PKS_4063) show differences in their domain organizations. PKS_3671 possesses two ACP-domains. Apart from that, only PKS_3671 contains a SAT-domain . These domains provide the first building block in the polyketide assembly, which usually is different from the extender unit malonyl-CoA (also known as the 'starter unit effect'). The ACA-synthesis however is believed to involve merely malonyl-CoA molecules. Even though the ACA-producing PKSs MdpG, ACAS, EncA, AptA and ClaG contain SAT-domains, an amino acid sequence alignment of these domains revealed that they all lack the active-site cysteine in the GXCXG motif and therefore most likely have no acyl transferase activity. Instead, all malonate building blocks are assumed to be loaded by the MAT. Under this aspect, the SAT-domain of PKS_3671 (that includes the correct GXCXG motif) likely incorporates a starter unit different from malonyl-CoA indicating that this enzyme is not involved in the biosynthesis of ACA. Therefore, the ACA-synthesizing PKS in C. asteris would rather be PKS_4063 that misses the SAT-domain .In the monodictyphenone and cladofulvin pathways, the cluster-encoded gene products MdpH and ClaH are crucial enzymes pushing the biosynthesis towards emodin. These EthD-domain-containing enzymes are suggested to catalyze the decarboxylation of ACA (3) into atrochrysone (4). Surprisingly, no such EthD-domain is encoded in the whole C. asteris genome. On the other hand, four genes directly attached to the putative ACA-synthase-coding gene pks_4063 show high similarity to genes of non-investigated PKS clusters in other fungi , which indicates an involvement in tailoring reactions of the respective polyketide pathways. According to InterProScan and BLASTp analyses, the genes sky_4060-62 encode a dehydratase and two dehydrogenases potentially catalyzing the multistep conversion of ACA (3) into emodin (1). Gene sky_4059 codes for a monooxygenase that putatively can connect two emodin molecules to the final product skyrin (2) in the style of the monooxygenase ClaM involved in the dimerization of the bisanthraquinone cladofulvin. Thus, the presence of these genes in the gene cluster gives further support to the hypothesis that PKS_4063 is the ACA-synthase in C. asteris. Mutational studies will be done in order to confirm these assumptions after a gene transfer system for this strain has been developed.
Click to Show/Hide
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor | Part | Location | NP Content | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
PDB medium (26℃ + 4 days)
|
Rhizomes | Himalayas |
NP Content: 54.6 % Relative area
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Species Name: Eugenia chlorophylla | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor Name: Developmental Stage Variation | [2] | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Experiment Detail |
Plant material was collected at vegetative stage (stems and leaves,September 2005) and at flowering stage (leaves and flowers,December 2004), inCuritiba,Parana state, Brazil.
Click to Show/Hide
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor Function |
Thirty-four components were identified, representing more than 80% of total oil. The major components were beta-caryophyllene (flowers-12.8%), caryophyllene oxide (stems-17.2%), globulol (stems-16.5%; leaves-22.5% at vegetative stage and 18.9% at flowering stage), 1-epi-cubenol (stems-10.9%), epi-alpha-muurolol (stems-16.8%) and alpha-cadinol (stems-12.1%; flowers-10.1%).
Click to Show/Hide
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor | Part | Location | NP Content | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Flower: flowering stage
|
Flowers | Brazil |
NP Content: 1.5 %
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Species Name: Rosa damascena | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor Name: Variety Comparison | [3] | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Experiment Detail |
Experimental site: The present study was conducted at the experimental farm of the CSIR-Institute of Himalayan Bioresource Technology, Palampur (1325 m amsl, 32° 06′ 05″ N, 76° 34′10″ E), India, in 2011. Minimum temperature ranges from 3.5 ℃ to 19.8 ℃, maximum temperature ranges from 15.2 ℃ to 31.4 ℃, relative humidity varies between 62.2% and 94.1% in the morning and 45.0% and 87.2% in the evening, and bright sunshine hour ranges from 2.9 to 8.9 hours. Plant material: A population of approximately 50,000 plants raised from mixed stem cuttings collected from perennial rose plantations at the University of Agriculture, Udaipur, Rajasthan, India, and maintained in the field of the CSIR-Institute of Himalayan Bioresource Technology, Palampur, Himachal Pradesh, India, were utilized as an original gene pool of R. damascena. Two varieties, Jwala and Himroz were diversified through selections of desirable traits (morphological/oil content) across 25,000 plants. The five elites, three of R. damascena var. Jwala, (Indica, Super jwala and Jwala) and two of R. damascena var. Himroz (Hot himroz and Himroz) were developed through field selections and maintained at the Natural Plant Products Division Experimental Farm of the Institute. Rosa bourboniana plants were collected from the Fragrance and Flavour Development Centre, Kannauj, UP, India, during 1992 and maintained at the Natural Plant Products Division Experimental Farm of the Institute.
Click to Show/Hide
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor Function |
The essential oil content of the varieties of R. damascena varied from 0.037% to 0.051% and that of R. bourboniana was 0.017%. Super jwala recorded the highest oil content (0.051%). A total of 32 components were identified in the different varieties of rose oil. These components constituted 78.1-93.5% of the total rose oil species. The main components of rose oil were citronellol + nerol (16.3-30.1%), geraniol (15.8-29.3%), linalool (0.7-1.9%), rose oxide (0.9-2.6%), phenyl ethyl alcohol (0.1-0.4%), eugenol (0.3-2.2%), nonadecane (7.3-14.7%). The content of citronellol + nerol (30.1%) and geraniol (29.3%) was the highest in Himroz compared with other varieties.
Click to Show/Hide
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor | Part | Location | NP Content | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Rosa damascena var. Himroz
|
Flowers | India |
NP Content: 0.1 %
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Rosa damascena var. Hot Himroz
|
Flowers | India |
NP Content: 0.1 %
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Rosa damascena var. Indica
|
Flowers | India |
NP Content: 0.2 %
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Rosa damascena var. Jwala
|
Flowers | India |
NP Content: 0.1 %
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Rosa damascena var. Super Jwala
|
Flowers | India |
NP Content: 0.1 %
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Species Name: Thymus pseudopulegioides | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor Name: Locality Variation | [4] | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Experiment Detail |
Plant materials were collected from the following localities in north western Turkey. A = Trabzon: Caykara, Soganli dag on July 28, 1994; B = Bayburt: Caykara, Mohakambo yaylasi on July 25, 1994; C = Trabzon: Koprubasi, Vizara yaylasi on July 20, 1994.
Click to Show/Hide
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor Function |
One hundred and four compounds were identified representing 97.5-99.5% of the total components detected in thymol/carvacrol (50.14/10.67%), thymol/linalool (23.14/20.24%) and linalool/alpha-terpinyl acetate/geraniol (21.55/16.70/11.17%) rich oils.
Click to Show/Hide
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor | Part | Location | NP Content | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Locality: Soganli dag, Caykara, Trabzon, Eskisehir, Turkey
|
Aerial parts | Eskisehir, Turkey |
NP Content: <0.1 %
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||