| The Content Variation of Natural Product Induced by Different Factor(s) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Species Name: Vitis vinifera | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor Name: Variety Comparison | [1] | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Experiment Detail |
Grape pomaces and stalks of Nero d'Avola and Frappato were donated by the ''Valle dell'Acate'' wine firm, Acate, RG, Italy - those from Nerello Mascalese and Cabernet Sauvignon were given by the ''Emanuele Scammacca Barone del Murgo'' wine firm, Santa Venerina, CT, Italy. The winemaking procedures were similar for all samples, namely grape clusters were crushed and destemmed using a destemmer-crusher. The crushed grapes were treated with sulphur dioxide (0.2-0.5% total mash) and with selected strains of Saccharomyces cerevisiae to start up the fermentation. After 6-8 days of maceration, when alcoholic fermentation was finished, the mash was pressed. Stalks coming from destemming procedure and grape pomace coming from the maceration procedure were subjected to the distillation procedures within 24 h of their collection. All materials were collected during the 2004 vintage.
Click to Show/Hide
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor Function |
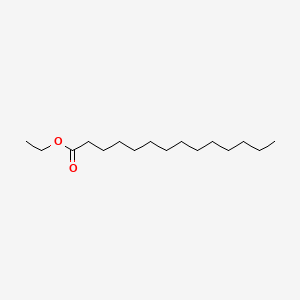
On the whole, 38 components have been characterized in the samples of grape pomaces, with Frappato cv. showing the richest composition; instead, 88 components have been detected in the stalks of Frappato, Nero d'Avola, Nerello Mascalese and Cabernet Sauvignon varieties.
Click to Show/Hide
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor | Part | Location | NP Content | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Vitis vinifera var. Cabernet Sauvignon
|
Pomaces | Italy |
NP Content: 1.6 %
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Vitis vinifera var. Frappato
|
Pomaces | Italy |
NP Content: 1.33 %
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Vitis vinifera var. Nerello Mascalese
|
Pomaces | Italy |
NP Content: 3.13 %
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Vitis vinifera var. Nero d'Avola
|
Pomaces | Italy |
NP Content: 1.94 %
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| References | ||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 1 | Volatile components of grape pomaces from different cultivars of Sicilian Vitis vinifera L. | |||||||||||||||||||||||