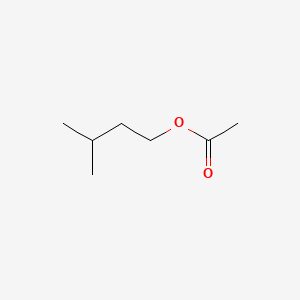
| The Content Variation of Natural Product Induced by Different Factor(s) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Species Name: Artemisia annua | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor Name: Cultivar Comparison | [1] | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Experiment Detail |
Populations of A. annua cultivar 'Jeevanraksha' and accession Suraksha were grown in the experimental field plot of the Institute at New Delhi. The seeds were sown in January 2004, seedlings transplanted in late February 2004 and aerial parts (flowers, leaves and stems from the upper 0.5 m of crop canopy) sampled in late October 2004.
Click to Show/Hide
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor Function |
Ninety-seven compounds comprising 91.3% of the total oil of 'Jeevanraksha' were identified. Forty-three monoterpenes (56.6%), 32 sesquiterpenes (31.1%), and 2 diterpenes (0.2%) comprised bulk of the oil (87.9%). The oil was devoid of artemisia ketone and contained camphor (13.5%), 1,8-cineole (9.4%), trans-sabinol (7.1%), p-mentha-1(7), 5-dien-2-ol (6.3%), myrcene (4.7%), germacrene D (4.4%), (E)-beta-farnesene (3.9%), beta-caryophyllene (3.7%), dihydroartemisinic lactone (3.0%) and p-cymene (2.0%) as the major constituents. Eighty-six compounds representing 93.3% of the composition were identified in the Suraksha oil. This oil contained artemisia ketone (47%), 1,8-cineole (8.4%), camphor (5.9%) and alpha-pinene (5.2%) as the major components.
Click to Show/Hide
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor | Part | Location | NP Content | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Artemisia annua accessions Suraksha
|
Aerial parts | India |
NP Content: <0.05 %
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Artemisia annua cv. Jeevanraksha
|
Aerial parts | India |
NP Content: <0.05 %
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Species Name: Chamaemelum nobile | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor Name: Locality Variation | [2] | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Experiment Detail |
Two different Roman chamomile oils were obtained from Giardino Officinale in Casola Valsenio (RA - Italy). One oil was distilled from chamomile plants grown in the Emilia-Romagna region (E), and the other one came from the Piemonte region (P) in Northern Italy
Click to Show/Hide
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor Function |
No significant differences were found in the composition of the two oils which were characterized by a high content of isobutyl angelate (36.3-38.5%), 2-methylbutyl angelate (18.2-20.3%) and other angelate esters.
Click to Show/Hide
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor | Part | Location | NP Content | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Locality: Emilia-Romagna region, Italy
|
Whole plant | Italy |
NP Content: 0.1 %
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Locality: Piemonte region, Italy
|
Whole plant | Italy |
NP Content: 0.1 %
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Species Name: Fragaria × ananassa Duch. | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor Name: Phosphate Treatment; AMF Inoculation; Nutrient Treatment | [3] | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Experiment Detail |
Seedlings of Fragaria × ananassa Duch var. Elyana F1 with continuous flowering habit, were transplanted in plastic pots (400 mL) in a soil (Brill Ortopack, Agrochimica, Bolzano, Italy; pH 5.5-6.5) previously sterilized by flowing steam (101℃ for 1 h), and kept in a greenhouse for rooting. After 1 month, they were transplanted in new plastic pots (900 mL) in a 2/1 v/v mixture of sterile soil (the same used in the first transplant) and sand (sterilized in oven at 180℃ for 3 h), and inoculated or not with one of three different AMF in combination with one of three different bacterial strains . Plants were initially irrigated 3 times per week. When the plants began to produce fruits, they were transplanted again in plastic pots of 3 L capacity. Starting from 1 week after the last transplant, they were irrigated daily: once per week with a Long Ashton (LA) nutrient solution , and with tap water on the other days. In particular, half of the control plants (C) were watered with LA 32 µM phosphate, while the remaining controls (C-P) and all the inoculated plants were fed with LA 16 µM phosphate until harvest. Strawberry plants were maintained in greenhouse for 16 weeks.The arbuscular mycorrhizal fungus Rhizophagus irregularis (Ri) (DAOM197-198) was provided by INRA (Recorbet and Bernaud, Dijon). Funneliformis mosseae (Fm) (BEG12) was provided by the European Bank of Glomales (Dijon). Septoglomus viscosum (Sv), collected from an Italian soil (Tuscany, Italy), was produced and provided by Mybasol S.r.l. (Alessandria, Italy). The three inocula, prepared as a mixture of soil, mycorrhizal roots, hyphae, and spores, were mixed (11% v/v) with the plant growth medium.Pseudomonas fluorescens strain Pf4 (Pf4) was isolated from a forest soil located in Sassello (Savona, Italy) and characterized by Berta et al. . Pseudomonas sp. 5Vm1K (5Vm) was isolated from the rhizosphere of blueberry plants grown in a larch woodland (Bellino, Cuneo, Italy) and characterized as described by Bona et al. . P. fluorescens strain 19Fv1t (19Fv) was provided by Mybasol s.r.l (Alessandria, Italy) and characterized by Bona et al. . Bacterial 16S rDNA sequences were deposited in the NCBI database GenBank with the accession numbers KF234076, KF233995, KF752592 for Pf4, 5Vm, and 19Fv, respectively.Bacterial cells were grown on tryptic soy agar (TSA) at 28℃ for 48 h and suspended in 0.1M MgSO4. Bacterial density (600 nm) was adjusted to 109 CFU/mL. Each plant was inoculated with 8 mL of bacterial suspension, except uninoculated ones that were irrigated with the same quantity of MgSO4. After 1 week the plants were inoculated again.
Click to Show/Hide
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor Function |
In general, AMF mostly affected the parameters associated with the vegetative portion of the plant, while plant growth promoting bacteria (PGPB) were especially relevant for fruit yield and quality. The plant physiological status was differentially affected by inoculations, resulting in enhanced root and shoot biomass. Inoculation with Pf4 bacterial strain increased flower and fruit production per plant and malic acid content in fruits, while decreased the pH value, regardless of the used fungus. Inoculations affected fruit nutritional quality, increasing sugar and anthocyanin concentrations, and modulated pH, malic acid, volatile compounds and elements.
Click to Show/Hide
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor | Part | Location | NP Content | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
32 µM phosphate (P) in Long Ashton (LA) nutrient solution
|
Fruits | NA |
NP Content: 0.020 ± 0.011 peak area
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
16 µM P in LA nutrient solution
|
Fruits | NA |
NP Content: 0.0150 ± 0.0070 peak area
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
(Funneliformis mosseae BEG12 (Fm) and Pseudomonas fluorescens 19Fv1t (19Fv) inoculation) + (16 µM P in LA)
|
Fruits | NA |
NP Content: 0.0090 ± 0.0040 peak area
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
(Fm and Pseudomonas sp. 5Vm1K (5Vm) inoculation) + (16 µM P in LA)
|
Fruits | NA |
NP Content: 0.0130 ± 0.0030 peak area
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
(Fm and Pseudomonas fluorescens Pf4 (Pf4) inoculation) + (16 µM P in LA)
|
Fruits | NA |
NP Content: 0.0070 ± 0.0010 peak area
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
(Septoglomus viscosum (Sv) and (19Fv) inoculation) + (16 µM P in LA)
|
Fruits | NA |
NP Content: 0.0200 ± 0.0070 peak area
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
(Sv and 5Vm inoculation) + (16 µM P in LA)
|
Fruits | NA |
NP Content: 0.0130 ± 0.0050 peak area
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
(Sv and Pf4 inoculation) + (16 µM P in LA)
|
Fruits | NA |
NP Content: 0.052 ± 0.017 peak area
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
(Rhizophagus irregularis DAOM197-198 (Ri) and 19Fv inoculation) + (16 µM P in LA)
|
Fruits | NA |
NP Content: 0.0110 ± 0.0040 peak area
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
(Ri and 5Vm inoculation) + (16 µM P in LA)
|
Fruits | NA |
NP Content: 0.0140 ± 0.0070 peak area
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
(Ri and Pf4 inoculation) + (16 µM P in LA)
|
Fruits | NA |
NP Content: 0.0160 ± 0.0050 peak area
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Species Name: Tanacetum cadmeum ssp. orientale | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor Name: Locality Variation | [4] | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Experiment Detail |
Plant materials were collected during the flowering period in July 2002 from the Dumluca Mountain in the vicinity of Divrigi village of Sivas city at 1900 m altitude and Saksagan Gorge in Saimbeyli village of Adana city at 1900 m altitude.
Click to Show/Hide
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor Function |
The flower, stem and root oils of T. cadmeum ssp. orientale collected from the Adana location were characterized with alpha-thujone (25%, 5.2%), cis-linalool oxide (6.8%, 12.8%), trans-chrysanthenyl acetate (5.8%, 8.5%) for flower and stem oils, and beta-eudesmol (10.3%, 6.2%, 13.8%); in addition, stem oil contained 1,8-cineole (6.6%) and root oil contained hexadecanoic acid (6.0%), spathulenol (5.8%) and beta-muurolol (5.3%). The flower and stem oils of T. cadmeum ssp. orientale collected from the Sivas location were characterized with camphor (25.9%, 14.8%), borneol (15.4%, 25.8%) and alpha-thujone (7.8%, 5.5%); in addition, stem oil contained 1,8-cineole (7.4%) and root oil contained nonacosane (16.2%), spathulenol (6.8%) and hexadecanoic acid (5.8%).
Click to Show/Hide
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor | Part | Location | NP Content | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Flower: (Locality: Adana, Turkey)
|
Flowers | Adana, Turkey |
NP Content: 0.1 %
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Flower: (Locality: Sivas, Turkey)
|
Flowers | Sivas, Turkey |
NP Content: 0.1 %
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Species Name: Vitis vinifera | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor Name: Variety Comparison | [5] | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Experiment Detail |
Grape pomaces and stalks of Nero d'Avola and Frappato were donated by the ''Valle dell'Acate'' wine firm, Acate, RG, Italy - those from Nerello Mascalese and Cabernet Sauvignon were given by the ''Emanuele Scammacca Barone del Murgo'' wine firm, Santa Venerina, CT, Italy. The winemaking procedures were similar for all samples, namely grape clusters were crushed and destemmed using a destemmer-crusher. The crushed grapes were treated with sulphur dioxide (0.2-0.5% total mash) and with selected strains of Saccharomyces cerevisiae to start up the fermentation. After 6-8 days of maceration, when alcoholic fermentation was finished, the mash was pressed. Stalks coming from destemming procedure and grape pomace coming from the maceration procedure were subjected to the distillation procedures within 24 h of their collection. All materials were collected during the 2004 vintage.
Click to Show/Hide
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor Function |
On the whole, 38 components have been characterized in the samples of grape pomaces, with Frappato cv. showing the richest composition; instead, 88 components have been detected in the stalks of Frappato, Nero d'Avola, Nerello Mascalese and Cabernet Sauvignon varieties.
Click to Show/Hide
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor | Part | Location | NP Content | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Vitis vinifera var. Nerello Mascalese
|
Pomaces | Italy |
NP Content: < 0.05 %
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Vitis vinifera var. Nero d'Avola
|
Pomaces | Italy |
NP Content: 0.29 %
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||