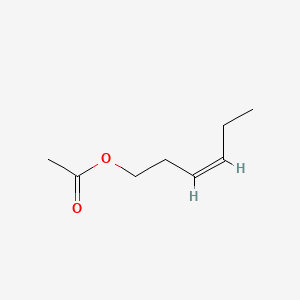
| The Content Variation of Natural Product Induced by Different Factor(s) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Species Name: Arabidopsis thaliana | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor Name: T-DNA Knock-Out Treatment | [1] | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Experiment Detail |
In vitro cultivation of Arabidopsis wildtype and mutant plants: Seeds were sterilized according to standard lab routines (EtOH, NaOCl/NaOH) prior to aseptical (in vitro) cultivation in 500 ml screw cap jars on MS medium (4.3 g/l; 50 ml/jar) containing Bacto- and Phytoagar (1:2; 6 g/l) and 30 g/l sucrose. Ten seeds were pipetted into each jar and plants grown for 6 weeks until flowering at a temperature of 20 ℃ under a 16/8 h day/ night regime using fluorescent tubes (Osram Lumilux Plus Eco 36 W). Both Arabidopsis thaliana wildtype plants of ecotype Columbia-0 (Col) and 4 Col-derived T-DNA knock-out mutants (homozygous lines) showing deficiencies in the GLS biosynthesis pathway were used in this study (five parallels for wildtype and mutants): TGG1 (Atg526000; Salk_130469), TGG2 (At5g25980; Salk_038730), Cyp83A1 (At4g13770) and Cyp83B1 (At4g31500; Salk_028573). Greenhouse-cultivation of Arabidopsis ecotypes: The following Arabidopsis ecotypes were used in the study: Columbia (Col), Cape Verde Islands (Cvi), Landsberg erecta (Ler) and Wassilewskija (Ws). Single plants were greenhouse-cultivated on fertilized soil (P-Jord; Emmaljunga Torvmull AB) in plug trays (9 × 6 cells) at a temperature of 20 ℃ (three parallels for each ecotype). Due to the 6-weeks growth period (November/December 2003), the plants were cultivated under a 16/8 h day/night regime using metal halide lamps (Osram HQI-T 400 W) placed 130 cm above the trays. Depending on the ecotypical plant development, whole plants were sampled after 3-4 weeks right before bolting for in vivo studies, while investigations of single plant organs (leaf, stem, inflorescence) were carried out after 5-6 weeks of cultivation.
Click to Show/Hide
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor Function |
Metabolites from methionine, leucine and phenylalanine-derived glucosinolates were most abundant (4-methylthiobutyl, 4-methylpentyl, 2-phenylethyl). In addition, 24 monoterpenes, 26 sesquiterpenes and 12 aromatic structures, predominantly observed in inflorescenses, are described. Excluding the vast group of straight chain aliphatic structures, a total of 102 volatile compounds were detected, of which 59 are reported in Arabidopsis thaliana for the first time, thus emphasizing the sensitivity and applicability of solid-phase microextraction for volatile profiling of plant secondary metabolites.
Click to Show/Hide
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor | Part | Location | NP Content | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Arabidopsis thaliana wildtype
|
Whole plant | Norway |
NP Content: 26.9 %
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
A. thaliana T-DNA knock-out mutant Cyp83A1: (At4g13770)
|
Whole plant | Norway |
NP Content: 12.25 %
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
A. thaliana T-DNA knock-out mutant Cyp83B: (At4g31500; Salk_028573)
|
Whole plant | Norway |
NP Content: 5.5 %
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
A. thaliana T-DNA knock-out mutant TGG1: (Atg526000; Salk_130469)
|
Whole plant | Norway |
NP Content: 17.69 %
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
A. thaliana T-DNA knock-out mutant TGG2: (At5g25980; Salk_038730)
|
Whole plant | Norway |
NP Content: 23.64 %
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Species Name: Teucrium chamaedrys | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor Name: Locality Variation | [2] | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Experiment Detail |
The aerial parts of T. chamaedrys were collected at the flowering stage in June 2004 near Corti, Corsica, France and near Oristano, Sardinia, Italy
Click to Show/Hide
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor Function |
The Corsican and Sardinian oils of T. chamaedrys investigated in this study were qualitatively similar but they differed by the amount of their major components. The major components were beta-caryophyllene (29.0% and 27.4%, respectively) and germacrene D (19.4% and 13.5%, respectively), followed by alpha-humulene (6.8%) and delta-cadinene (5.4%) in the Corsican oil and by caryophyllene oxide (12.3%) and alpha-humulene (6.5%) in the Sardinian oil. These quantitative differences are also noticeable on the amounts of the different class compounds. Especially, the monoterpene hydrocarbons amounted for 10.3% and 4.1% in Sardinian and Corsican oils respectively and the oxygenated sesquiterpenes amounted for 18.9% and only 7.4% in both oils, respectively. Both oils were qualitatively rather similar in comparison with those reported in the literature from various geographic regions. However, among the 87 components identified in this study, 47 minor components (< 0.6%) reported were identified for the first time in T. chamaedrys oil. This study confirms the quantitative variability of the major components according to the plant origin.
Click to Show/Hide
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor | Part | Location | NP Content | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Locality: Corti, Corsica, France
|
Aerial parts | France |
NP Content: <0.05 %
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Species Name: Vitis vinifera | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor Name: Variety Comparison | [3] | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Experiment Detail |
Grape pomaces and stalks of Nero d'Avola and Frappato were donated by the ''Valle dell'Acate'' wine firm, Acate, RG, Italy - those from Nerello Mascalese and Cabernet Sauvignon were given by the ''Emanuele Scammacca Barone del Murgo'' wine firm, Santa Venerina, CT, Italy. The winemaking procedures were similar for all samples, namely grape clusters were crushed and destemmed using a destemmer-crusher. The crushed grapes were treated with sulphur dioxide (0.2-0.5% total mash) and with selected strains of Saccharomyces cerevisiae to start up the fermentation. After 6-8 days of maceration, when alcoholic fermentation was finished, the mash was pressed. Stalks coming from destemming procedure and grape pomace coming from the maceration procedure were subjected to the distillation procedures within 24 h of their collection. All materials were collected during the 2004 vintage.
Click to Show/Hide
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor Function |
On the whole, 38 components have been characterized in the samples of grape pomaces, with Frappato cv. showing the richest composition; instead, 88 components have been detected in the stalks of Frappato, Nero d'Avola, Nerello Mascalese and Cabernet Sauvignon varieties.
Click to Show/Hide
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor | Part | Location | NP Content | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Vitis vinifera var. Frappato
|
Stalks | Italy |
NP Content: < 0.05 %
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Vitis vinifera var. Nero d'Avola
|
Stalks | Italy |
NP Content: 0.3 %
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||