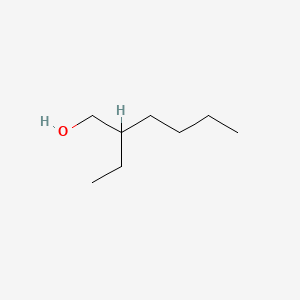
| The Content Variation of Natural Product Induced by Different Factor(s) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Species Name: Aquilaria agallocha | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor Name: Plant Pathogen Infection | [1] | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Experiment Detail |
Artificial inoculation of fungal isolates: The most frequently isolated fungi from infected agarwood (e.g. Chaetomium globosum and Fusarium oxysporum) were inoculated to the healthy plants by artifi cial boring on to the plants. Inoculation was made with two different fungi alone and in their combination. Observations were made at an interval of 30 days after inoculation.
Click to Show/Hide
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor Function |
This investigation showed a marked difference in the oil compositions among the treatments with regards to their quality. Valerianol (3.0%) and tetradec-anioc acid (7.1%) contents were recorded higher in the oils of naturally infected plants than in that of healthy ones (0.1% and 6.9%, respectively). Pentadecenoic acid was totally absent in the oils of healthy, whereas it was found in a greater amount (6.8%) in the oil of naturally infected plants. In contrast, dodecanoic acid (3.1%), pentadecanoic acid (6.2%), hexadecanoic acid (31.5%) and octadecanoic acid (4.1%) were found in a higher amount in the oils of healthy plants, while the oils obtained from naturally infected plants contained lower amounts of these components (2.5%, 4.8%, 20.0% and 1.0%, respectively). The oils obtained from the inoculated plants showed almost similar distribution of the components with healthy plants.
Click to Show/Hide
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor | Part | Location | NP Content | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Artificial inoculation plants (Fusarium oxysporum)
|
Wood chips | India |
NP Content: trace %
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Artificial inoculation plants (C. globosum and F. oxysporum)
|
Wood chips | India |
NP Content: trace %
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Species Name: Arabidopsis thaliana | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor Name: T-DNA Knock-Out Treatment | [2] | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Experiment Detail |
In vitro cultivation of Arabidopsis wildtype and mutant plants: Seeds were sterilized according to standard lab routines (EtOH, NaOCl/NaOH) prior to aseptical (in vitro) cultivation in 500 ml screw cap jars on MS medium (4.3 g/l; 50 ml/jar) containing Bacto- and Phytoagar (1:2; 6 g/l) and 30 g/l sucrose. Ten seeds were pipetted into each jar and plants grown for 6 weeks until flowering at a temperature of 20 ℃ under a 16/8 h day/ night regime using fluorescent tubes (Osram Lumilux Plus Eco 36 W). Both Arabidopsis thaliana wildtype plants of ecotype Columbia-0 (Col) and 4 Col-derived T-DNA knock-out mutants (homozygous lines) showing deficiencies in the GLS biosynthesis pathway were used in this study (five parallels for wildtype and mutants): TGG1 (Atg526000; Salk_130469), TGG2 (At5g25980; Salk_038730), Cyp83A1 (At4g13770) and Cyp83B1 (At4g31500; Salk_028573). Greenhouse-cultivation of Arabidopsis ecotypes: The following Arabidopsis ecotypes were used in the study: Columbia (Col), Cape Verde Islands (Cvi), Landsberg erecta (Ler) and Wassilewskija (Ws). Single plants were greenhouse-cultivated on fertilized soil (P-Jord; Emmaljunga Torvmull AB) in plug trays (9 × 6 cells) at a temperature of 20 ℃ (three parallels for each ecotype). Due to the 6-weeks growth period (November/December 2003), the plants were cultivated under a 16/8 h day/night regime using metal halide lamps (Osram HQI-T 400 W) placed 130 cm above the trays. Depending on the ecotypical plant development, whole plants were sampled after 3-4 weeks right before bolting for in vivo studies, while investigations of single plant organs (leaf, stem, inflorescence) were carried out after 5-6 weeks of cultivation.
Click to Show/Hide
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor Function |
Metabolites from methionine, leucine and phenylalanine-derived glucosinolates were most abundant (4-methylthiobutyl, 4-methylpentyl, 2-phenylethyl). In addition, 24 monoterpenes, 26 sesquiterpenes and 12 aromatic structures, predominantly observed in inflorescenses, are described. Excluding the vast group of straight chain aliphatic structures, a total of 102 volatile compounds were detected, of which 59 are reported in Arabidopsis thaliana for the first time, thus emphasizing the sensitivity and applicability of solid-phase microextraction for volatile profiling of plant secondary metabolites.
Click to Show/Hide
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor | Part | Location | NP Content | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Leaves: A. thaliana wildtype detected in vivo
|
Leaves | Norway |
NP Content: 0.24 %
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Stem: A. thaliana wildtype detected in vivo
|
Stems | Norway |
NP Content: 0.16 %
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Species Name: Tanacetum cadmeum ssp. orientale | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor Name: Locality Variation | [3] | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Experiment Detail |
Plant materials were collected during the flowering period in July 2002 from the Dumluca Mountain in the vicinity of Divrigi village of Sivas city at 1900 m altitude and Saksagan Gorge in Saimbeyli village of Adana city at 1900 m altitude.
Click to Show/Hide
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor Function |
The flower, stem and root oils of T. cadmeum ssp. orientale collected from the Adana location were characterized with alpha-thujone (25%, 5.2%), cis-linalool oxide (6.8%, 12.8%), trans-chrysanthenyl acetate (5.8%, 8.5%) for flower and stem oils, and beta-eudesmol (10.3%, 6.2%, 13.8%); in addition, stem oil contained 1,8-cineole (6.6%) and root oil contained hexadecanoic acid (6.0%), spathulenol (5.8%) and beta-muurolol (5.3%). The flower and stem oils of T. cadmeum ssp. orientale collected from the Sivas location were characterized with camphor (25.9%, 14.8%), borneol (15.4%, 25.8%) and alpha-thujone (7.8%, 5.5%); in addition, stem oil contained 1,8-cineole (7.4%) and root oil contained nonacosane (16.2%), spathulenol (6.8%) and hexadecanoic acid (5.8%).
Click to Show/Hide
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor | Part | Location | NP Content | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Root: (Locality: Adana, Turkey)
|
Roots | Adana, Turkey |
NP Content: 0.1 %
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||