| The Content Variation of Natural Product Induced by Different Factor(s) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Species Name: Aquilaria agallocha | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor Name: Plant Pathogen Infection | [1] | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Experiment Detail |
Artificial inoculation of fungal isolates: The most frequently isolated fungi from infected agarwood (e.g. Chaetomium globosum and Fusarium oxysporum) were inoculated to the healthy plants by artifi cial boring on to the plants. Inoculation was made with two different fungi alone and in their combination. Observations were made at an interval of 30 days after inoculation.
Click to Show/Hide
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor Function |
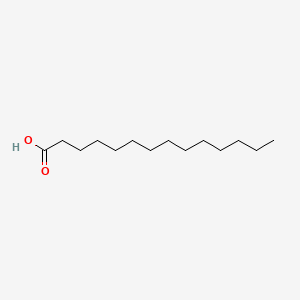
This investigation showed a marked difference in the oil compositions among the treatments with regards to their quality. Valerianol (3.0%) and tetradec-anioc acid (7.1%) contents were recorded higher in the oils of naturally infected plants than in that of healthy ones (0.1% and 6.9%, respectively). Pentadecenoic acid was totally absent in the oils of healthy, whereas it was found in a greater amount (6.8%) in the oil of naturally infected plants. In contrast, dodecanoic acid (3.1%), pentadecanoic acid (6.2%), hexadecanoic acid (31.5%) and octadecanoic acid (4.1%) were found in a higher amount in the oils of healthy plants, while the oils obtained from naturally infected plants contained lower amounts of these components (2.5%, 4.8%, 20.0% and 1.0%, respectively). The oils obtained from the inoculated plants showed almost similar distribution of the components with healthy plants.
Click to Show/Hide
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor | Part | Location | NP Content | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Healthy plants
|
Wood chips | India |
NP Content: 6.9 %
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Naturally infected plants (C. globosum or F. oxysporum)
|
Wood chips | India |
NP Content: 7.1 %
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Artificial inoculation plants (Chaetomium globosum)
|
Wood chips | India |
NP Content: 6.4 %
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Artificial inoculation plants (Fusarium oxysporum)
|
Wood chips | India |
NP Content: 5.8 %
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Artificial inoculation plants (C. globosum and F. oxysporum)
|
Wood chips | India |
NP Content: 6.2 %
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Species Name: Lagenaria siceraria | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor Name: Cultivar Comparison; Developmental Stage Variation | [2] | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Experiment Detail |
The experiment was conducted from July to November 2007 at the experimental station of the University of Abobo-Adjame (Abidjan, Ivory Coast) (latitude between 5° 17′ and 5° 31′ N, longitude between 3° 45′ and 4° 22′ W). During this period, rainfall, mean temperature and humidity varied from 5.33 to 192.28 mm, from 23.3 to 26.4 ℃ and from 86.7 to 96% respectively. Open-pollinated accessions from two edible-seeded L. siceraria (Molina) Standl. cultivars recognisable by their fruit shape (oval or round) were used. Seeds from the round fruit cultivar are characterised by the presence of a cap on the distal side, whereas those from the oval fruit cultivar lack this cap. Differences are also noted between the two cultivars in their rates of seed germination and seedling emergence, the best performances being observed for the round fruit cultivar. However, the visual changes in fruits during their growth as well as at plant whiteness are the same in the two cultivars. Both round and oval fruit cultivars were obtained from the cucurbit germplasm of the university, where they are identified by the alphanumeric codes NI354 and NI260 respectively. Each cultivar was sown on a plot of 20 m × 20 m in 12 holes. Female flowers were tagged after their closure in order to monitor the fruits until the date determined for harvesting. Fruits were harvested at three stages of maturation: (i) 30 days after fruit set (DAFS), at which stage fruits do not grow any more; (ii) 50 DAFS, at which stage the colour of fruits no longer changes; (iii) complete plant whiteness (CPW), indicating the end of plant growth. For each of the three fruit maturation times, five fruits per cultivar were selected. The seeds were extracted from each fruit, washed and dried in the sun for 1 week. After drying, the seeds of all five fruits were grouped and decorticated to obtain seed kernels that were used for analysis.
Click to Show/Hide
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor Function |
The results of this study showed that there were considerable modifications during fruit maturation in the oilseed gourd L. siceraria and that chemical characterisation of the seed kernel is important for controlling the processes of maturation. The two cultivars of L. siceraria studied should be harvested at 50 days after fruit set (DAFS) owing to their high contents of proteins, lipids, energy and minerals at this stage. At 50 DAFS the leaves of L. siceraria were still green and could serve as a source of nutrients for livestock. However, to obtain the best amino acid composition and biological values of proteins, the round and oval berry cultivars should be harvested at 30 DAFS and complete plant whiteness (CPW) respectively. At these stages their proteins could be used as a supplement. The low digestibility of the proteins at this stage could be improved by appropriate technological treatment.
Click to Show/Hide
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor | Part | Location | NP Content | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Lagenaria siceraria cv. Round berry + Harvesting time: 30 days after fruit set
|
Seed kernels | Abidjan, Cote d'lvoire |
NP Content: 1.2 ± 0.1 g/kg dry matter
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Lagenaria siceraria cv. Round berry + Harvesting time: 50 days after fruit set
|
Seed kernels | Abidjan, Cote d'lvoire |
NP Content: 0.8 ± 0.0 g/kg dry matter
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Lagenaria siceraria cv. Round berry + Harvesting time: complete plant whiteness stage
|
Seed kernels | Abidjan, Cote d'lvoire |
NP Content: 1.0 ± 0.1 g/kg dry matter
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Lagenaria siceraria cv. Oval berry + Harvesting time: 30 days after fruit set
|
Seed kernels | Abidjan, Cote d'lvoire |
NP Content: 1.0 ± 0.1 g/kg dry matter
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Lagenaria siceraria cv. Oval berry + Harvesting time: 50 days after fruit set
|
Seed kernels | Abidjan, Cote d'lvoire |
NP Content: 0.8 ± 0.1 g/kg dry matter
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Lagenaria siceraria cv. Oval berry + Harvesting time: complete plant whiteness stage
|
Seed kernels | Abidjan, Cote d'lvoire |
NP Content: 0.7 ± 0.1 g/kg dry matter
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Species Name: Stachys pilifera | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor Name: Locality Variation | [3] | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Experiment Detail |
Plant material and isolation procedure: Aerial parts of the plant were collected from two regions, from Kazeroon in southern Iran and Shahr-e-kord in western Iran at the time of flowering in June 2002.
Click to Show/Hide
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor Function |
The main components of the oil of S. pilifera collected from Kazeroon, in southern Iran, were spathulenol (15.8%), cis-chrysanthenol (15.3%), beta-caryophyllene (8.4%) and cis-chrysanthenyl acetate (6.9%), while for the plant collected from Shahr-e-kord, in western Iran, they were cis-chrysanthenyl acetate (21.8%), linalool (18.9%), terpinen-4-ol (11.9%) and cis-chrysanthenol (9.2%).
Click to Show/Hide
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor | Part | Location | NP Content | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Locality: Kazeroon, southern Iran
|
Aerial parts | Iran |
NP Content: 0.1 %
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Species Name: Tanacetum larvatum | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor Name: Locality Variation | [4] | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Experiment Detail |
Aerial parts of T. larvatum were collected in July and August during a five-year period, starting in 2001, in Montenegro on several locations: Planinica (Sample a), Visitor (Sample b) and Sinjajevina (Sample c).
Click to Show/Hide
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor Function |
Sixty-four components were identified, representing 83.1%, 96.6% and 89.4% of the total oils content in the Planinica [Sample a], Visitor [Sample b] and Sinjajevina [Sample c], respectively. The major constituent in Samples a and b , was oxygenated monoterpene, trans-sabinyl acetate (38.1% and 55.8% respectively). Monoterpene hydrocarbons, beta-pinene (13.5%) and santolinatriene (30.6%), were found to be the dominant components in Sample c. The toxic trans-sabinyl acetate was present only in traces in this sample. trans-Chrysanthenyl acetate, as one of major components in feverfew essential oil, has not been previously identified in the investigated essential oils.
Click to Show/Hide
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor | Part | Location | NP Content | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Locality: Planinica, Montenegro
|
Aerial parts | Montenegro |
NP Content: 0.1 %
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Locality: Sinjajevina, Montenegro
|
Aerial parts | Montenegro |
NP Content: 0.4 %
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Locality: Visitor, Montenegro
|
Aerial parts | Montenegro |
NP Content: <0.1 %
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||