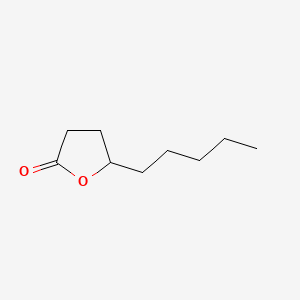
| The Content Variation of Natural Product Induced by Different Factor(s) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Species Name: Arabidopsis thaliana | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor Name: T-DNA Knock-Out Treatment | [1] | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Experiment Detail |
In vitro cultivation of Arabidopsis wildtype and mutant plants: Seeds were sterilized according to standard lab routines (EtOH, NaOCl/NaOH) prior to aseptical (in vitro) cultivation in 500 ml screw cap jars on MS medium (4.3 g/l; 50 ml/jar) containing Bacto- and Phytoagar (1:2; 6 g/l) and 30 g/l sucrose. Ten seeds were pipetted into each jar and plants grown for 6 weeks until flowering at a temperature of 20 ℃ under a 16/8 h day/ night regime using fluorescent tubes (Osram Lumilux Plus Eco 36 W). Both Arabidopsis thaliana wildtype plants of ecotype Columbia-0 (Col) and 4 Col-derived T-DNA knock-out mutants (homozygous lines) showing deficiencies in the GLS biosynthesis pathway were used in this study (five parallels for wildtype and mutants): TGG1 (Atg526000; Salk_130469), TGG2 (At5g25980; Salk_038730), Cyp83A1 (At4g13770) and Cyp83B1 (At4g31500; Salk_028573). Greenhouse-cultivation of Arabidopsis ecotypes: The following Arabidopsis ecotypes were used in the study: Columbia (Col), Cape Verde Islands (Cvi), Landsberg erecta (Ler) and Wassilewskija (Ws). Single plants were greenhouse-cultivated on fertilized soil (P-Jord; Emmaljunga Torvmull AB) in plug trays (9 × 6 cells) at a temperature of 20 ℃ (three parallels for each ecotype). Due to the 6-weeks growth period (November/December 2003), the plants were cultivated under a 16/8 h day/night regime using metal halide lamps (Osram HQI-T 400 W) placed 130 cm above the trays. Depending on the ecotypical plant development, whole plants were sampled after 3-4 weeks right before bolting for in vivo studies, while investigations of single plant organs (leaf, stem, inflorescence) were carried out after 5-6 weeks of cultivation.
Click to Show/Hide
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor Function |
Metabolites from methionine, leucine and phenylalanine-derived glucosinolates were most abundant (4-methylthiobutyl, 4-methylpentyl, 2-phenylethyl). In addition, 24 monoterpenes, 26 sesquiterpenes and 12 aromatic structures, predominantly observed in inflorescenses, are described. Excluding the vast group of straight chain aliphatic structures, a total of 102 volatile compounds were detected, of which 59 are reported in Arabidopsis thaliana for the first time, thus emphasizing the sensitivity and applicability of solid-phase microextraction for volatile profiling of plant secondary metabolites.
Click to Show/Hide
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor | Part | Location | NP Content | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Inflorescences: A. thaliana wildtype detected in vivo
|
Flowers | Norway |
NP Content: 0.36 %
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Leaves: A. thaliana wildtype detected in vivo
|
Leaves | Norway |
NP Content: 0.21 %
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Stem: A. thaliana wildtype detected in vivo
|
Stems | Norway |
NP Content: 0.2 %
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| References | ||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 1 | Volatile profiling of Arabidopsis thaliana - Putative olfactory compounds in plant communication | |||||||||||||||||||||||