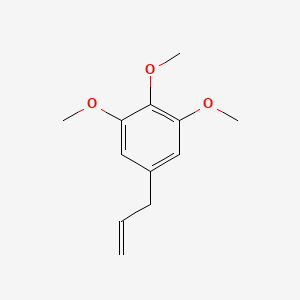
| The Content Variation of Natural Product Induced by Different Factor(s) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Species Name: Eugenia chlorophylla | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor Name: Developmental Stage Variation | [1] | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Experiment Detail |
Plant material was collected at vegetative stage (stems and leaves,September 2005) and at flowering stage (leaves and flowers,December 2004), inCuritiba,Parana state, Brazil.
Click to Show/Hide
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor Function |
Thirty-four components were identified, representing more than 80% of total oil. The major components were beta-caryophyllene (flowers-12.8%), caryophyllene oxide (stems-17.2%), globulol (stems-16.5%; leaves-22.5% at vegetative stage and 18.9% at flowering stage), 1-epi-cubenol (stems-10.9%), epi-alpha-muurolol (stems-16.8%) and alpha-cadinol (stems-12.1%; flowers-10.1%).
Click to Show/Hide
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor | Part | Location | NP Content | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Flower: flowering stage
|
Flowers | Brazil |
NP Content: 0.6 %
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Species Name: Pimpinella tragioides | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor Name: Developmental Stage Variation | [2] | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Experiment Detail |
Plant materials were collected from Chalous Road (north of Tehran province) both at the flowering stage in June and the seed stage in September 2003. The fresh plants were dried at room temperature. Dried stems/leaves (S/L) (collected during flowering stage), seeds (S) were hydrodistilled for 3 h in a Clevenger-type apparatus to produce the oils.
Click to Show/Hide
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor Function |
The major constituent in the stem/leaf oil was trans-alpha-bergamotene (77.1%), whereas the major constituent of the seed oil was pregeijerene (87.0%). Nonadecane (8.6%) were the other major constituents in the stem/leaf.
Click to Show/Hide
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor | Part | Location | NP Content | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Leaf and stem: flowering stage
|
Leaves and stems | Iran |
NP Content: 0.9 %
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Species Name: Portenschlagiella ramosissima | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor Name: Developmental Stage Variation | [3] | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Experiment Detail |
Plant material was collected in October 2003. in Herceg Novi, Montenegro. The air-dried roots (54 g), seeds (73.5 g) and aerial parts during vegetative phase (V, 150 g) and aerial parts during flowering period (F, 110 g) of P. ramosissima were submitted for 3 h to water-distillation using a Clevenger type apparatus.
Click to Show/Hide
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor Function |
In all the oils samples the main component was myristicin. In the root oil myristicin was present with 68.5%, in oil from aerial parts during vegetative phase, myristicin was present with 88.9%, while in oil from aerial parts during flowering period this component was present with 91.5%, in the seed oil myristicin was found with 61.1%. It can be seen that myristicin was the most abundant component in all oil samples that we investigated with very high percentage. But, it can also be seen that the season of plant collection influenced the oil characteristics. The highest content of myristicin was present in the oil sample isolated from plants collected during the flowering period (91.5%), than in oil isolated during the vegetative phase.
Click to Show/Hide
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor | Part | Location | NP Content | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Aerial parts: vegetative stage
|
Aerial parts | Montenegro |
NP Content: 0.1 %
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Aerial parts: flowering stage
|
Aerial parts | Montenegro |
NP Content: 0.6 %
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Root: vegetative stage
|
Roots | Montenegro |
NP Content: 4.1 %
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Seed: vegetative stage
|
Seeds | Montenegro |
NP Content: 0.9 %
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||