| The Content Variation of Natural Product Induced by Different Factor(s) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
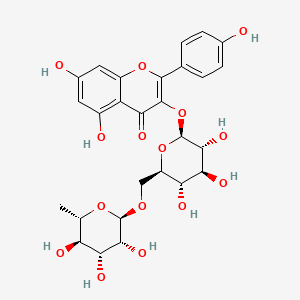
| Species Name: Chelidonium majus | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor Name: Locality Variation; Harvest Time Variation | [1] | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Experiment Detail |
Aerial parts from five populations of Chelidonium majus were collected from the wild at the flowering stage (hereafter referred to as 'wild') for chemical analysis and biological activity testing during May 2019. Ten randomly selected plantlets were also collected from the same five populations in 2019 and planted in an organically certified experimental field of IES (57° 19′ 11.7″ N 25° 19′ 18.8″ E, 115 m altitude). The plot size was 0.8 m2, and the plant spacing was 0.2 × 0.5 m. A year later, aerial parts were collected during the flowering stage from the same populations in the experimental field (hereafter referred to as 'cultivated').
Click to Show/Hide
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor Function |
The total content of alkaloids in aqueous ethanol extracts prepared from cultivated C. majus specimens was higher than that observed in extracts prepared from wild-grown plant material. Chelidonine, sanguinarine, and chelerythrine were the main contributors to the total increase in alkaloid content. The cultivation of C. majus did not significantly affect the total content of flavonol glycosides. The observed differences in the phytochemical compositions of the C. majus extracts resulted in significant increases in the cytotoxic activities of the preparations.
Click to Show/Hide
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor | Part | Location | NP Content | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Locality: wild + Harvesting time: 2019
|
Aerial parts | Latvia |
NP Content: 653.8 ± 377.4 µg/g
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Locality: experimental field + Harvesting time: 2020
|
Aerial parts | Latvia |
NP Content: 600.7 ± 216.4 µg/g
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Species Name: Prunus persica Batsch cv. 'Yuhua No. 2' | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor Name: Low Temperature Treatment; Glycine betaine Treatment | [2] | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Experiment Detail |
Peach fruit (Prunus persica Batsch cv. 'Yuhua No. 2') was hand-harvested at commercial maturity (about 9-12N firmness, 10-12% total soluble solids) from a local orchard in Nanjing, China. The fresh weight of 'Yuhua No. 2' peach is about 215g and the dry weight is about 30g. The fruit shape is round and the diameter size is about 72 mm. The peaches were selected in uniform size and color and absence of any damage. The selected peaches were randomly divided into two groups, each with 360 fruits for 3 replicates. According to our previous study, 10 mmol/LGB was selected as the treatment concentration. Peach fruits were immersed in 10 mmol/LGB solution for 10 min to ensure that GB could be equally distributed on the fruits. The control fruits were soaked in sterile deionized water for 10 min. After treatment, all fruits were air dried about 30 min and stored at 0℃ with a relative humidity of 85-90% for 35 days. Mesocarp samples were collected from 18 fruits on the 7th, 14th, 21th, 28th, 35th day and frozen in liquid nitrogen, then stored at -80℃ until biochemical analysis. Another 18 fruits were removed from 0℃ after 7th, 14th, 21th, 28th, 35th day, and held at 20℃ for three days to simulate shelf condition, and then evaluated CI index, firmness and extractable juice. Each treatment was replicated three times and the experiment was conducted twice with similar results.
Click to Show/Hide
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor Function |
Glycine betaine (GB) treatment enhanced chilling tolerance throughout regulating phenolic and sugar metabolisms in peach fruit during cold storage. The alleviation of chilling injury (CI) by GB treatment may be attributed to enhancement of individual of phenolic compounds and sucrose content, and induce the activities of enzymes related to phenolic and sugar metabolisms.
Click to Show/Hide
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor | Part | Location | NP Content | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Cold storage(days): 0
|
Flesh tissues | Nanjing, China |
NP Content: 0.36 ± 0.02 mg/g fresh weight
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Cold storage(days): 7
|
Flesh tissues | Nanjing, China |
NP Content: 0.45 ± 0.03 mg/g fresh weight
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
10 mmol/L Glycine betaine + Cold storage(days): 7
|
Flesh tissues | Nanjing, China |
NP Content: 0.82 ± 0.05 mg/g fresh weight
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Cold storage(days): 21
|
Flesh tissues | Nanjing, China |
NP Content: 0.52 ± 0.03 mg/g fresh weight
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
10 mmol/L Glycine betaine + Cold storage(days): 10
|
Flesh tissues | Nanjing, China |
NP Content: 0.63 ± 0.03 mg/g fresh weight
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Cold storage(days): 35
|
Flesh tissues | Nanjing, China |
NP Content: 0.26 ± 0.02 mg/g fresh weight
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
10 mmol/L Glycine betaine + Cold storage(days): 35
|
Flesh tissues | Nanjing, China |
NP Content: 0.43 ± 0.03 mg/g fresh weight
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||