| The Content Variation of Natural Product Induced by Different Factor(s) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Species Name: Solanum lycopersicum L.(cv. ACE 55 VF) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor Name: Jasmonic Treatment; Drought Stress Treatment; AMF Inoculation | [1] | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Experiment Detail |
Solanum lycopersicum L. seedlings were grown from commercial seeds (cv. ACE 55 VF). Seeds were surface sterilized by gently shaking them in a 1% NaClO solution for 5 min and rinsed successively 10 times for 5 min in sterile demineralized water. The seeds were pregerminated in seed trays containing autoclaved peat substrate in a climate-controlled chamber (16 h photoperiod at a light intensity of approximately 300 µmol m-2 s-1 photosynthetically active radiation, 23-25 ℃ and 60% Relative Humidity). Ninety-five percent of the seeds germinated at 5 seeds/pot, after one week all the seedlings were showing the apical bud and two cotyledons. Seedlings were then selected for uniformity (plant height and number of leaves) from a large population, and were individually transplanted to 1200 ml pots containing autoclaved soil:sand mix (1:2, v/v). Half of the seedlings received the mycorrhizal treatment as described below. Mycorrhizal colonization of germinated tomato seedlings was induced by transplanting the plants into pots containing autoclaved substrate mixed with inoculum. Mycorrhizal plants (AM): plants were inoculated with 20 ml of Endorize IV commercial inoculum containing Glomus mosseae, Glomus intraradices, Glomus sp., infective units not specified (Biorize, Dijon, France) . Plants were supplied weekly with 20 ml/pot of Long Ashton nutrient solution with half of the content of phosphorus .We attempted to obtain mycorrhizal plants (AM) with size and tissue nutrient content similar to those of non-mycorrhizal plants (NAM) by supplementing NAM plants with more phosphate, since AM symbiosis enhances P uptake and this may alter the plant response to drought . Moreover, the use of plants with similar size allows the detection of drought direct effects not mediated by plant size when working with plants in containers, since unequal plant size can be responsible of differences in soil water depletion and plant transpiration, and consequently plants can be exposed to unequal stress. Thus, not mycorrhizal plants were grown on the same autoclaved substrate, without inoculum material, and supplied weekly with 20 ml/pot of full-strength Long Ashton solution containing 41 ppm of P.All of the seedlings were maintained in a climate-controlled chamber (16 h photoperiod at a light intensity of approximately 370 µmol m-2 s-1 photosynthetically active radiation, 23-25 ℃ and 60% Relative Humidity). One month after inoculation, plants were transferred to 3 L pots filled with the sterile substrate and kept in the climate chamber described above. Plants were watered with tap water and fertilized as indicated above.To induce an almost natural, reversible drought stress, thus allowing the plant enough time to acclimate, irrigation was stopped 24 h before measurements were taken. These treatments resulted in moderate water stress (lower than -2 MPa). Two stems, each containing 5-6 mature leaves, and one apical stem were selected in each plant for jasmonic acid (JA) treatment. In the JA treatment, the abaxial and adaxial surfaces of six leaves from two different branches were sprayed until runoff with a solution of 0.5 mM of JA (Sigma-Aldrich, St. Louis, MO, USA). The solution was prepared by dissolving JA in acetone and them diluting this mixture with water to 1 mM. Approximately 1.5 ml of JA solution, corresponding to 0.157 mg of JA, were sprayed onto each single leaf. JA-treated leaves were isolated with a protective plastic that prevented the rest of the plant from being treated. The plastic was removed after the spray has dried. Treatments were coordinated so that all plants were tested approximately 14-15 h after JA application to avoid any diurnal effects.Two months after inoculation, gas exchange and VOCs measurements were performed.
Click to Show/Hide
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor Function |
Root colonization by AM fungi favoured the leaf production of essential isoprenoids rather than nonessential ones, especially under drought stress conditions or after JA application.
Click to Show/Hide
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor | Part | Location | NP Content | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Leaf volatile emission: No jasmonic + Well-watered + Non-AMF inoculation
|
Leaves | NA |
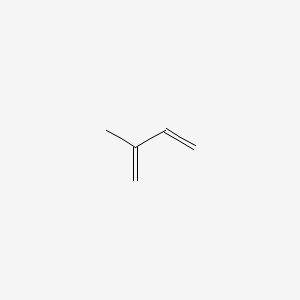
NP Content: 19.6 ± 7.9 ng m-2 s-1
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Leaf volatile emission: No jasmonic + Well-watered + Glomus mosseae, Glomus intraradices and Glomus sp. (AMF) inoculation
|
Leaves | NA |
NP Content: 3.4 ± 2.1 ng m-2 s-1
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Leaf volatile emission: No jasmonic + Drought + Non-AMF inoculation
|
Leaves | NA |
NP Content: 24.7 ± 5.9 ng m-2 s-1
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Leaf volatile emission: No jasmonic + Drought + AMF inoculation
|
Leaves | NA |
NP Content: 32.0 ± 7.4 ng m-2 s-1
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Leaf volatile emission: Jasmonic + Well-watered + Non-AMF inoculation
|
Leaves | NA |
NP Content: 2.5 ± 1.6 ng m-2 s-1
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Leaf volatile emission: Jasmonic + Well-watered + AMF inoculation
|
Leaves | NA |
NP Content: 18.0 ± 10.8 ng m-2 s-1
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Leaf volatile emission: Jasmonic + Drought + Non-AMF inoculation
|
Leaves | NA |
NP Content: 2.0 ± 2.0 ng m-2 s-1
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Leaf volatile emission: Jasmonic + Drought + AMF inoculation
|
Leaves | NA |
NP Content: 4.8 ± 4.8 ng m-2 s-1
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| References | ||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 1 | AM fungi root colonization increases the production of essential isoprenoids vs. nonessential isoprenoids especially under drought stress conditions or after jasmonic acid application | |||||||||||||||||||||||