| The Content Variation of Natural Product Induced by Different Factor(s) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Species Name: Chelidonium majus | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor Name: Locality Variation; Harvest Time Variation | [1] | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Experiment Detail |
Aerial parts from five populations of Chelidonium majus were collected from the wild at the flowering stage (hereafter referred to as 'wild') for chemical analysis and biological activity testing during May 2019. Ten randomly selected plantlets were also collected from the same five populations in 2019 and planted in an organically certified experimental field of IES (57° 19′ 11.7″ N 25° 19′ 18.8″ E, 115 m altitude). The plot size was 0.8 m2, and the plant spacing was 0.2 × 0.5 m. A year later, aerial parts were collected during the flowering stage from the same populations in the experimental field (hereafter referred to as 'cultivated').
Click to Show/Hide
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor Function |
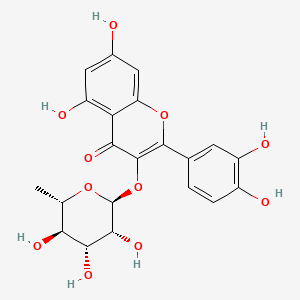
The total content of alkaloids in aqueous ethanol extracts prepared from cultivated C. majus specimens was higher than that observed in extracts prepared from wild-grown plant material. Chelidonine, sanguinarine, and chelerythrine were the main contributors to the total increase in alkaloid content. The cultivation of C. majus did not significantly affect the total content of flavonol glycosides. The observed differences in the phytochemical compositions of the C. majus extracts resulted in significant increases in the cytotoxic activities of the preparations.
Click to Show/Hide
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor | Part | Location | NP Content | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Locality: wild + Harvesting time: 2019
|
Aerial parts | Latvia |
NP Content: 1.4 ± 1.2 µg/g
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Locality: experimental field + Harvesting time: 2020
|
Aerial parts | Latvia |
NP Content: 2.0 ± 0.9 µg/g
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Species Name: Crocus sativus L. (saffron) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor Name: AMF Inoculation; Harvest Time Variation | [2] | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Experiment Detail |
AMF Inoculation in Pot : Saffron corms with horizontal diameters of 1.3 to 2.8 cm were sown in pots (4 L; 1 corm per pot) in the last ten days of August 2016. Pots were filled with sterile quartz sand (3 L per pot) on a layer of sterilized expanded clay (1 L per pot). Corms were treated with two inocula (MycAgro Lab, Breteniere, FR), one composed of a single fungus Rhizophagus intraradices (Ri) and one of R. intraradices and Funneliformis mosseae (Ri + Fm). Ten grams of each inoculum were placed under each corm in order to guarantee the contact between the inoculum and the roots and therefore to favor the symbiosis between AMF and roots. Saffron corms used as controls were not inoculated (AMF-). Corms were not treated against fungal pathogens. A randomized block design was used with a total of 48 pots displayed in two experimental plot units (24 pots per unit) and three treatments (8 pots per treatment). Cultivation lasted for one cycle (August 2016-April 2017) in a heated glasshouse of the Department of Agricultural Forest and Food Sciences (DISAFA) of the University of Torino (Italy, 45° 06′ 23.21″ N Lat, 7° 57′ 82.8″ E Long; 293 m a.s.l.), with an average temperature of 22 ℃ during the day and 16 ℃ in the night. Irrigation water (pH 7.4, EC 505 µS cm) was added weekly (250 mL per pot) with a drip system. The corms were fertilized by fertigation (VIGORFLOR, AL.FE. srl, MN, Italy) every two weeks starting from the emergence of the spate, in quantities of 1.5 g/L of water. No flowering occurred because of the small size of the corms.AMF Inoculation in Open Field : Saffron corms with horizontal diameters of 2.5 to 3.5 cm were planted in the last ten days of August 2016 in two Alpine experimental sites located in the municipality of Morgex (45° 45′ 35″ N; 7° 02′ 37.3″ E; 1000 m a.s.l.) and Saint Cristophe (45° 45′ 06″ N; 7° 20′ 37″ E; 700 m a.s.l.) in Italy and cultivation lasted for two cycles (2016-2017 and 2017-2018). Both sites were cultivated with saffron for at least the previous three years. Before starting the experiment both fields were milled. To assess the effects of AMF inocula on saffron cultivation and production, the same treatments used in the pot trial were applied (Ri, Ri + Fm or AMF-). A randomized block design was used, with three experimental plot units (blocks). Each plot unit consisted of 56 corms, planted in a 1.44 m2 area (39 corms m-2). Inter-row planting distance was of 7 cm, while between-row distance was 25 cm. Plots were separated from each other with at least 4 m distance. Before planting, 10 g of inoculum was placed under the corms to ensure contact between plant and the treatment. Irrigation was provided when needed and hand weeding control was conducted during cultivation, while no preplanting fertilization, tillage, or treatments against pathogens were applied. The two Alpine sites were characterized by semicontinental climate, with a long and cold winter . In general, both sites had a sandy-loam texture according to the USDA classification and similar chemical characteristics.
Click to Show/Hide
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor Function |
The inoculum composed by R. intraradices and F. mosseae was particularly effective in increasing flower production and saffron yield, while R. intraradices alone increased the content of some bioactive compounds-picrocrocin, quercitrin, crocin II-as well as antioxidant activity.
Click to Show/Hide
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor | Part | Location | NP Content | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Harvesting time: 2016-2017
|
Powdered saffrons | Italy |
NP Content: 22 mg/100g dry weight
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Harvesting time: 2017-2018
|
Powdered saffrons | Italy |
NP Content: 16 mg/100g dry weight
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Rhizophagus intraradices and Funneliformis mosseae inoculation
|
Powdered saffrons | Italy |
NP Content: 17 mg/100g dry weight
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Rhizophagus intraradices inoculation
|
Powdered saffrons | Italy |
NP Content: 22 mg/100g dry weight
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Non-AMF inoculation (Control)
|
Powdered saffrons | Italy |
NP Content: 18 mg/100g dry weight
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||