| The Content Variation of Natural Product Induced by Different Factor(s) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Species Name: Satureja parnassica ssp. parnassica | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor Name: Month Variation; Developmental Stage Variation | [1] | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Experiment Detail |
Fresh plant materials were obtained in 2004 and 2005. S. thymbra 1(vegetative stage: just before flowering, date: June 7, 2004, location: Mt. Immitos, altitude(m): 350); S. thymbra 2(vegetative stage: full flowering, date: July 7, 2004, location: Mt. Immitos, altitude(m): 350); S. thymbra 3(vegetative stage: after flowering, date: Aug 7, 2004, location: Mt. Immitos, altitude(m): 350); S. thymbra 4(vegetative stage: fruiting, date: Sept 7, 2004, location: Mt. Immitos, altitude(m): 350); S. thymbra 5(vegetative stage: fruiting, date: Nov 7, 2004, location: Mt. Immitos, altitude(m): 350); S. thymbra 6(vegetative stage: fruiting, date: Feb 7, 2005, location: Mt. Immitos, altitude(m): 350); S. thymbra 7(vegetative stage: before flowering, date: May 7, 2005, location: Mt. Immitos, altitude(m): 350); S. parnassica 8(vegetative stage: before flowering, date: June 16, 2004, location: Mt. Parnon, altitude(m): 1800); S. parnassica 9(vegetative stage: just before flowering, date: July 16, 2004, location: Mt. Parnon, altitude(m): 1800); S. parnassica 10(vegetative stage: full flowering, date: Aug 16, 2004, location: Mt. Parnon, altitude(m): 1800); S. parnassica 11(vegetative stage: after flowering, date: Sept 16, 2004, location: Mt. Parnon, altitude(m): 1800).
Click to Show/Hide
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor Function |
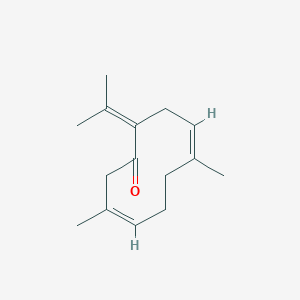
It is evident that the phytochemical content of the essential oils for both Satureja species varied greatly, depending on the period examined, and showed large prevalence of phenolic content. It must also be pointed out that regardless of the vegetative stage of the plant collected, the sum of the two isomeric phenol monoterpenes (carvacrol and thymol) and their biosynthetic monoterpene precursors p-cymene and gamma-terpinene represented always the bulk of each essential oil (~76%). More specificallysfor both species-during their premature vegetative stage, gamma-terpinene constitutes the major component of their essential oils. The approach of the flowering period results in the simultaneous gradual diminishment of monoterpene precursors and the prevalence of their phenolic metabolites. Thus, essential oils obtained from plants collected during the 'just before their flowering' stage contain thymol as their major component, which constitutes 27.88 and 38.51% of the total oil content for S. thymbra and S. parnassica, respectively. On the other hand, during their full flowering period carvacrol prevails as the major component, accounting for 39.10% for S. thymbra and for 34.61% for S. parnassica. The end of the flowering stage delineates a sharp decrease of carvacrol levels and the predominance of thymol as the major component of the essential oils. A few months later, as the premature vegetative stage approached, the level of gamma-terpinene was restored. The content of p-cymenesthe other major monoterpene precursor-fluctuated seasonally in a manner similar to that shown by gamma-terpinene. Other monoterpene hydrocarbons such as myrcene and alpha-terpinene were also detected in smaller quantities, whereas various monoterpene alcohols such as linalool, borneol, and terpin-4-ol were found mainly in the oils obtained after the flowering stage. Finally, it is notable that the oils obtained during the just before the full flowering period contain beta-caryophyllene as one of their major components.
Click to Show/Hide
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor | Part | Location | NP Content | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Harvesting time: just before flowering satge; 16-June-2004
|
Leaves and stems | Mt. Parnon, Peloponnese |
NP Content: 0.35 %
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| References | ||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 1 | Characterization of the essential oil volatiles of Satureja thymbra and Satureja parnassica: influence of harvesting time and antimicrobial activity | |||||||||||||||||||||||