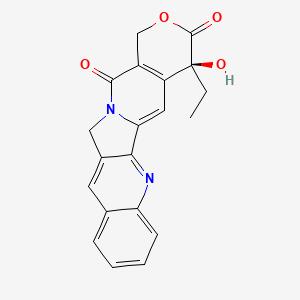
| The Content Variation of Natural Product Induced by Different Factor(s) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Species Name: Alternaria alternata isolate MDF1.2 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor Name: PDB medium | [1] | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Experiment Detail |
Endophytic fungi were isolated from fruits of M. dentata (Icacinaceae; sub-order Asterid). The species is a slow growing climber and is sparsely distributed in the Western Ghats, India. For each pure isolate, single hyphal tips were incubated in 250 ml conical flasks containing 50 ml of pre-sterilized PDB. Flasks were agitated at 200 rpm on a rotary shaker at 28 ℃ for 4 days.
Click to Show/Hide
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor | Part | Location | NP Content | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
PDB medium (28℃ + 6 days + PH 6.5)
|
Fruits | Ghats, India |
NP Content: 73.9 µg/g dry weight
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Species Name: Fomitopsis sp. MDF11 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor Name: PDB medium | [1] | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Experiment Detail |
Endophytic fungi were isolated from fruits of M. dentata (Icacinaceae; sub-order Asterid). The species is a slow growing climber and is sparsely distributed in the Western Ghats, India. For each pure isolate, single hyphal tips were incubated in 250 ml conical flasks containing 50 ml of pre-sterilized PDB. Flasks were agitated at 200 rpm on a rotary shaker at 28 ℃ for 4 days.
Click to Show/Hide
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor | Part | Location | NP Content | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
PDB medium (28℃ + 6 days + PH 6.5)
|
Fruits | Ghats, India |
NP Content: 55.49 µg/g dry weight
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Species Name: Fusarium solani culture-collection MTCC:9667 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor Name: PDA medium | [2] | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Experiment Detail |
The endophytic fungal strains were isolated from trees of the species A. dimidiata E. Mey. ex Arn (Icacinaceae) growing in the Western Ghats region, a mega-bio-diversity hot spot in India. The location coordinates of Periya, the collection locality are 11° 50′ 0″ North, 75° 50′ 0″ East. For each pure isolate, single hyphal tips were incubated in 250 ml conical flasks containing 50 ml of pre-sterilized PDA broth. Flasks were agitated at 200 rpm on a rotary shaker at 28℃ for 4 days.
Click to Show/Hide
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor | Part | Location | NP Content | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
PDA medium (28℃ + 4 days)
|
Leaves; Stem bark | Ghats, India |
NP Content: 37 µg/100 g
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Species Name: Fusarium solani culture-collection MTCC:9668 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor Name: PDA medium | [2] | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Experiment Detail |
The endophytic fungal strains were isolated from trees of the species A. dimidiata E. Mey. ex Arn (Icacinaceae) growing in the Western Ghats region, a mega-bio-diversity hot spot in India. The location coordinates of Periya, the collection locality are 11° 50′ 0″ North, 75° 50′ 0″ East. For each pure isolate, single hyphal tips were incubated in 250 ml conical flasks containing 50 ml of pre-sterilized PDA broth. Flasks were agitated at 200 rpm on a rotary shaker at 28℃ for 4 days.
Click to Show/Hide
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor | Part | Location | NP Content | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
PDA medium (28℃ + 4 days)
|
Leaves; Stem bark | Ghats, India |
NP Content: 53.6 µg/100 g
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Species Name: Fusarium solani isolate S19 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor Name: PDA medium | [3] | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Experiment Detail |
Samples of C. acuminata were collected in April from the Botanical Garden, South of Guiyang, China. Small terminal, limbs (1x25 cm) from one to two years old were harvested and stored in a sealed plastic bag at room temperature and processed within 2 days. The fungi were statically cultured for 10 days at 28 ℃ in petri dishes. They were incubated into 1000 mL flasks containing 500 mL of PDA broth. After 4 days of incubation at 28 ℃ on rotary shaker at 150 rotations per minute (rpm), mycelia and broth were separated by filtration.
Click to Show/Hide
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor | Part | Location | NP Content | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
PDA medium (28℃ + 7 days)
|
Stems | Guiyang, China. |
NP Content: 40 ± 5 µg/g dry weight mycelia
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Species Name: Phomopsis sp. MDF3(1) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor Name: PDB medium | [1] | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Experiment Detail |
Endophytic fungi were isolated from fruits of M. dentata (Icacinaceae; sub-order Asterid). The species is a slow growing climber and is sparsely distributed in the Western Ghats, India. For each pure isolate, single hyphal tips were incubated in 250 ml conical flasks containing 50 ml of pre-sterilized PDB. Flasks were agitated at 200 rpm on a rotary shaker at 28 ℃ for 4 days.
Click to Show/Hide
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor | Part | Location | NP Content | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
PDB medium (28℃ + 6 days + PH 6.5)
|
Fruits | Ghats, India |
NP Content: 42.06 µg/g dry weight
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||