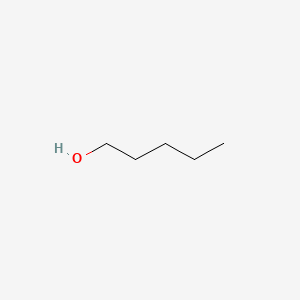
| The Content Variation of Natural Product Induced by Different Factor(s) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Species Name: phaseolorum sp. PR4 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor Name: PDB medium | [1] | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Experiment Detail |
PR4 was isolated as an endophyte from the rhizome of Picrorhiza kurroa. Picrorhiza kurroa Royle ex. Benth (Plantaginaceae) is a perennial herb endemic to the north western alpine Himalayas. The endophyte PR4 was grown on PDA and in PDB at 26 ℃ for 15 days with constant shaking at 200 rpm in the latter case.
Click to Show/Hide
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Mechanism |
The two candidate NR-PKSs (PKS_3671 and PKS_4063) show differences in their domain organizations. PKS_3671 possesses two ACP-domains. Apart from that, only PKS_3671 contains a SAT-domain . These domains provide the first building block in the polyketide assembly, which usually is different from the extender unit malonyl-CoA (also known as the 'starter unit effect'). The ACA-synthesis however is believed to involve merely malonyl-CoA molecules. Even though the ACA-producing PKSs MdpG, ACAS, EncA, AptA and ClaG contain SAT-domains, an amino acid sequence alignment of these domains revealed that they all lack the active-site cysteine in the GXCXG motif and therefore most likely have no acyl transferase activity. Instead, all malonate building blocks are assumed to be loaded by the MAT. Under this aspect, the SAT-domain of PKS_3671 (that includes the correct GXCXG motif) likely incorporates a starter unit different from malonyl-CoA indicating that this enzyme is not involved in the biosynthesis of ACA. Therefore, the ACA-synthesizing PKS in C. asteris would rather be PKS_4063 that misses the SAT-domain .In the monodictyphenone and cladofulvin pathways, the cluster-encoded gene products MdpH and ClaH are crucial enzymes pushing the biosynthesis towards emodin. These EthD-domain-containing enzymes are suggested to catalyze the decarboxylation of ACA (3) into atrochrysone (4). Surprisingly, no such EthD-domain is encoded in the whole C. asteris genome. On the other hand, four genes directly attached to the putative ACA-synthase-coding gene pks_4063 show high similarity to genes of non-investigated PKS clusters in other fungi , which indicates an involvement in tailoring reactions of the respective polyketide pathways. According to InterProScan and BLASTp analyses, the genes sky_4060-62 encode a dehydratase and two dehydrogenases potentially catalyzing the multistep conversion of ACA (3) into emodin (1). Gene sky_4059 codes for a monooxygenase that putatively can connect two emodin molecules to the final product skyrin (2) in the style of the monooxygenase ClaM involved in the dimerization of the bisanthraquinone cladofulvin. Thus, the presence of these genes in the gene cluster gives further support to the hypothesis that PKS_4063 is the ACA-synthase in C. asteris. Mutational studies will be done in order to confirm these assumptions after a gene transfer system for this strain has been developed.
Click to Show/Hide
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor | Part | Location | NP Content | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
PDB medium (26℃ + 15 days)
|
Rhizomes | Himalayas |
NP Content: 0.609 % Relative area
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor Name: PDA medium | [1] | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Experiment Detail |
PR4 was isolated as an endophyte from the rhizome of Picrorhiza kurroa. Picrorhiza kurroa Royle ex. Benth (Plantaginaceae) is a perennial herb endemic to the north western alpine Himalayas. The endophyte PR4 was grown on PDA and in PDB at 26 ℃ for 15 days with constant shaking at 200 rpm in the latter case.
Click to Show/Hide
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Mechanism |
The two candidate NR-PKSs (PKS_3671 and PKS_4063) show differences in their domain organizations. PKS_3671 possesses two ACP-domains. Apart from that, only PKS_3671 contains a SAT-domain . These domains provide the first building block in the polyketide assembly, which usually is different from the extender unit malonyl-CoA (also known as the 'starter unit effect'). The ACA-synthesis however is believed to involve merely malonyl-CoA molecules. Even though the ACA-producing PKSs MdpG, ACAS, EncA, AptA and ClaG contain SAT-domains, an amino acid sequence alignment of these domains revealed that they all lack the active-site cysteine in the GXCXG motif and therefore most likely have no acyl transferase activity. Instead, all malonate building blocks are assumed to be loaded by the MAT. Under this aspect, the SAT-domain of PKS_3671 (that includes the correct GXCXG motif) likely incorporates a starter unit different from malonyl-CoA indicating that this enzyme is not involved in the biosynthesis of ACA. Therefore, the ACA-synthesizing PKS in C. asteris would rather be PKS_4063 that misses the SAT-domain .In the monodictyphenone and cladofulvin pathways, the cluster-encoded gene products MdpH and ClaH are crucial enzymes pushing the biosynthesis towards emodin. These EthD-domain-containing enzymes are suggested to catalyze the decarboxylation of ACA (3) into atrochrysone (4). Surprisingly, no such EthD-domain is encoded in the whole C. asteris genome. On the other hand, four genes directly attached to the putative ACA-synthase-coding gene pks_4063 show high similarity to genes of non-investigated PKS clusters in other fungi , which indicates an involvement in tailoring reactions of the respective polyketide pathways. According to InterProScan and BLASTp analyses, the genes sky_4060-62 encode a dehydratase and two dehydrogenases potentially catalyzing the multistep conversion of ACA (3) into emodin (1). Gene sky_4059 codes for a monooxygenase that putatively can connect two emodin molecules to the final product skyrin (2) in the style of the monooxygenase ClaM involved in the dimerization of the bisanthraquinone cladofulvin. Thus, the presence of these genes in the gene cluster gives further support to the hypothesis that PKS_4063 is the ACA-synthase in C. asteris. Mutational studies will be done in order to confirm these assumptions after a gene transfer system for this strain has been developed.
Click to Show/Hide
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor | Part | Location | NP Content | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
PDA medium (26℃)
|
Rhizomes | Himalayas |
NP Content: 0.411 % Relative area
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| References | ||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 1 | An endophyte of Picrorhiza kurroa Royle ex. Benth, producing menthol, phenylethyl alcohol and 3-hydroxypropionic acid, and other volatile organic compounds | |||||||||||||||||||||||