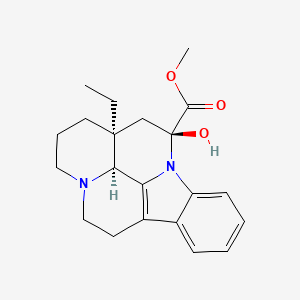
| Synonyms |
vincamine; 1617-90-9; Pervincamine; Devincan; Angiopac; Minorine; Vincamidol; Novicet; Arteriovinca; Equipur; Monorin; (+)-Vincamine; Methyl vincaminate; Anasclerol; Oxygeron; Vincafor; Perval; Vincimax; Vincasaunier; Decincan; Devinkan; Tripervan; Vincadar; Vincafolina; Vincagil; Vincamin; Vincapront; Vinkametrin; UNII-996XVD0JHT; Minorin; Pervone; Vincachron; Vincapan; Vinodrel retard; Ocu-vinc; Vinca-Ecobi; Vinca-Minor; Anasclerol (base); 14,15-Dihydro-14-hydroxyeburnamenine-14-carboxylic acid methyl ester; 996XVD0JHT; CHEBI:9985; MFCD00078054; Eburnamenine-14-carboxylic acid, 14,15-dihydro-14-hydroxy-, methyl ester, (3alpha,14beta,16alpha)-; DSSTox_CID_20134; DSSTox_RID_79448; DSSTox_GSID_40134; Vraap; Vincamina [DCIT]; Vincaminum; Vincamina; Alkaloid obtained from Vinca minor; Vincaminum [INN-Latin]; methyl 14beta-hydroxy-14,15-dihydro-3alpha,16alpha-eburnamenine-14alpha-carboxylate; Vincamine [INN:BAN:DCF]; HSDB 7150; EINECS 216-576-3; NSC 91998; Oxybral; Cetal retard; NSC-91998; (3?,14?,16?)-14,15-Dihydro-14-hydroxyeburnamenine-14-carboxylic acid methyl ester; Vincamine (FP); NCGC00094824-01; Vincamine (INN); Prestwick_495; CAS-1617-90-9; Cetal retard (TN); Vincamine, 98%; Prestwick0_000271; Prestwick1_000271; Prestwick2_000271; Prestwick3_000271; Eburnamenine-14-carboxylic acid, 14,15-dihydro-14-hydroxy-, methyl ester, (3alpha,14beta,16.); BSPBio_000142; GTPL349; MLS002154249; SCHEMBL147179; SPBio_002361; BPBio1_000158; CHEMBL1165342; DTXSID9040134; BCBcMAP01_000080; HMS1568H04; HMS2095H04; HMS2268C20; HMS3712H04; AMY39091; BCP05837; HY-B1021; ZINC1069082; Tox21_111342; Tox21_301968; s3891; AKOS015896471; Tox21_111342_1; CCG-208544; CS-4536; DB13374; KS-5179; methyl (3alpha,14beta,16alpha)-14-hydroxy-14,15-dihydroeburnamenine-14-carboxylate; SMP1_000314; Vincamine, analytical reference material; NCGC00184983-01; NCGC00184983-03; NCGC00255542-01; AC-22625; SMR000112509; C09251; D08677; J10447; 617V909; A851579; Q416225; Q-100193; BRD-K40902647-001-03-7; Eburnamenine-14-carboxylic acid, 14,15-dihydro-14-hydroxy-, methylester, (3a,14b,16a)-; (3alpha,14beta,16alpha)-14,15-Dihydro-14-hydroxyeburnamenine-14-carboxylic acidmethyl ester; Eburnamenine-14-carboxylic acid, 14,15-dihydro-14-hydroxy-,methyl ester, (3alpha,14beta,16alpha)-; methyl (15S,17S,19S)-15-ethyl-17-hydroxy-1,11-diazapentacyclo[9.6.2.0?,?.0?,??.0??,??]nonadeca-2,4,6,8(18)-tetraene-17-carboxylate; methyl (15S,17S,19S)-15-ethyl-17-hydroxy-1,11-diazapentacyclo[9.6.2.0^{2,7}.0^{8,18}.0^{15,19}]nonadeca-2,4,6,8(18)-tetraene-17-carboxylate; methyl (15S,17S,19S)-15-ethyl-17-hydroxy-1,11-diazapentacyclo[9.6.2.02,7.08,18.015,19]nonadeca-2,4,6,8(18)-tetraene-17-carboxylate; Methyl (41S,12S,13aS)-13a-ethyl-12-hydroxy-2,3,41,5,6,12,13,13a-octahydro-1H-indolo[3,2,1-de]pyrido[3,2,1-ij][1,5]naphthyridine-12-carboxylate
Click to Show/Hide
|