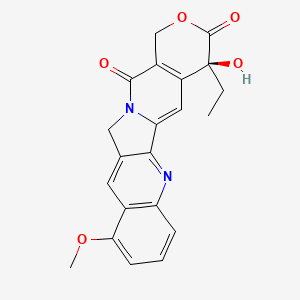
| The Content Variation of Natural Product Induced by Different Factor(s) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Species Name: Fusarium solani culture-collection MTCC:9667 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor Name: PDA medium | [1] | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Experiment Detail |
The endophytic fungal strains were isolated from trees of the species A. dimidiata E. Mey. ex Arn (Icacinaceae) growing in the Western Ghats region, a mega-bio-diversity hot spot in India. The location coordinates of Periya, the collection locality are 11° 50′ 0″ North, 75° 50′ 0″ East. For each pure isolate, single hyphal tips were incubated in 250 ml conical flasks containing 50 ml of pre-sterilized PDA broth. Flasks were agitated at 200 rpm on a rotary shaker at 28℃ for 4 days.
Click to Show/Hide
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor | Part | Location | NP Content | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
PDA medium (28℃ + 4 days)
|
Leaves; Stem bark | Ghats, India |
NP Content: 38.3 µg/100 g
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Species Name: Fusarium solani culture-collection MTCC:9668 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor Name: PDA medium | [1] | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Experiment Detail |
The endophytic fungal strains were isolated from trees of the species A. dimidiata E. Mey. ex Arn (Icacinaceae) growing in the Western Ghats region, a mega-bio-diversity hot spot in India. The location coordinates of Periya, the collection locality are 11° 50′ 0″ North, 75° 50′ 0″ East. For each pure isolate, single hyphal tips were incubated in 250 ml conical flasks containing 50 ml of pre-sterilized PDA broth. Flasks were agitated at 200 rpm on a rotary shaker at 28℃ for 4 days.
Click to Show/Hide
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor | Part | Location | NP Content | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
PDA medium (28℃ + 4 days)
|
Leaves; Stem bark | Ghats, India |
NP Content: 44.9 µg/100 g
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| References | ||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 1 | Endophytic fungal strains of Fusarium solani, from Apodytes dimidiata E. Mey. ex Arn (Icacinaceae) produce camptothecin, 10-hydroxycamptothecin and 9-methoxycamptothecin | |||||||||||||||||||||||