| Synonyms |
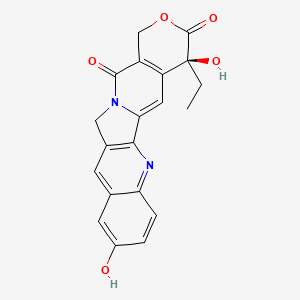
10-Hydroxycamptothecin; 19685-09-7; (S)-10-Hydroxycamptothecin; Hydroxycamptothecin; 10-hydroxycamptothecine; 10-Hydroxy camptothecin; Hydroxycamptothecine; (S)-4-Ethyl-4,9-dihydroxy-1H-pyrano[3',4':6,7]indolizino[1,2-b]quinoline-3,14(4H,12H)-dione; Camptothecin, hydroxy-; 10-Hydroxy-Camptothecin; 10-HCPT; (4S)-4-Ethyl-4,9-dihydroxy-1H-pyrano[3',4':6,7]indolizino[1,2-b]quinoline-3,14(4H,12H)-dione; UNII-9Z01632KRV; NSC107124; CHEMBL273862; CHEBI:81395; 9Z01632KRV; Hydroxy camptothecine; MFCD02093100; NSC-107124; (+)-(S)-10-HYDROXYCAMPTOTHECIN; (S)-4-ethyl-4,9-dihydroxy-1,12-dihydro-14H-pyrano[3',4':6,7]indolizino[1,2-b]quinoline-3,14(4H)-dione; C20H16N2O5; Camptothecin, 10-hydroxy-; Camptothecine, 10-hydroxy-; (20S)-4-Ethyl-4,9-dihydroxy-1,12-dihydro-4H-2-oxa-6,12a-diaza-dibenzo[b,h]fluorene-3,13-dione; NSC 107124; 10-Hydroxycamptochecin; (S)-10-Hydroxycamptothecin hydrate; (20S)-10-Hydroxycamptothecin; CAMPTOTHECIN, 10-HYDROXY; 10-Hydroxy-CPT; Spectrum_001639; SpecPlus_000763; Spectrum2_001660; Spectrum3_001621; Spectrum4_001815; Spectrum5_000549; ethyl(dihydroxy)[?]dione; SCHEMBL25875; BSPBio_003281; Irinotecan related compound a; KBioGR_002454; KBioSS_002119; 1H-Pyrano(3',4':6,7)indolizino(1,2-b)quinoline-3,14(4H,12H)-dione, 4-ethyl-4,9-dihydroxy-, hydrate, (S)-; DivK1c_006859; SPECTRUM1504123; SPBio_001819; KBio1_001803; KBio2_002119; KBio2_004687; KBio2_007255; KBio3_002501; DTXSID00941444; EX-A988; AOB33756; BCP01385; HY-N0095; ZINC3979155; Camptothecine, 10-hydroxy- (8CI); 2136AH; BDBM50008922; CCG-38770; s2423; s3898; AKOS015919293; AC-5502; ACN-035239; BCP9000058; CS-5193; DB12385; NCGC00095986-01; NCGC00095986-02; NCGC00095986-03; NCGC00095986-04; NCGC00178165-01; AC-13221; AS-13196; H577; NCI60_000173; SY010687; H1463; N2591; 85H097; A25382; C17939; SR-05000002620; Q-100241; SR-05000002620-1; BRD-K63784565-001-02-1; BRD-K63784565-001-03-9; Q27155328; 4-Ethyl-4,9-dihydroxy-1,12-dihydro-4H-2-oxa-6,12a-diaza-dibenzo[b,h]fluorene-3,13-dione; (19S)-19-ethyl-7,19-dihydroxy-17-oxa-3,13-diazapentacyclo[11.8.0.02,11.04,9.015,20]henicosa-1(21),2(11),3,5,7,9,15(20)-heptaene-14,18-dione; (alphaS)-alpha,2-Dihydroxy-alpha-ethyl-8-(hydroxymethyl)-9-oxo-9,11-dihydroindolizino[1,2-b]quinoline-7-acetic acid 7,8-lactone; (S)-10-Hydroxycamptothecin;-;(+/-)-4-ethyl-4,9-dihydroxy-1h-pyrano[3',4':6,7]indolizino[1,2-b]quinoline-3,14(4h,12h)-dione; (S)-4-Ethyl-4,9-dihydroxy-1H-pyrano[3 inverted exclamation mark ,4 inverted exclamation mark :6,7]indolizino[1,2-b]quinoline-3,14-(4H,12H)-dione; (S)-4-Ethyl-4,9-dihydroxy-1H-pyrano[3',4':6,7]indolizino[1,2-b]quinoline-3,14(4H,12H)-dione ((S)-10-Hydroxycamptothecin); (S)-4-Ethyl-4,9-dihydroxy-1H-pyrano[3',4':6,7]indolizino[1,2-b]quinoline-3,14-(4H,12H)-dione; 1H-Pyrano(3',4':6,7)indolizino(1,2-b)quinoline-3,14(4H,12H)-dione-,4-ethyl-4,9-dihydroxy-, (S)- (9CI); 1H-Pyrano[3',7]indolizino[1,2-b]quinoline-3,14(4H,12H)-dione-,4-ethyl-4,9-dihydroxy-, (S)-; 4-Ethyl-4,10-dihydroxy-1,12-dihydro-4H-2-oxa-6,12a-diaza-dibenzo[b,h]fluorene-3,13-dione; 4-Ethyl-4,9-dihydroxy-1,12-dihydro-4H-2-oxa-6,12a-diaza-dibenzo[b,h]fluorene-3,13-dione (10-hydroxycamptothecin)
Click to Show/Hide
|