| The Content Variation of Natural Product Induced by Different Factor(s) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Species Name: Aeromicrobium sp. strain LGMB491 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor Name: SG medium | [1] | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Experiment Detail |
V. divergens leaves with no marks or injuries were collected from 21 plants located in the Pantanal sul-mato-grossense/Brazil, specifically in two regions of the Pantanal of Miranda, Abobral (19° 30′ 09.5″ S, 57° 02′ 32.2″ W) and Sao Bento (19° 28′ 53.9″ S, 57° 02′ 36.9″ W). A large-scale fermentation (10 L) of strain LGMB491 was performed using SG culture medium at 36 ℃ for 10 days.
Click to Show/Hide
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor | Part | Location | NP Content | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
SG medium (36℃ + 10 days)
|
Leaves | Pantanal, Brazil |
NP Content: 50.1 mg
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Species Name: Streptomyces sp. SUK10 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor Name: NB medium | [2] | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Experiment Detail |
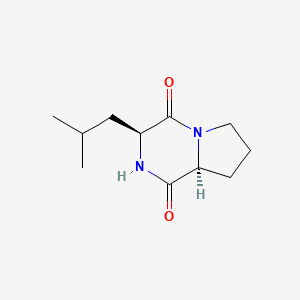
In this work, the DKP gancidin W (GW) was isolated from the crude extract of an endophytic Streptomyces strain designated as SUK10, which was obtained from the bark of the Shorea ovalis tree. Nutrient broth (NB) (Sigma-Aldrich, Kuala Lumpur, Malaysia) was prepared at pH 7.0. To produce sufficient amount of isolates for extraction, five blocks of 5x5 mm International Streptomyces Project 2 agar enriched with Streptomyces SUK10 were cut and inoculated into 400 mL of autoclaved NB at pH 7.0 in a 1,000 mL conical flask. Fermentation was carried out using an orbital shaker at 28℃ at rotation rate of 200 rpm for 21 days.
Click to Show/Hide
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor | Part | Location | NP Content | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
NB medium (28℃ + 21 days)
|
Bark | Malaysian tropical forests, Malaysia |
NP Content: 16.2 mg
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||