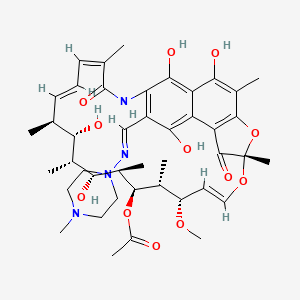
| Synonyms |
Rifampicin; rifampin; Rifadin; Rimactane; Rimactan; Rifamycin AMP; 13292-46-1; Rifaldazine; Rifaprodin; Rifampicinum; Rifoldin; Riforal; Tubocin; Rifa; Archidyn; Rifadine; Rifaldin; Rifoldine; Rimactizid; Rifagen; Rimazid; Rifampicin SV; L-5103 Lepetit; R/AMP; Abrifam; Arficin; Benemicin; Doloresum; Eremfat; Fenampicin; Rifaldazin; Rifamor; Rifinah; Rifobac; Rimactazid; Sinerdol; Arzide; Rifcin; Rifam; Dione 21-acetate; Rifampicine [French]; Rifampicina; Rifampicinum [INN-Latin]; Ba 41166/E; Rifampicina [INN-Spanish]; 3-(((4-Methyl-1-piperazinyl)imino)methyl)rifamycin SV; CCRIS 551; Rifadin I.V.; HSDB 3181; C43H58N4O12; UNII-VJT6J7R4TR; BA-41166E; NSC113926; VJT6J7R4TR; NSC-113926; Rifampicine; Rifamycin, 3-(((4-methyl-1-piperazinyl)imino)methyl)-; RFP; CHEBI:28077; 3-(4-Methylpiperazinyliminomethyl)-rifamycin SV; 8-(4-Methylpiperazinyliminomethyl) rifamycin SV; L-5103; (2S,12Z,14E,16S,17S,18R,19R,20R,21S,22R,23S,24E)-5,6,9,17,19-pentahydroxy-23-methoxy-2,4,12,16,18,20,22-heptamethyl-8-{(E)-[(4-methylpiperazin-1-yl)imino]methyl}-1,11-dioxo-1,2-dihydro-2,7-(epoxypentadeca[1,11,13]trienoimino)naphtho[2,1-b]furan-21-yl acetate; 8-(((4-Methyl-1-piperazinyl)imino)methyl)rifamycin SV; Rifamycin, 3-[[(4-methyl-1-piperazinyl)imino]methyl]-; Rifomycin SV, 8-(N-(4-methyl-1-piperazinyl)formidoyl)-; DSSTox_CID_1244; 3-(4-Methylpiperazinyliminomethyl)rifamycin SV; DSSTox_RID_76035; DSSTox_GSID_21244; [(7S,9E,11S,12R,13S,14R,15R,16R,17S,18S,19E,21Z)-2,15,17,27,29-pentahydroxy-11-methoxy-3,7,12,14,16,18,22-heptamethyl-26-[(E)-(4-methylpiperazin-1-yl)iminomethyl]-6,23-dioxo-8,30-dioxa-24-azatetracyclo[23.3.1.14,7.05,28]triaconta-1(29),2,4,9,19,21,25,27-octaen-13-yl] acetate; RIF; (7S,9E,11S,12R,13S,14R,15R,16R,17S,18S,19E,21Z)-2,15,17,27,29-pentahydroxy-11-methoxy-3,7,12,14,16,18,22-heptamethyl-26-{(E)-[(4-methylpiperazin-1-yl)imino]methyl}-6,23-dioxo-8,30-dioxa-24-azatetracyclo[23.3.1.1(4,7).0(5,28)]triaconta-1(28),1(29),2,4,9,19,21,25,27-nonaen-13-yl acetate; CAS-13292-46-1; 3-([(4-Methyl-1-piperazinyl)imino]methyl)rifamycin SV; 8-[[(4-Methyl-1-piperazinyl)imino]methyl]rifamycin sv; rifamcin; [1,11,13]trienimino)naphto[2,1-b]furan-21-yl acetate; Rifomycin sv, 8-[N-(4-Methyl-1-piperazinyl)formidoyl]-; Rifamsolin; Rifampicin [INN:BAN:JAN]; Rifapiam; Famcin; MFCD00151389; Rimactane (TN); NCGC00094777-01; AZT + Rifampin; Rifampin (USP); (2S,12Z,14E,16S,17S,18R,19R,20R,21S,22R,23S,24E)-5,6,9,17,19-Pentahydroxy-23-methoxy-2,4,12,16,18,20,22-heptamethyl-8-[[(4-methylpiperazin-1-yl)imino]methyl]-1,11-dioxo-1,2-dihydro-2,7-(epoxypentadeca; (2S,12Z,14E,16S,17S,18R,19R,20R,21S,22R,23S,24E)-5,6,9,17,19-pentahydroxy-23-methoxy-2,4,12,16,18,20,22-heptamethyl-8-{(E)-[(4-methylpiperazin-1-yl)imino]methyl}-1,11-dioxo-1,2-dihydro-2,7-(epoxypentadeca[1,11,13]trienoimino)naphtho[2,1-b]furan-21-yl acet; (2S,14E,16S,17S,18R,19R,20R,21S,22R,23S,24E)-5,6,9,17,19-Pentahydroxy-23-methoxy-2,4,12,16,18,20,22-heptamethyl-8-{[(4-methylpiperazin-1-yl)imino]methyl}-1,11-dioxo-1,2-dihydro-2,7-(epoxypentadeca[1,11,13]trienoimino)naphtho[2,1-b]furan-21-yl acetate; [pentahydroxy-methoxy-heptamethyl-[(E)-(4-methylpiperazin-1-yl)iminomethyl]-dioxo-[?]yl] acetate; Rifampicin & EEP; Prestwick_833; EINECS 236-312-0; R-Cin; Rifadin (TN); NSC 113926; Piperine & Rifampicin; Rifampicin & Propolis; Reserpine & Rifampicin; Rifampin [USAN:USP]; Prestwick2_000525; Prestwick3_000525; Spectrum5_002018; 3-[[(4-Methyl-1-piperazinyl)imino]-methyl]rifamycin; Rifampicin (JP17/INN); SCHEMBL23490; BSPBio_000509; L-5103-LEPETIT; 8CI); BPBio1_000561; CHEMBL374478; DTXSID6021244; HMS1569J11; HMS2089F12; HMS2096J11; HMS3713J11; DRG-0109; Rifampicin, >=97.0% (HPLC); Rifampicin, powder, gamma-irradiated; Tox21_111329; Tox21_201385; Tox21_300550; Ba 41166; BDBM50370232; GR-306; NIH-10782; AKOS015951372; Rifampicin - CAS 13292-46-1; Rifampicin, >=97% (HPLC), powder; Tox21_111329_1; BA 411661E; CCG-208267; DB01045; NCGC00022678-03; NCGC00022678-04; NCGC00022678-05; NCGC00022678-06; NCGC00179536-02; NCGC00254537-01; NCGC00258936-01; 5,6,9,17,19,21-Hexahydroxy-23-methoxy-2,4,12,16,18,20,22-heptamethyl-8-(N-(4-methyl-1-piperazinyl)formimidoyl)-2,7-(epoxypentadeca(1,11,13)trienimino)naphtho(2,1-b)furan-1,11(2H)-dione 21-acetate; M880; C06688; D00211; J10110; Rifampicin, VETRANAL(TM), analytical standard; AB00383022_07; 292R461; SR-05000002118; 8-[[(4-Methyl-1-piperazinyl)imino[methyl]rifamycin; 8-[[(4-Methylpiperazinyl)imino]methyl]rifamycin sv; SR-05000002118-3; WLN: V1 WQ A&1 E&1 E1UN- AT6N DNTJ D1; 3-[(4-Methyl-1-piperazinyl)iminomethyl]rifamycin SV; Rifampicin, European Pharmacopoeia (EP) Reference Standard; Rifampin, United States Pharmacopeia (USP) Reference Standard; Rifampin, Pharmaceutical Secondary Standard; Certified Reference Material; Rifampicin, plant cell culture tested, BioReagent, >=97% (HPLC), crystalline; yl]-, (2S,12Z,14E,16S,17S,18R,19R,20R,21S,22R,23S,24E)-; (2S,12Z,14E,16S,17S,18R,19R,20R,21S,22R,23S,24E)-5,6,9,17,19-pentahydroxy-23-methoxy-2,4,12,16,18,20,22-heptamethyl-8-{(E)-[(4-methylpiperazin-1-yl)imino]methyl}-1,11-dioxo-1,2-dihydro-2,7-(epoxypentadeca[1,11,13]trienoimino)naphtho[2,1-b]furan-21-yl ac; (7S,9E,11S,12R,13S,14R,15R,16R,17S,18S,19E,21Z)-2,15,17,27,29-pentahydroxy-11- methoxy-3,7,12,14,16,18,22-heptamethyl-26-{(E)-[(4-methylpiperazin-1-yl)imino] methyl}-6,23-dioxo-8,30-dioxa-24-azatetracyclo[23.3.1.14,7.05,28]triaconta-1 (28),2,4,9,19,21,25(29),26-octaen-13-yl acetate; 2,11,13]trienimino)naphtho[2,1-b]furan-1,11(2H)-dione, 5,6,9,17,19,21-hexahydroxy-23-methoxy-2,4,12,16,18,20,22-heptamethyl-8-[N-(4-methyl-1-piperazinyl)formimidoyl]-, 21-acetate; 2,7-(epoxy[1,11,13]pentadecatrienoimino)naphtho[2,1-b]furan-1,11(2H)-dione, 21-(acetyloxy)-5,6,9,17,19-pentahydroxy-23-methoxy-2,4,12,16,18,20,22-heptamethyl-8-[(E)-[(4-methyl-1-piperazinyl)imino]meth; 2,7-(Epoxypentadeca(1,11,13)trienimino)naphtho(2,1-b)furan-1,11(2H)-dione, 5,6,9,17,19,21-hexahydroxy-23-methoxy-2,4,12,16,18,20,22-heptamethyl-8-(N-(4-methyl-1-piperazinyl)formimidoyl)-, 21-acetate (; 2,7-(Epoxypentadeca(1,11,13)trienimino)naphtho(2,1-b)furan-1,11(2H)-dione, 5,6,9,17,19,21-hexahydroxy-23-methoxy-2,4,12,16,18,20,22-heptamethyl-8-(N-(4-methyl-1-piperazinyl)formimidoyl)-, 21-acetate (8CI); 5,6,9,17,19,21-Hexahydroxy-23-methoxy-2,4,12,16,18,20,22-heptamethyl-8-(N-(4-methyl-1-piperazinyl)formimidoyl)-2,7-(epoxypentadeca(1,11,13)trienimino)naphtho(2,1-beta)furan-1,11(2H)-dione 21-acetate; 5,6,9,17,19,21-hexahydroxy-23-methoxy-2,4,12,16,18,20,22-heptamethyl-8-[N-(4-methyl-1-piperazinyl)formimidoyl]-, 21-acetate; Rifampicin in combination with actinonin, BB-3497, hydroxylamine hydrochloride, and 1,10-phenanthroline; Stereoisomer of 5,9,17,19,21-Hexahydroxy-23-methoxy-2,4,12,16,18,20,22-he@ptamethyl-8-[N-(4-methyl-1-piperazinyl)formimidoyl]-2,7-(epoxypentadeca[1,11,13]trienimino)naphtho[2,1-b]furan-1,11(2H)-dione 21-acetate
Click to Show/Hide
|