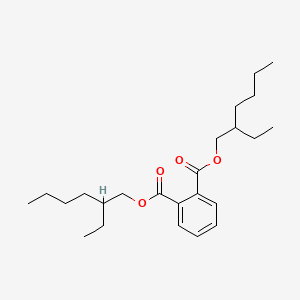
| Synonyms |
Bis(2-ethylhexyl) phthalate; DEHP; 117-81-7; BIS(2-ETHYLHEXYL)PHTHALATE; Di(2-ethylhexyl)phthalate; Di(2-ethylhexyl) phthalate; Diethylhexyl phthalate; 2-Ethylhexyl phthalate; Di-sec-octyl phthalate; Octyl phthalate; Fleximel; Octoil; Ethylhexyl phthalate; Palatinol AH; Celluflex DOP; Vestinol AH; Bisoflex DOP; Kodaflex DOP; Staflex DOP; Truflex DOP; Flexol DOP; Vinicizer 80; Bisoflex 81; Eviplast 80; Eviplast 81; Hercoflex 260; RC plasticizer DOP; Compound 889; Witcizer 312; Platinol dop; Di-2-ethylhexyl phthalate; Nuoplaz dop; Platinol ah; Hatcol dop; Reomol dop; Pittsburgh PX-138; Sansocizer DOP; Ergoplast FDO; Monocizer DOP; Plasthall DOP; Flexol plasticizer DOP; Mollan O; Jayflex DOP; Sicol 150; Ergoplast FDO-S; Di(2-ethylhexyl)orthophthalate; Good-rite gp 264; Reomol D 79P; Bis(2-ethylhexyl) benzene-1,2-dicarboxylate; Di(ethylhexyl) phthalate; Bis(ethylhexyl) phthalate; Rcra waste number U028; Px-138; Phthalic acid dioctyl ester; NCI-C52733; Di(2-ethylhexyl) o-phthalate; Phthalic acid di(2-ethylhexyl) ester; 1,2-Benzenedicarboxylic acid, bis(2-ethylhexyl) ester; DOP; Bis(2-ethylhexyl) 1,2-benzenedicarboxylate; UNII-C42K0PH13C; Bis(2-ethylhexyl) o-phthalate; Phthalic acid, bis(2-ethylhexyl) ester; CHEBI:17747; 1,2-Benzenedicarboxylic acid bis(2-ethylhexyl) ester; Benzenedicarboxylic acid, bis(2-ethylhexyl) ester; Phthalic Acid Bis(2-ethylhexyl) Ester; Bis-(2-ethylhexyl)ester kyseliny ftalove; C42K0PH13C; 1,2-Benzenedicarboxylic acid, 1,2-bis(2-ethylhexyl) ester; DTXSID5020607; Etalon; Bis-(2-ethylhexyl)ester kyseliny ftalove [Czech]; Di-(2-ethylhexyl) phthalate; BIS-(2-ETHYLHEXYL) PHTHALATE; NCGC00091499-05; Sconamoll DOP; Diacizer DOP; Kodaflex DEHP; 15495-94-0; Etalon (plasticizer); Sansocizer R 8000; Caswell No. 392K; Dioctylphthalate; Behp; Di-2-ethylhexylphthalate; Diplast O; ESBO-D 82; Ergoplast FDO; Ergoplast FDO-S; Etalon; Phthalic acid, bis-2-ethylhexyl ester; DOF [Russian plasticizer]; SMR000777878; CCRIS 237; Ethyl hexyl phthalate; HSDB 339; Di(2-ethylhexyl) orthophthalate; Bis-(2-ethylhexyl)ester kyseliny ftalove (czech); EINECS 204-211-0; NSC 17069; Diethylhexylphthalate (Bis-(2-ethylhexyl) Phthalate); RCRA waste no. U028; Union carbide flexol 380; EPA Pesticide Chemical Code 295200; BRN 1890696; AI3-04273; DAF 68; Palatinol DOP; Polycizer DOP; Merrol DOP; Palatinol AH-L; Hatco DOP; Vinycizer 80; Di(2-ethylhexyl)phthalate (DEHP); N-Dioctyl phthalate; MFCD00009493; Corflex 400; Dioctyl phthalate, 99%; DSSTox_CID_607; 1, bis(ethylhexyl) ester; Epitope ID:140107; EC 204-211-0; WLN: 8OVR BVO8; Di(2-Ethylhexyl phthalate); DSSTox_RID_75688; DSSTox_GSID_20607; SCHEMBL20271; 14C -DEHP; 8033-53-2; MLS001333173; MLS001333174; MLS002454397; Dioctyl phthalate, >=99.5%; 1,2-Benzenedicarboxylic acid, bis-(1-ethylhexyl) ester; CHEMBL1242017; SCHEMBL21733281; HMS2233C15; HMS3374J09; AMY40790; HY-B1945; NSC17069; Tox21_400084; Bis(2-ethylhexyl)ester phthalic acid; NSC-17069; s3360; AKOS024318875; Bis(2-ethylhexyl) phthalate-[13C6]; Phthalic acid bis(2-ethylhexyl ester); MCULE-4692716107; NCGC00091499-01; NCGC00091499-02; NCGC00091499-04; NCGC00091499-06; NCGC00091499-07; CAS-117-81-7; I887; Bis(2-ethylhexyl) 1, 2-benzenedicarboxylate; CS-0014050; FT-0624576; FT-0663286; P0297; WLN: 4Y2 & 1OVR BVO1Y4 & 2; Bis(2-ethylhexyl) phthalate, Selectophore(TM); C03690; A937603; Q418492; 1,2-Benzenedicarboxylic acid bis-(1-ethylhexyl) ester; benzene-1,2-dicarboxylic acid bis(2-ethylhexyl) ester; BRD-A89471977-001-05-2; Bis(2-ethylhexyl) phthalate 100 microg/mL in Methanol; Bis(2-ethylhexyl) phthalate 5000 microg/mL in Methanol; F0001-0292; Bis(2-ethylhexyl) phthalate, SAJ first grade, >=98.0%; Bis(2-ethylhexyl) phthalate, PESTANAL(R), analytical standard; Phthalic acid, bis-2-ethylhexyl ester 10 microg/mL in Cyclohexane; Plastic additive 01, European Pharmacopoeia (EP) Reference Standard; Bis(2-ethylhexyl) phthalate, certified reference material, TraceCERT(R); Plastic additive 14, United States Pharmacopeia (USP) Reference Standard; 1,2-Benzenedicarboxylic acid, bis(2-ethylhexyl) ester, labeled with carbon-14; 50885-87-5; 82208-43-3
Click to Show/Hide
|