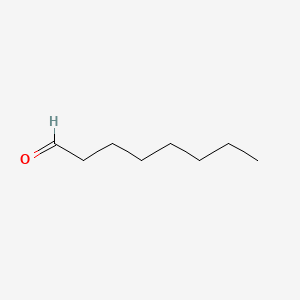
| The Content Variation of Natural Product Induced by Different Factor(s) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Species Name: Arabidopsis thaliana | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor Name: T-DNA Knock-Out Treatment | [1] | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Experiment Detail |
In vitro cultivation of Arabidopsis wildtype and mutant plants: Seeds were sterilized according to standard lab routines (EtOH, NaOCl/NaOH) prior to aseptical (in vitro) cultivation in 500 ml screw cap jars on MS medium (4.3 g/l; 50 ml/jar) containing Bacto- and Phytoagar (1:2; 6 g/l) and 30 g/l sucrose. Ten seeds were pipetted into each jar and plants grown for 6 weeks until flowering at a temperature of 20 ℃ under a 16/8 h day/ night regime using fluorescent tubes (Osram Lumilux Plus Eco 36 W). Both Arabidopsis thaliana wildtype plants of ecotype Columbia-0 (Col) and 4 Col-derived T-DNA knock-out mutants (homozygous lines) showing deficiencies in the GLS biosynthesis pathway were used in this study (five parallels for wildtype and mutants): TGG1 (Atg526000; Salk_130469), TGG2 (At5g25980; Salk_038730), Cyp83A1 (At4g13770) and Cyp83B1 (At4g31500; Salk_028573). Greenhouse-cultivation of Arabidopsis ecotypes: The following Arabidopsis ecotypes were used in the study: Columbia (Col), Cape Verde Islands (Cvi), Landsberg erecta (Ler) and Wassilewskija (Ws). Single plants were greenhouse-cultivated on fertilized soil (P-Jord; Emmaljunga Torvmull AB) in plug trays (9 × 6 cells) at a temperature of 20 ℃ (three parallels for each ecotype). Due to the 6-weeks growth period (November/December 2003), the plants were cultivated under a 16/8 h day/night regime using metal halide lamps (Osram HQI-T 400 W) placed 130 cm above the trays. Depending on the ecotypical plant development, whole plants were sampled after 3-4 weeks right before bolting for in vivo studies, while investigations of single plant organs (leaf, stem, inflorescence) were carried out after 5-6 weeks of cultivation.
Click to Show/Hide
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor Function |
Metabolites from methionine, leucine and phenylalanine-derived glucosinolates were most abundant (4-methylthiobutyl, 4-methylpentyl, 2-phenylethyl). In addition, 24 monoterpenes, 26 sesquiterpenes and 12 aromatic structures, predominantly observed in inflorescenses, are described. Excluding the vast group of straight chain aliphatic structures, a total of 102 volatile compounds were detected, of which 59 are reported in Arabidopsis thaliana for the first time, thus emphasizing the sensitivity and applicability of solid-phase microextraction for volatile profiling of plant secondary metabolites.
Click to Show/Hide
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor | Part | Location | NP Content | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Inflorescences: A. thaliana wildtype detected in vivo
|
Flowers | Norway |
NP Content: 1.38 %
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Leaves: A. thaliana wildtype detected in vivo
|
Leaves | Norway |
NP Content: 1.64 %
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Stem: A. thaliana wildtype detected in vivo
|
Stems | Norway |
NP Content: 1.23 %
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Arabidopsis thaliana wildtype
|
Whole plant | Norway |
NP Content: 0.14 %
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
A. thaliana T-DNA knock-out mutant Cyp83A1: (At4g13770)
|
Whole plant | Norway |
NP Content: 0.85 %
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
A. thaliana T-DNA knock-out mutant Cyp83B: (At4g31500; Salk_028573)
|
Whole plant | Norway |
NP Content: 0.33 %
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
A. thaliana T-DNA knock-out mutant TGG1: (Atg526000; Salk_130469)
|
Whole plant | Norway |
NP Content: 0.27 %
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
A. thaliana T-DNA knock-out mutant TGG2: (At5g25980; Salk_038730)
|
Whole plant | Norway |
NP Content: 0.26 %
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Species Name: Artemisia absinthium | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor Name: Chemotype Comparison | [2] | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Experiment Detail |
Ten different plants of wormwood were collected in March 1997 from each one of the following four wild populations in the Spanish Pyrenees: Tallo de Aulet (prov. Huesca) and Pont de Suert, Sort and Farga de Moles (prov. Lleida). In three of the four populations studied, there was another chemotype, with 25-65% of cis-epoxyocimene and 15-50% of chrysanthenyl acetate. This chemotype, called chemotype B, was less frequent in the Pyrenees than the chemotype A, appearing only in 17% of the samples (two samples in TallO de Aulet and in Pont de Suert and three samples in Farga de Moles).
Click to Show/Hide
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor Function |
Two chemotypes were detected; a cis-epoxyocimene type (with more than 50% of this compound) which was predominant in all the populations, and a cis-epoxyocimene + chrysanthenyl acetate type (with 25-65% of cis-epoxyocimene and 15-50% of chrysanthenyl acetate). The distribution of these chemotypes had no relation with the altitude of the samples.
Click to Show/Hide
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor | Part | Location | NP Content | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Chemotype (cis-epoxyocimene type)
|
Leaves | Spain |
NP Content: <0.03 %
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Chemotype (cis-epoxyocimene + chrysanthenyl acetate type)
|
Leaves | Spain |
NP Content: <0.03 %
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Species Name: Citrus sinensis (Hongjiang) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor Name: Variety Comparison | [3] | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Experiment Detail |
Four kinds of fresh sweet oranges were obtained in the same season, November 2000, in Guangzhou. Citrus sinensis var. Hongjiang (called 'hong jiang chen' in Chinese) and C. sinensis Osbeck var. Anliu (called 'luo gang chen') were obtained at an orchard in Luo gang in Guangzhou (25 km from the center of Guangzhou). Citrus sinensis var. Sihui (called 'sihui ju') was harvested at the Shigou Experimental Farm in Sihui City in Guangdong Province (75 km far away from Guangzhou). Citrus sinensis var. Washington navel (called 'qi chen') which was produced in Jiangxi Province (200 km from Guangzhou; bordering Guangdong Province), was purchased at the wholesale market in Guangzhou. All oranges were kept in a cold room until prepared a few days later.
Click to Show/Hide
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor Function |
The peel oil compositions of four kinds of sweet oranges in China, Citrus sinensis Osbeck var. Hongjian, C. sinensis Osbeck var. Anliu, C. sinensis Osbeck var. Sihui and C. sinensis Osbeck var. Washington navel, were investigated by GC and GC/MS. The essential oils were extracted by cold-pressing method. Forty-two to 53 compounds were quantitatively determined for each variety. Their percentages, respectively, were: > 97.3%, > 98.4%, > 97.5% and > 98.0% in hydrocarbons; > 1.5%, > 0.7%, > 0.8% and > 0.9% in total aldehydes; 0.8%, 0.5%, 0.5% and 0.5% in alcohols. Either cis-or trans-limonene oxide was detected in small amounts in each of the four samples, with Hongjiang containing both limonene oxides. delta-3-Carene was commonly quantified at a level of 0.1% in all the samples. The content of aliphatic aldehydes, including octanal, nonanal, decanal and dodecanal, exceeded that of terpene aldehydes, such as neral and geranial in Hongjiang (0.9%) and Washington navel (0.6%), whereas the aliphatic aldehydes in Anliu and Sihui were present to a lesser degree than the terpene aldehydes. Either alpha- or beta-sinensal was detected in trace amounts in each of the four samples. Linalool was the major alcohol in all the samples. Nootkatone was not detected.
Click to Show/Hide
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor | Part | Location | NP Content | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Citrus sinensis var. Anliu
|
Fruits | China |
NP Content: < 0.005 %
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Citrus sinensis var. Hongjiang
|
Fruits | China |
NP Content: 0.4 %
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Citrus sinensis var. Sihui
|
Fruits | China |
NP Content: 0.2 %
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Citrus sinensis var. Washington navel
|
Fruits | China |
NP Content: 0.4 %
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Species Name: Coriandrum sativum | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor Name: NaCl Treatment | [4] | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Experiment Detail |
Plant material: Coriander (Coriandrum sativum L.) fruits were collected from cultivated plants in the region of Korba (northeastern Tunisia) in April 2006. Seeds were set to germinate at 25 ℃. Ten-day-old coriander seedlings were grown in quarter-strength Hoagland's solution laced with 0 mM, 25 mM, 50 mM and 75 mM of NaCl. The culture was placed in a greenhouse with 25 ℃ day maximum and 18 ℃ night minimum, under artificial light of 141 µmol/m2 /s (6000 lux) with 16 h photoperiod and 60-80% air humidity. Nutrient solution was continuously aerated. Growth parameters: Plants were harvested at the seedling stage 3 weeks after treatment.
Click to Show/Hide
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor Function |
Essential oil content was 1762.64 µg/g dry weight (DW) (0.18%) and 1255.77 µg/g DW (0.12%) in stems and leaves, respectively. At low and moderate stress, a significant difference in the essential oil content was developed between stems, with a significant decrease, and leaves, with an increase up to 43%. Under high salinity, the oil content of both organs decreased significantly. The major volatile compound of stems and leaves was (E)-2-decenal with 24% and 52%, respectively. Other important components were decanal, (E)-2-dodecenal, dodecanal, (E)-2-undecenal, (E)-2-tridecenal and (E)-2-undecanal. Further, the content of these compounds were affected differently by the treatment level and by the organ type.
Click to Show/Hide
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor | Part | Location | NP Content | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
0 mM NaCl (Control)
|
Leaves | Tunisia |
NP Content: 1.93 %
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
0 mM NaCl (Control)
|
Stems | Tunisia |
NP Content: 0.16 %
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
25mM NaCl
|
Leaves | Tunisia |
NP Content: 5.96 %
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
25mM NaCl
|
Stems | Tunisia |
NP Content: 0.51 %
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
50 mM NaCl
|
Leaves | Tunisia |
NP Content: 3.29 %
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
50 mM NaCl
|
Stems | Tunisia |
NP Content: 0.1 %
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
75 mM NaCl
|
Leaves | Tunisia |
NP Content: 4.1 %
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
75 mM NaCl
|
Stems | Tunisia |
NP Content: 0.67 %
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Species Name: Ducrosia anethifolia | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor Name: Locality Variation | [5] | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Experiment Detail |
The aerial parts of Ducrosia anethifolia (DC.) Boiss. were collected in the wild from Mehdi Abad (Kerman province, in southern Iran) at the flowering stage in June 2006. The material was dried at room temperature.
Click to Show/Hide
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor Function |
The 63 components of this interesting plant were identified in the oil of D. anethifolia, representing 94.0% of the oil. alpha-Pinene (11.6%), terpinolene(3.2%) and (z)-beta-ocimene (2.8%) were the main hydrocarbon components present in the oil, while decanal (54.0%), cis-chrysanthenyl acetate(3.2%) and decanoic acid (1.3%) were the major oxygen-containing constituents.
Click to Show/Hide
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor | Part | Location | NP Content | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Locality: Karaj, Iran
|
Aerial parts | Iran |
NP Content: 0.2 %
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Locality: Kerman, Iran
|
Aerial parts | Iran |
NP Content: 0.4 %
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Species Name: Ducrosia assadii | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor Name: Locality Variation | [6] | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Experiment Detail |
The aerial parts of D. assadii Alava. were collected in the wild from Lalehzar (Kerman Province, in southern Iran) at the flowering stage, in July 2007. The material was dried at room temperature and used for distillation. Distillation: A direct-fired field distillation unit containing a distillation tank (capacity: 1,000 L), a condensation column and receiver, all made of stainless steel, and which can process 30-50 kg of dried aerial parts from the plants/batch, was installed at an altitude of 2600 m (boiling point: 87 ℃). Dried aerial parts from the plants (40 kg) were charged into the distillation unit along with 500 L fresh water and the unit was heated by steam. The system was kept open to atmospheric pressure until the temperature reached to 70 ℃, when the air present in the unit was replaced by the vapor. After complete removal of air from the unit, the air vent was closed and the whole unit was operated as a closed system under pressure to distill the oil. The pressure, temperature and rate of distillation were controlled manually. The process was completed after the collection of 500 L of water distillate. The oil collected in the receiver and dried over anhydrous Na2SO4. Extraction of Ducrosia Second Oil From Ducrosia Water by Redistillation: The seprated distillate water collected in the receiver was redistilled in a 1,000 L still to yield more Doucrosia oil (this oil is known as secondary essential oil, second oil, cooked oil or indirect oil).
Click to Show/Hide
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor Function |
Fifty components were identified in a second oil of D. assadii from Lalehzar with decanal (35.2%), nonadecane (12%) and citronellyl acetate (11.6%) as the main constituents. The oil from Dehbakrii also contained decanal (36.4%) as the main component of an oil recovered from the distillate water. The results showed that the amount of decanal is remarkably high in the oils of D. assadii.
Click to Show/Hide
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor | Part | Location | NP Content | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Locality: Lalehzar, Kerman Province, southern Iran
|
Aerial parts | Iran |
NP Content: 0.1 %
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Species Name: Micromeria biflora | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor Name: Seasonal Variation | [7] | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Experiment Detail |
The aerial parts of M. biflora collected during November 1993 and June 1994 were used for the investigation.
Click to Show/Hide
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor Function |
The major constituents of the oil were neral (25.3-32.2%) and geranial (26.7-41.3%). The oil produced in the winter was found to contain higher amounts of oxygenated monoterpenes than the oil produced in the summer.
Click to Show/Hide
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor | Part | Location | NP Content | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Harvesting time: Summer
|
Aerial parts | South India |
NP Content: 0.09 %
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Harvesting time: Winter
|
Aerial parts | South India |
NP Content: 0.1 %
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Species Name: Pulicaria dysenterica | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor Name: Locality Variation | [8] | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Experiment Detail |
Aerial parts of P. dysenterica were collected during the flowering stage from two different locations in Greece in August 2002. Sample A: Katara (Perfecture Trikala). Sample B: Arahova (Perfecture Viotia).
Click to Show/Hide
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor Function |
Fifty-four components were identified representing 80.5% (sample A) and 72.6% (sample B) of the total oils. The main components in sample A were (Z)-nerolidol (11.2%), caryophyllene oxide (9.1%) and (E)-nerolidol (6.6%), while those of sample B were beta-caryophyllene (12.8%), caryophyllene oxide (12.8%) and (E)-nerolidol (6.9%).
Click to Show/Hide
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor | Part | Location | NP Content | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Locality: Arahova, Perfecture Viotia, Greece
|
Aerial parts | Greece |
NP Content: <0.1 %
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Species Name: Tanacetum cadmeum ssp. orientale | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor Name: Locality Variation | [9] | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Experiment Detail |
Plant materials were collected during the flowering period in July 2002 from the Dumluca Mountain in the vicinity of Divrigi village of Sivas city at 1900 m altitude and Saksagan Gorge in Saimbeyli village of Adana city at 1900 m altitude.
Click to Show/Hide
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor Function |
The flower, stem and root oils of T. cadmeum ssp. orientale collected from the Adana location were characterized with alpha-thujone (25%, 5.2%), cis-linalool oxide (6.8%, 12.8%), trans-chrysanthenyl acetate (5.8%, 8.5%) for flower and stem oils, and beta-eudesmol (10.3%, 6.2%, 13.8%); in addition, stem oil contained 1,8-cineole (6.6%) and root oil contained hexadecanoic acid (6.0%), spathulenol (5.8%) and beta-muurolol (5.3%). The flower and stem oils of T. cadmeum ssp. orientale collected from the Sivas location were characterized with camphor (25.9%, 14.8%), borneol (15.4%, 25.8%) and alpha-thujone (7.8%, 5.5%); in addition, stem oil contained 1,8-cineole (7.4%) and root oil contained nonacosane (16.2%), spathulenol (6.8%) and hexadecanoic acid (5.8%).
Click to Show/Hide
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor | Part | Location | NP Content | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Flower: (Locality: Adana, Turkey)
|
Flowers | Adana, Turkey |
NP Content: <0.1 %
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Flower: (Locality: Sivas, Turkey)
|
Flowers | Sivas, Turkey |
NP Content: 0.1 %
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Species Name: Teucrium flavum | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor Name: Month Variation; Developmental Stage Variation | [10] | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Experiment Detail |
The aerial parts of T. flavum were collected in different periods from December to July 2006, from plants growing along the Ionic coast of Sicily (Italy). LF 1-LF 2-LF 3: represent the composition of leaf oils of plant samples collected in December (vegetative stage), February (pre-flowering stage) and April (budding stage) respectively; FL: flower oil; FR: fruit oil.
Click to Show/Hide
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor Function |
Some components, in all investigated plant parts, remained more or less constant during all the different phases of the plant cycle life. Worthy of note, considering the leaf oils, was that beta-pinene, limonene and germacrene D increased in the pre-flowering stage, while a series of esters and alpha-copaene, beta-caryophyllene, viridiflorol, Tmuurolol and phytol increased in the budding stage (LF3); the vegetative stage oil is generally characterized by a rich chemical composition and some constituents such as isoamyl hexanoate, alpha-humulene, bicyclogermacrene, beta-bisabolene and alpha-bisabolol reached their highest levels in this oil. In the flower oil, linalool and 1-octen-3-yl acetate were the main components compared to the amounts found in the other oils. Fruit oil composition was relatively oil poor, with beta-bisabolene, caryophyllene oxide, cadin-4-en-1-ol and phytone as the major constituents.
Click to Show/Hide
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor | Part | Location | NP Content | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Harvesting time: April; budding stage
|
Leaves | Italy |
NP Content: <0.01 %
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Species Name: Vitis vinifera cv. Pinot noir | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor Name: Drought Stress Treatment | [11] | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Experiment Detail |
3-year old single shoot V. vinifera plants (cultivar Pinot noir 18 Gm grafted on Kober 5BB, 51 plants) potted in 3L pots in a sandy loam soil were used. All plants were well watered (200 mL per day) at the beginning of the experiment (04.06.2010; DAY 0; 5 plants) and water was supplied to all control plants once every day (250 mL per day), whereas water supply of stressed plants was stopped. Physiological measurements and sampling of leaves took place on 07.06.2010 (DAY 3; 5 control, 5 stressed plants), 10.06.2010 (DAY 6; 5 control, 5 stressed plants) and 12.06.2010 (DAY 8; 5 control, 10 stressed plants). Due to very hot weather conditions in June 2010 the experiment was stopped after 8 days and 12 available control plants were used to restart the drought treatment with 6 control and 6 stressed plants on 11.06.2010 and all plants were measured on 15.06.2010 (DAY 5). The mean leaf temperatures at midday were: 25 ℃ (04.06.2010; DAY 0), 31.9 ℃ (07.06.2010; DAY 3), 30.8 ℃ (15.06.2010; DAY 5), 35.8 ℃ (10.06.2010; DAY 6) and 35.7 ℃ (12.06.2010; DAY 8). The mean PAR radiation per day (measured from 6:00 am till 7:00 pm) was 144.1 µmol m-2 s-1. Each plant was used only once for physiological measurements and sampling of leaves.On every day of the experiment (day 0, 3, 5, 6, 8) the pot weight and the volumetric soil moisture content (ThetaProbe ML2x and handheld data logger Moisture Meter HH2, Delta-T Devices, Cambridge, United Kingdom) was recorded. The water potential (PWSC Model 3000, Soilmoisture Equipment Corporation, Santa Barbara, USA) was determined for the 6th leaf (representing the insertion level of the shoot from the basis) of every plant and measurement day. Chlorophyll fluorescence and gas exchange parameters of light adapted leaves were determined with the 4th and 5th leaf, whereas dark adaptation was performed only with the 5th leaf. Immediately after these non-invasive measurements, the 5th leaf was harvested, frozen in liquid nitrogen and further used for the measurement of polyphenols, selected primary metabolites and volatiles (VOCs).
Click to Show/Hide
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor Function |
The content of different groups of primary and secondary metabolites is significantly influenced by severe drought stress in grapevine leaves. The content of the majority of the metabolites (around 60% of primary metabolites, around 85% of polyphenols and about 40% of the detected and identified VOCs) increased upon drought stress treatment. Among these especially the primary metabolites citric acid and glyceric acid were strongly influenced by the short as well as the prolonged drought stress treatment, whereas all polyphenols were only induced upon the prolonged drought stress treatment.
Click to Show/Hide
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor | Part | Location | NP Content | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Normal condition
|
Leaves | Vienna, Austria |
NP Content: 12369.4 ± 8042.2 peak areas
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Dry 3-5 days
|
Leaves | Vienna, Austria |
NP Content: 12140.9 ± 6877.6 peak areas
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Dry 6-8 days
|
Leaves | Vienna, Austria |
NP Content: 13811.4 ± 10127.7 peak areas
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||