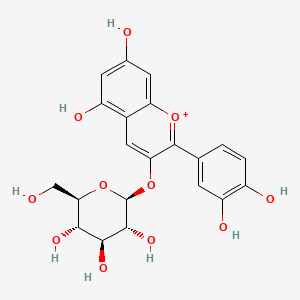
| The Content Variation of Natural Product Induced by Different Factor(s) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Species Name: Fragaria ananassa cv. Camarosa | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor Name: Organic Fertilization; Traditional Fertilization; AMF Inoculation | [1] | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Experiment Detail |
Clonal commercial strawberry plantlets (Fragaria ananassa cv. Camarosa) acquired from Vivero Llahuen, a local producer from the Biobio Region in Chile, were cropped in soil and further propagated from secondary stolons in a sterile substrate mix containing sand/vermiculite/perlite (1/1/1; v/v/v). Cultivation of the plantlets was carried out in a greenhouse under controlled light (16/8 h) and temperature (25 ℃ /18 ℃ day/night) conditions at the Departamento de Ciencias Quimicas y Recursos Naturales, University of La Frontera, Temuco, Chile. Fertilization conditions were based on the recommendations given by the Instituto de Desarrollo Agropecuario (INDAP). Briefly, the fertilization scheme involved application of soluble forms of urea, calcium nitrate, monoammonium phosphate, and urea phosphate (UP) to achieve the equivalent of 140 kg N/ha, 30 kg P/ha and 130 kg K/ha (traditional fertilization, TF). When the plants that originated from the stolons were prepared (when three-to-four leaves were completely extended), some of them were selected and used in the greenhouse experiment.The experimental unit was a mesocosm of 50 × 15 × 15 cm (l, w, h) including three F. ananassa plants randomly distributed on the substrate described above. In detail, each experimental unit or mesocosm included three plants, which were homogeneously sampled to obtain a composite sample (experimental sample) at harvest time when all of the treatment groups presented mature fruits. Ten treatments were established with organic or chemical fertilization and two levels of fertilization including 100% or 50% of the recommendation described above for each type of fertilization. For traditional fertilization, the scheme described above was applied in a solution. Commercial red manure was acquired from ANASAC (Santiago, Chile). The main components corresponded to 8% organic matter, 0.5% N, 10% P, and 0.8% K, and contains traces of Mg, Ca, S, Fe, Mn, Cu, Pb and I. The equivalent amount of red manure was applied as a solid in the organic treatments (OF). Because one of the main factors studied here was the source and concentration of P, the doses of red manure were evaluated considering the P content. In the case of N and K, their concentration in red manure allowed a similar supply of both nutrients; thus, no other fertilizer additions were required. All conditions were also cropped, and either inoculated or not with the AM fungus Claroideoglomus claroideum (N.C. Schenck & G.S. Sm.) C. Walker & A. Schubler (named as +M, -M, respectively). This fungus is a habitual component of AM fungal communities in Andisols from southern Chile,4 and the strain used here is maintained in trap crops at the germplasm bank of AM fungi in CIMYSA (Universidad de La Frontera, Temuco, Chile). For the inoculation process, the pots were first prepared with a sterile substrate composed of sepiolite, vermiculite and sand (1:1:1, v:v:v). Then, at the trasplanting time, 5 g of a similar substrate containing the fungal propagules, previously produced in trap pots cropped with maize and clover, were placed in the space of the roots ensuring complete contact with all the root surfaces. Subsequently, the free space was filled with sterile substrate. Transplanting was done on January 26, 2017, and harvesting was completed seven months later (September, 2017). In the inoculated treatments, 5 g of inoculum (approximately 70 spores per gram) was added to each plant at transplanting time. In the noninoculated plants, 10 mL of an inoculum filtrate (1/10, inoculum/water) was added to homogenize the nonmycorrhizal microbiota. The control was the treatment without any kind of fertilization or inoculation.
Click to Show/Hide
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor Function |
The type and dose of fertilizer and AM colonization have significant effects on important traits of strawberry fruits, such as the concentration of anthocyanins, which, interestingly, are higher in general in treatments that were not inoculated with AM, in contrast to their antioxidant capability, which was higher after AM-inoculated treatment. This suggests the participation of compounds other than anthocyanins as important sources of antioxidant activity, which should be studied further.
Click to Show/Hide
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor | Part | Location | NP Content | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Without fertilization + AMF inoculation
|
Fruits | Temuco, Chile |
NP Content: 17.8 ± 1.0 µg/g
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Without fertilization
|
Fruits | Temuco, Chile |
NP Content: 19.0 ± 2.7 µg/g
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
50% of Traditional fertilization + Claroideoglomus claroideum (AMF) inoculation
|
Fruits | Temuco, Chile |
NP Content: 18.5 ± 0.7 µg/g
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
50% of Traditional fertilization
|
Fruits | Temuco, Chile |
NP Content: 18.1 ± 1.9 µg/g
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
100% of Traditional fertilization + Claroideoglomus claroideum (AMF) inoculation
|
Fruits | Temuco, Chile |
NP Content: 17.4 ± 0.9 µg/g
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
100% of Traditional fertilization
|
Fruits | Temuco, Chile |
NP Content: 20.1 ± 0.6 µg/g
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
50% of Organic fertilization + Claroideoglomus claroideum (AMF) inoculation
|
Fruits | Temuco, Chile |
NP Content: 18.6 ± 0.7 µg/g
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
50% of Organic fertilization
|
Fruits | Temuco, Chile |
NP Content: 20.3 ± 2.8 µg/g
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
100% of Organic fertilization + Claroideoglomus claroideum (AMF) inoculation
|
Fruits | Temuco, Chile |
NP Content: 20.2 ± 0.8 µg/g
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
100% of Organic fertilization
|
Fruits | Temuco, Chile |
NP Content: 16.2 ± 0.7 µg/g
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Species Name: Fragaria × ananassa Duch. | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor Name: Nitrogen Treatment; AMF Inoculation | [2] | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Experiment Detail |
The experiment was conducted in a 'shade'-type greenhouse with 30% shade at the Instituto de Investigaciones Agropecuarias y Forestales (IIAF), Universidad Michoacana de San Nicolas de Hidalgo (UMSNH), Morelia, Michoacan, Mexico. Maximum and minimum temperatures in the greenhouse varied between 28 and 32 ℃ and between 8 and 18 ℃ respectively. Plants of the strawberry cultivar 'Aromas' were used that had previously been grown in a sterilised (95 ℃ water/steam, 40 min) substrate of coconut fibre/perlite (1:3 v/v) under greenhouse conditions. Before the experiment was established, the absence of AMF in the roots was verified by the ink and vinegar technique, modifying the duration of immersion in KOH and ink/vinegar solution (7 and 5 min respectively). Before planting, roots were disinfected by submerging them for 20 s in 20 g/L sodium hypochlorite solution and rinsing them in water. The inoculum was prepared with spores of Glomus intraradices cultivated in liquid medium (3.5 × 106 spores/L, 90% viability; Premier Tech Biotechnologies Company, Quebec, Canada), which was diluted with fitagel (Sigma P-8169, Saint Louis, MO, USA) solution at 50 g/L to obtain a final concentration of about 5 × 104 spores/L. The viability of spores was determined according to the method of An and Hendrix. Eighteen days after setting up the experiment, each plant received 2 mL of inoculum applied directly to the recently formed roots. One month later, after staining, the percentage of root colonisation was determined by the gridline intersect method. The experiment was organised as a full factorial, completely randomised design with two factors: inoculation (two levels: mycorrhizal and non-mycorrhizal plants) and N concentration in the nutrient solution (three levels: 3, 6 and 18 mmol/L). The six treatments were replicated four times, producing 24 experimental units with ten plants each. Every second day, all plants were irrigated up to substrate saturation. Nitrogen was supplied as NO and the cation/anion ratio was kept constant by varying the concentration of SO. When N was below 18 mmol/L, the cation concentrations were maintained as follows: K+, 3; Ca2+, 3.5; Mg2+, 1.5 mmol/L. They were increased in the 18 mmol/L N treatment: K+, 6.5; Ca2+, 7.5; Mg2+, 3.25 mmol/L. In all nutrient solutions the concentration of phosphorus (P) was 0.3 mmol/L. The other nutrients in the solutions were: H3BO3, 20; CuSO4. 5H2O, 0.5; Fe-EDTA (Ethylenediaminetetraacetic acid iron (III) sodium salt), 15; MnSO4.H2O, 12; (NH4)6Mo7O24 . 4H2O, 0.05; ZnSO4 . 7H2O, 3 µmol/L. The pH was adjusted to 5.5 at every application date.
Click to Show/Hide
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor Function |
Mycorrhization did not modify the weight, diameter or length of strawberry fruits but had a negative effect on most colour parameters. Moreover, fruits of mycorrhizal plants had higher K and Cu concentrations and showed greater accumulation of most phenolic compounds.
Click to Show/Hide
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor | Part | Location | NP Content | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Nitrogen concentration (mmol/L): 3
|
Mature fruits | Morelia, Michoacan, Mexico |
NP Content: 0.001 g/kg dry matter
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Nitrogen concentration (mmol/L): 6
|
Mature fruits | Morelia, Michoacan, Mexico |
NP Content: 0.002 g/kg dry matter
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Nitrogen concentration (mmol/L): 18
|
Mature fruits | Morelia, Michoacan, Mexico |
NP Content: 0.001 g/kg dry matter
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Glomus intraradices inoculation
|
Mature fruits | Morelia, Michoacan, Mexico |
NP Content: 0.001 g/kg dry matter
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Non-AMF inoculation
|
Mature fruits | Morelia, Michoacan, Mexico |
NP Content: 0.002 g/kg dry matter
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Nitrogen concentration (mmol/L): 3 + G. intraradices inoculation
|
Mature fruits | Morelia, Michoacan, Mexico |
NP Content: 0.001 g/kg dry matter
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Nitrogen concentration (mmol/L): 3 + Non-AMF inoculation
|
Mature fruits | Morelia, Michoacan, Mexico |
NP Content: 0.001 g/kg dry matter
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Nitrogen concentration (mmol/L): 6 + G. intraradices inoculation
|
Mature fruits | Morelia, Michoacan, Mexico |
NP Content: 0.001 g/kg dry matter
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Nitrogen concentration (mmol/L): 6 + Non-AMF inoculation
|
Mature fruits | Morelia, Michoacan, Mexico |
NP Content: 0.002 g/kg dry matter
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Nitrogen concentration (mmol/L): 18 + G. intraradices inoculation
|
Mature fruits | Morelia, Michoacan, Mexico |
NP Content: 0.001 g/kg dry matter
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Nitrogen concentration (mmol/L): 18 + Non-AMF inoculation
|
Mature fruits | Morelia, Michoacan, Mexico |
NP Content: 0.001 g/kg dry matter
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Species Name: Vitis vinifera cv. Pinot noir | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor Name: Drought Stress Treatment | [3] | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Experiment Detail |
3-year old single shoot V. vinifera plants (cultivar Pinot noir 18 Gm grafted on Kober 5BB, 51 plants) potted in 3L pots in a sandy loam soil were used. All plants were well watered (200 mL per day) at the beginning of the experiment (04.06.2010; DAY 0; 5 plants) and water was supplied to all control plants once every day (250 mL per day), whereas water supply of stressed plants was stopped. Physiological measurements and sampling of leaves took place on 07.06.2010 (DAY 3; 5 control, 5 stressed plants), 10.06.2010 (DAY 6; 5 control, 5 stressed plants) and 12.06.2010 (DAY 8; 5 control, 10 stressed plants). Due to very hot weather conditions in June 2010 the experiment was stopped after 8 days and 12 available control plants were used to restart the drought treatment with 6 control and 6 stressed plants on 11.06.2010 and all plants were measured on 15.06.2010 (DAY 5). The mean leaf temperatures at midday were: 25 ℃ (04.06.2010; DAY 0), 31.9 ℃ (07.06.2010; DAY 3), 30.8 ℃ (15.06.2010; DAY 5), 35.8 ℃ (10.06.2010; DAY 6) and 35.7 ℃ (12.06.2010; DAY 8). The mean PAR radiation per day (measured from 6:00 am till 7:00 pm) was 144.1 µmol m-2 s-1. Each plant was used only once for physiological measurements and sampling of leaves.On every day of the experiment (day 0, 3, 5, 6, 8) the pot weight and the volumetric soil moisture content (ThetaProbe ML2x and handheld data logger Moisture Meter HH2, Delta-T Devices, Cambridge, United Kingdom) was recorded. The water potential (PWSC Model 3000, Soilmoisture Equipment Corporation, Santa Barbara, USA) was determined for the 6th leaf (representing the insertion level of the shoot from the basis) of every plant and measurement day. Chlorophyll fluorescence and gas exchange parameters of light adapted leaves were determined with the 4th and 5th leaf, whereas dark adaptation was performed only with the 5th leaf. Immediately after these non-invasive measurements, the 5th leaf was harvested, frozen in liquid nitrogen and further used for the measurement of polyphenols, selected primary metabolites and volatiles (VOCs).
Click to Show/Hide
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor Function |
The content of different groups of primary and secondary metabolites is significantly influenced by severe drought stress in grapevine leaves. The content of the majority of the metabolites (around 60% of primary metabolites, around 85% of polyphenols and about 40% of the detected and identified VOCs) increased upon drought stress treatment. Among these especially the primary metabolites citric acid and glyceric acid were strongly influenced by the short as well as the prolonged drought stress treatment, whereas all polyphenols were only induced upon the prolonged drought stress treatment.
Click to Show/Hide
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor | Part | Location | NP Content | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Normal condition
|
Leaves | Vienna, Austria |
NP Content: 0.1 ± 0.1 µg/g dry weight
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Dry 3-5 days
|
Leaves | Vienna, Austria |
NP Content: 0.1 ± 0.0 µg/g dry weight
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Dry 6-8 days
|
Leaves | Vienna, Austria |
NP Content: 1.2 ± 1.6 µg/g dry weight
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||