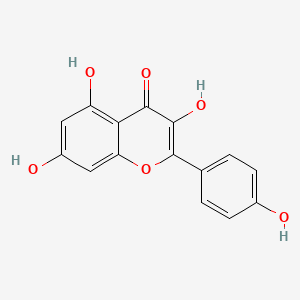
| The Content Variation of Natural Product Induced by Different Factor(s) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Species Name: Brassica juncea (var. RLC-1) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor Name: CdCl2 Treatment; Earthworms Treatment | [1] | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Experiment Detail |
The experiments were conducted under controlled conditions using plastic pots having lower diameter of 7.8 cm, upper diameter of 13.5 cm and 12 cm in height. The soil was collected from the top layer (0-20 cm) from the Botanical Garden of the university. Soil was air dried crushed and sieved through 2 mm filter autoclaved at 121 ℃ for 2 h. The soil was autoclaved to exclude soil pathogens and other microorganisms if any. The autoclaved soil was poured in pots and kept in the growth chamber. The pots were filled with 500 g uncontaminated soil and partially decayed compost (cow manure) (2:1) and was used as growing medium. The cow dung was added into the soil for better performance of earthworms. A subsample of the study soil before mixing with compost was analyzed for its physicochemical characteristics. The soil used for the experiment was sandy loam soil having pH 7.8 , EC (Electrical conductivity) (µS/cm) =184.25 , TDS (Total Dissolved Solids) (mg/kg) = 130 , N (Nitrogen) (mg/kg) = 103 , P (Phosphorus) (mg/kg) = 10.6 , K (Potassium) (mg/kg) = 0.343 , %OC = 0.894, Cd (mg/kg) = ND (not detected by AAS).The Cd treatment was given by using anhydrous CdCl2 (Minimum assay: 95.0%) procured from Hi-Media laboratories. The CdCl2 anhydrous was added to the soil to make different concentrations of Cd 0.50 mM, 0.75 mM, 1.00 mM, and 1.25 mM (i.e. 56 mg/Kg , 84 mg/Kg , 112 mg/Kg and 140 mg/Kg respectively). The various treatments given are as shown below:(1)C0 (Control): (Cadmium absence);(2)C1: (0.5 mM Cd);(3)C2: (0.75 mM Cd);(4)C3: (1.00 mM Cd);(5)C4: (1.25 mM Cd).Each Cd treatment was given in soils without as well as with earthworms (WTE = without, WE = with earthworms). Earthworms (3 earthworms per pot) were inoculated after seven days of Cd treatment and incubated for 7 d in soil with earthworms. The seeds after surface sterilization were sown in soil containing different concentration of Cd and earthworms in plastic pots. These pots were kept in seed germinator under controlled conditions i.e. 25 ℃ temperature and 16:8 h dark: light photoperiod (1700 lx) for 15 d. Seedlings were harvested after 15 d followed by washing with distilled water. The growth and biochemical analysis was done on these seedlings.
Click to Show/Hide
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor Function |
Increased Cd uptake in plants in presence of earthworms enhances the total antioxidative capacity, metal chelating compounds and content of other antioxidants in plants grown under metal polluted soils. Earthworms can improve plant growth by improving nutrient availability to plants through their vermicasting activity. Their role in modifying soil pH and increasing metal phytoavailability made their use ideal in phytoremediation of polluted soils. Increased uptake and accumulation of Cd in plants activates the antioxidative system of plants takes place by addition of earthworms to soil.
Click to Show/Hide
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Mechanism |
The gene expression for the key enzymes involved in organic acid metabolism was studied to understand the role of earthworms in organic acid metabolism in plants under Cd metal stress. It was observed that in comparison to control (C0) seedlings the expression of CS, SUCLG1, SDH and FH was enhanced 1.72, 1.58, 1.65 and 1.88 folds in seedlings given C4 treatment with 1.25 mM dose of Cd respectively . However, after supplementation of earthworms to Cd treated soils given C4 treatment resulted in further enhancement in expression of CS (2.53 fold), SUCLG1 (2.35 fold), SDH (2.13 fold) and FH (3.06 fold) .
Click to Show/Hide
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor | Part | Location | NP Content | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
0.5 mM CdCl2 + without earthworms
|
NA | Ludhiana, India. |
NP Content: 0.000494 ± 0.000023 mg/g
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
1.25 mM CdCl2 + with earthworms
|
NA | Ludhiana, India. |
NP Content: 0.010295 ± 0.000626 mg/g
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor Name: 24-epibrassinolide Treatment; Imidacloprid Treatment | [2] | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Experiment Detail |
Seeds of B. juncea (cv. RLC-1) were given pre-sowing treatment with 24-epibrassinolide (EBR) solutions (0 and 100 nM EBR/L) for 8 h. Petri-plates were lined with Whatman1 filter paper and were supplemented with different imidacloprid (IMI) concentrations (0, 150, 200, and 250 mg IMI/L). The EBR treated seeds were rinsed with distilled water and grown in Petri-plates supplemented with IMI solutions (three petri-plates for each treatment). The Petri-plates were kept in seed germinator (temperature = 25 ℃ , photoperiod = 16 h, light intensity = 175 µmol m -2 s-1) and the seedlings were harvested 10 days after sowing for further analysis.
Click to Show/Hide
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor Function |
Seed soaking with 24-epibrassinolide recovers the impaired growth of B. juncea seedlings under imidacloprid stress by modulating the expression of genes encoding key enzymes including chlorophyllase, citrate synthase, succinyl Co-A ligase, succinate dehydrogenase, fumarate hydratase, malate synthase, phytoene synthase, chalcone synthase, and phenylalanine ammonialyase.
Click to Show/Hide
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Mechanism |
In the present study, as compared to control seedlings, the expression of gene CHLASE (encoding chlorophyllase) was observed to increase by 2.66-fold under IMI toxicity, but seed soaking with EBR significantly reduced the expression of CHLASE to 1.07-fold in the seedlings under IMI toxicity . Data analysis using two-way ANOVA and Tukey's HSD showed significant difference for CHLASE expression in B. juncea seedlings (FIMI p < 0.01, FEBR p < 0.01, FIMI * EBR p < 0.001). MLR analysis of the fold change in CHLASE expression also revealed the increased expression of gene with IMI toxicity and EBR application (positive betaIMI-value), whereas interaction between IMI and EBR was observed to be negative .Further, in comparison to control seedlings, the expression of PSY (encoding phytoene synthase) and CHS (encoding chalcone synthase) was significantly enhanced by 5.22 and 4.54-folds respectively in the seedlings raised from EBR treated as well as untreated seeds grown under IMI stress . Significant differences in expression PSY (FIMI p < 0.001, FEBR P<0.05) and CHS (FIMI * EBR p < 0.001) were observed after analyzing the data using two-way ANOVA and Tukey's HSD. MLR analysis of fold change in gene expression also revealed the role of EBR in modulation of gene expression of PSY and CHS. Concentrations of IMI as well as EBR were regressed positively on the fold change in gene expression of PSY and CHS, thus revealing enhanced expressions of these genes under both the treatments. Moreover, interaction between IMI and EBR was positive for PSY expression, whereas negative interaction was observed for the expression of CHS .In the present study, the expression of PAL was also observed to enhance significantly by 6.68-fold in the seedlings raised from EBR treated seeds and grown under IMI stress . After analyzing the data using two-way ANOVA and Tukey's HSD, significant difference in the expression of PAL was observed (FIMI p < 0.01, FEBR p < 0.01, FIMI * EBR P<0.05). MLR analysis of the fold change in gene expression also confirmed the role of EBR in increasing the PAL gene expression under IMI pesticide stress. Positive beta-regression coefficients were observed for IMI, EBR, and IMI * EBR .The expression of genes encoding the key enzymes involved in organic acid metabolism was also studied to understand the role of EBR in organic acid metabolism under IMI pesticide stress. It was observed that as compared to control seedlings, the expression of CS (encoding citrate synthase, 2.35-fold), SUCLG1 (encoding succinyl-Co-A ligase, 1.57-fold), SDH (encoding succinate dehydrogenase, 2.01-fold), FH (encoding fumarate hydratase, 1.57-fold), and MS (encoding malate synthase, 1.91-fold) were increased in B. juncea seedlings raised from untreated seeds and grown under IMI pesticide toxicity . However, seed soaking with 100 nM EBR and germinating them under IMI toxicity resulted in further enhancement in expression of CS (2.61-fold), SUCLGD1 (4.18-fold), SDH (2.55-fold), FH (3.73-fold), and MS (4.03-fold). Data analysis using two-way ANOVA and Tukey's HSD showed significant differences in the expression of CS (FEBR p < 0.01, FIMI * EBR p < 0.01), SUCLG1 (FEBR p < 0.001, FIMI * EBR P<0.05), SDH (FEBR p < 0.01), FH (FEBR p < 0.001), and MS (FEBR p < 0.001). MLR analysis showed that gene expression in seedlings under IMI stress as well as after the EBR seed treatment was increased as indicated by positive beta-regression coefficients. Whereas, negative interactions were noticed between IMI and EBR treatments for the expression of all genes studied related to organic acid metabolism.
Click to Show/Hide
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor | Part | Location | NP Content | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
0 nM 24-epibrassinolide + 0 mg/L Imidacloprid
|
Fresh seedlings | NA |
NP Content: 0.0978 ± 0.01022 mg/g fresh weight
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
100 nM 24-epibrassinolide + 0 mg/L Imidacloprid
|
Fresh seedlings | NA |
NP Content: 0.0892 ± 0.01663 mg/g fresh weight
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
0 nM 24-epibrassinolide + 150 mg/L Imidacloprid
|
Fresh seedlings | NA |
NP Content: 0.10034 ± 0.01153 mg/g fresh weight
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
100 nM 24-epibrassinolide + 150 mg/L Imidacloprid
|
Fresh seedlings | NA |
NP Content: 0.09818 ± 0.01276 mg/g fresh weight
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
0 nM 24-epibrassinolide + 200 mg/L Imidacloprid
|
Fresh seedlings | NA |
NP Content: 0.16486 ± 0.03181 mg/g fresh weight
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
100 nM 24-epibrassinolide + 200 mg/L Imidacloprid
|
Fresh seedlings | NA |
NP Content: 0.08003 ± 0.01769 mg/g fresh weight
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
0 nM 24-epibrassinolide + 250 mg/L Imidacloprid
|
Fresh seedlings | NA |
NP Content: 0.08827 ± 0.01858 mg/g fresh weight
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
100 nM 24-epibrassinolide + 250 mg/L Imidacloprid
|
Fresh seedlings | NA |
NP Content: 0.21633 ± 0.02873 mg/g fresh weight
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Species Name: Chelidonium majus | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor Name: Locality Variation; Harvest Time Variation | [3] | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Experiment Detail |
Aerial parts from five populations of Chelidonium majus were collected from the wild at the flowering stage (hereafter referred to as 'wild') for chemical analysis and biological activity testing during May 2019. Ten randomly selected plantlets were also collected from the same five populations in 2019 and planted in an organically certified experimental field of IES (57° 19′ 11.7″ N 25° 19′ 18.8″ E, 115 m altitude). The plot size was 0.8 m2, and the plant spacing was 0.2 × 0.5 m. A year later, aerial parts were collected during the flowering stage from the same populations in the experimental field (hereafter referred to as 'cultivated').
Click to Show/Hide
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor Function |
The total content of alkaloids in aqueous ethanol extracts prepared from cultivated C. majus specimens was higher than that observed in extracts prepared from wild-grown plant material. Chelidonine, sanguinarine, and chelerythrine were the main contributors to the total increase in alkaloid content. The cultivation of C. majus did not significantly affect the total content of flavonol glycosides. The observed differences in the phytochemical compositions of the C. majus extracts resulted in significant increases in the cytotoxic activities of the preparations.
Click to Show/Hide
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor | Part | Location | NP Content | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Locality: wild + Harvesting time: 2019
|
Aerial parts | Latvia |
NP Content: 13.1 ± 9.2 µg/g
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Locality: experimental field + Harvesting time: 2020
|
Aerial parts | Latvia |
NP Content: 6.9 ± 4.2 µg/g
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Species Name: Fragaria × ananassa Duch. | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor Name: Nitrogen Treatment; AMF Inoculation | [4] | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Experiment Detail |
The experiment was conducted in a 'shade'-type greenhouse with 30% shade at the Instituto de Investigaciones Agropecuarias y Forestales (IIAF), Universidad Michoacana de San Nicolas de Hidalgo (UMSNH), Morelia, Michoacan, Mexico. Maximum and minimum temperatures in the greenhouse varied between 28 and 32 ℃ and between 8 and 18 ℃ respectively. Plants of the strawberry cultivar 'Aromas' were used that had previously been grown in a sterilised (95 ℃ water/steam, 40 min) substrate of coconut fibre/perlite (1:3 v/v) under greenhouse conditions. Before the experiment was established, the absence of AMF in the roots was verified by the ink and vinegar technique, modifying the duration of immersion in KOH and ink/vinegar solution (7 and 5 min respectively). Before planting, roots were disinfected by submerging them for 20 s in 20 g/L sodium hypochlorite solution and rinsing them in water. The inoculum was prepared with spores of Glomus intraradices cultivated in liquid medium (3.5 × 106 spores/L, 90% viability; Premier Tech Biotechnologies Company, Quebec, Canada), which was diluted with fitagel (Sigma P-8169, Saint Louis, MO, USA) solution at 50 g/L to obtain a final concentration of about 5 × 104 spores/L. The viability of spores was determined according to the method of An and Hendrix. Eighteen days after setting up the experiment, each plant received 2 mL of inoculum applied directly to the recently formed roots. One month later, after staining, the percentage of root colonisation was determined by the gridline intersect method. The experiment was organised as a full factorial, completely randomised design with two factors: inoculation (two levels: mycorrhizal and non-mycorrhizal plants) and N concentration in the nutrient solution (three levels: 3, 6 and 18 mmol/L). The six treatments were replicated four times, producing 24 experimental units with ten plants each. Every second day, all plants were irrigated up to substrate saturation. Nitrogen was supplied as NO and the cation/anion ratio was kept constant by varying the concentration of SO. When N was below 18 mmol/L, the cation concentrations were maintained as follows: K+, 3; Ca2+, 3.5; Mg2+, 1.5 mmol/L. They were increased in the 18 mmol/L N treatment: K+, 6.5; Ca2+, 7.5; Mg2+, 3.25 mmol/L. In all nutrient solutions the concentration of phosphorus (P) was 0.3 mmol/L. The other nutrients in the solutions were: H3BO3, 20; CuSO4. 5H2O, 0.5; Fe-EDTA (Ethylenediaminetetraacetic acid iron (III) sodium salt), 15; MnSO4.H2O, 12; (NH4)6Mo7O24 . 4H2O, 0.05; ZnSO4 . 7H2O, 3 µmol/L. The pH was adjusted to 5.5 at every application date.
Click to Show/Hide
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor Function |
Mycorrhization did not modify the weight, diameter or length of strawberry fruits but had a negative effect on most colour parameters. Moreover, fruits of mycorrhizal plants had higher K and Cu concentrations and showed greater accumulation of most phenolic compounds.
Click to Show/Hide
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor | Part | Location | NP Content | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Nitrogen concentration (mmol/L): 3
|
Mature fruits | Morelia, Michoacan, Mexico |
NP Content: 3.37 g/kg dry matter
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Nitrogen concentration (mmol/L): 6
|
Mature fruits | Morelia, Michoacan, Mexico |
NP Content: 3.71 g/kg dry matter
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Nitrogen concentration (mmol/L): 18
|
Mature fruits | Morelia, Michoacan, Mexico |
NP Content: 3.545 g/kg dry matter
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Glomus intraradices inoculation
|
Mature fruits | Morelia, Michoacan, Mexico |
NP Content: 3.692 g/kg dry matter
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Non-AMF inoculation
|
Mature fruits | Morelia, Michoacan, Mexico |
NP Content: 3.391 g/kg dry matter
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Nitrogen concentration (mmol/L): 3 + G. intraradices inoculation
|
Mature fruits | Morelia, Michoacan, Mexico |
NP Content: 3.564 g/kg dry matter
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Nitrogen concentration (mmol/L): 3 + Non-AMF inoculation
|
Mature fruits | Morelia, Michoacan, Mexico |
NP Content: 3.176 g/kg dry matter
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Nitrogen concentration (mmol/L): 6 + G. intraradices inoculation
|
Mature fruits | Morelia, Michoacan, Mexico |
NP Content: 4.198 g/kg dry matter
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Nitrogen concentration (mmol/L): 6 + Non-AMF inoculation
|
Mature fruits | Morelia, Michoacan, Mexico |
NP Content: 3.222 g/kg dry matter
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Nitrogen concentration (mmol/L): 18 + G. intraradices inoculation
|
Mature fruits | Morelia, Michoacan, Mexico |
NP Content: 3.314 g/kg dry matter
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Nitrogen concentration (mmol/L): 18 + Non-AMF inoculation
|
Mature fruits | Morelia, Michoacan, Mexico |
NP Content: 3.776 g/kg dry matter
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Nitrogen concentration (mmol/L): 18 + Non-AMF inoculation
|
Mature fruits | Morelia, Michoacan, Mexico |
NP Content: 0.012 g/kg dry matter
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Species Name: Gynostemma pentaphyllum | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor Name: Heat Stress Treatment; CO2 Treatment | [5] | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Experiment Detail |
The air temperature was controlled at 23/18 ℃ or 28/23 ℃ (day/night). Whilst the CO2 concentration was maintained at 360 or 720 µmol/mol. The temperature and CO2 treatments were randomly assigned in each of the four groups. One G. pentaphyllum plant (5-foliolate) was obtained from Beishan of Jinhua, Zhejiang Province and then planted in Zhejiang Normal University of botany experimental garden. Reproduction of new plants was used by cutting propagation. After 5 years we obtained sufficient plant material for this study. The seedlings were planted in a temperature-controlled greenhouse (24 ℃) from October to December (2014). Prior to treatment in a growth chamber, healthy plants were transplanted into pots (18 cm × 16 cm). The pots were filled with 3 kg of red soil combined with organic fertilizer of peat (19:1, w/w; total of organic matter content is approximately 60 g/kg). 50 plants were moved to each growth chamber. All plants were watered sparingly twice a week with 100 mL of modified Hoagland nutrient solution. The plant samples were evaluated at 60 days after treatment.
Click to Show/Hide
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor Function |
Elevated CO2 increased the level of total sugars and gypenoside A, but decreased the total antioxidant capacity and main antioxidant compounds in different organs of G. pentaphyllum. Also, TP content at CT was lower than C. Similarly, TP content of leaves significantly decreased at T compared to CK, with a drop of 25.65%. Furthermore, high temperature and elevated CO2 level significantly decreased the TP contents of leaves and stems. These results suggest that elevated CO2 and increased temperature does not favor accumulation of phenolics in G. pentaphyllum organs.
Click to Show/Hide
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor | Part | Location | NP Content | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Stem: Elevated CO2 (23/18 ℃, 720 µmol/mol CO2)
|
Stem | Zhejiang Normal University, Zhejiang Province, China |
NP Content: 0.58 mg/g dry weight
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Leaf: Elevated CO2 (23/18 ℃, 720 µmol/mol CO2)
|
Leaf | Zhejiang Normal University, Zhejiang Province, China |
NP Content: 36.14 mg/g dry weight
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Inflorescence: Elevated CO2 (23/18 ℃, 720 µmol/mol CO2)
|
Inflorescence | Zhejiang Normal University, Zhejiang Province, China |
NP Content: 11.66 mg/g dry weight
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Stem: Elevated temperature (28/23 ℃, 360 µmol/mol CO2)
|
Stem | Zhejiang Normal University, Zhejiang Province, China |
NP Content: 0 mg/g dry weight
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Leaf: Elevated temperature (28/23 ℃, 360 µmol/mol CO2)
|
Leaf | Zhejiang Normal University, Zhejiang Province, China |
NP Content: 50.5 mg/g dry weight
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Inflorescence: Elevated temperature (28/23 ℃, 360 µmol/mol CO2)
|
Inflorescence | Zhejiang Normal University, Zhejiang Province, China |
NP Content: 1.21 mg/g dry weight
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Stem: Elevated temperature and CO2 (28/23 ℃, 720 µmol/mol CO2)
|
Stem | Zhejiang Normal University, Zhejiang Province, China |
NP Content: 3.73 mg/g dry weight
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Leaf: Elevated temperature and CO2 (28/23 ℃, 720 µmol/mol CO2)
|
Leaf | Zhejiang Normal University, Zhejiang Province, China |
NP Content: 34.31 mg/g dry weight
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Inflorescence: Elevated temperature and CO2 (28/23 ℃, 720 µmol/mol CO2)
|
Inflorescence | Zhejiang Normal University, Zhejiang Province, China |
NP Content: 1.92 mg/g dry weight
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Species Name: Lentil var. Tina | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor Name: H2O2 Treatment; Mannitol Treatment; NaCl Treatment; High Temperature Treatment; Low Temperature Treatment | [6] | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Experiment Detail |
Seeds were sterilized in 1% (v/v) sodium hypochloride (Sigma-Aldrich, USA) for 10 min, then drained and washed with distilled water until they reached neutral pH. They were placed in distilled water and soaked for 6 h at 25 ℃ . Seeds were dark germinated for 8 days in a growth chamber (SANYO MLR-350H) on Petri dishes (125 mm) lined with absorbent paper. Seedlings were watered with 5 ml of Milli-Q water daily. Sprout (8-day-old) samples were gently collected, weighed (fresh mass), rapidly frozen and kept in polyethylene bags at -20 ℃ . For each treatment, three replicates were performed.Elicitation conditions were selected in previous screening studies. For the experiments, temperature (4 ℃ and 40 ℃ - TC and TH, respectively), H2O2 (20 mM and 200 mM - Ox1 and Ox2, respectively), mannitol (200 mM and 600 mM - Os1 and Os2, respectively) and NaCl (100 mM and 300 mM - S-Os1 and S-Os2, respectively) were selected as abiotic elicitors. All solutions were freshly prepared before each application. Mannitol (Os1, Os2), NaCl (S-O1, S-O2) and H2O2 (Ox1) treatments were applied by watering daily (not soaking) 2-day-old sprouts with 5 ml of test solution. For Ox2 (200 mM H2O2) treatment 2-day-old seedlings were only once watered with 5 ml of 200 mM H2O2 and then cultivated under standard conditions. For temperature conditioning treatment, 2-day-old sprouts were incubated at 4 ℃ and 40 ℃ (TC and TH, respectively) for 1 h and then cultivated under standard conditions. Sprout (8-day-old) samples were gently collected, weighed (fresh mass), rapidly frozen and kept in polyethylene bags at -20 ℃ .
Click to Show/Hide
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor Function |
Application of abiotic elicitors (environmental shocks) was an effective method for improvement of sprout pro-health potential via an increase of phenolic contents and subsequent elevation of antioxidant potential. Innovative application of elicitors on 2-day-old sprouts (not seed) allowed the elimination of the unfavorable influence of the factors employed on germination yield and biomass production. Assuming that the optimal germination conditions are those which most effectively increase the antioxidant potential without any negative influence on biomass accumulation and nutritional quality the elicitation with 20 mM H2O2 for the future applications is recommended.
Click to Show/Hide
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor | Part | Location | NP Content | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Normal condition
|
Sprouts | NA |
NP Content: 0.00269 ± 0.00011 mg/g flour
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Induction with 20 mM H2O2
|
Sprouts | NA |
NP Content: 0.00432 ± 0.0002 mg/g flour
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Induction with 200 mM H2O2
|
Sprouts | NA |
NP Content: 0.00009 ± 0.00004 mg/g flour
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Induction with 200 mM mannitol
|
Sprouts | NA |
NP Content: 0.00002 ± 0 mg/g flour
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Induction with 600 mM mannitol
|
Sprouts | NA |
NP Content: 0.00001 ± 0 mg/g flour
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Induction with 100 mM NaCl
|
Sprouts | NA |
NP Content: 0.0068 ± 0.00027 mg/g flour
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Induction with 300 mM NaCl
|
Sprouts | NA |
NP Content: 0.00002 ± 0 mg/g flour
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Induction at 4 ℃
|
Sprouts | NA |
NP Content: 0.00093 ± 0.00004 mg/g flour
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Induction at 40 ℃
|
Sprouts | NA |
NP Content: 0.0013 ± 0.00005 mg/g flour
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||