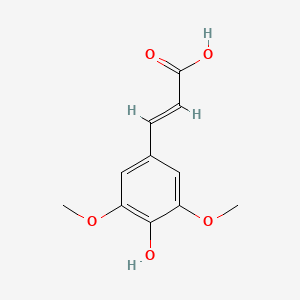
| The Content Variation of Natural Product Induced by Different Factor(s) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Species Name: Amaranthus tricolor genotype VA13 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor Name: NaCl Treatment | [1] | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Experiment Detail |
On the basis of previous studies, an antioxidant enriched high yield potential genotype (Accession VA13) was selected for this investigation. This genotype was grown in pots of a rain shelter open field of Bangabandhu Sheikh Mujibur Rahman Agricultural University, Bangladesh (AEZ-28, 24° 23′ north latitude, 90° 08′ east longitude, 8.4 m.s.l.). The seeds were sown in plastic pots (15 cm in height and 40 cm length and 30 cm width) in a randomized complete block design (RCBD) with three replications. N: P2O5:K2O were applied @92:48:60 kg/ha as a split dose. First, in pot soil, @46:48:60 kg ha 1 N: P2O5:K2O and second, at 7 days after sowing (DAS) @46:0:0 kg/ha N: P2O5:K2O. The genotype was grouped into three sets and subjected to four salinity stress treatments that are, 100 mM NaCl, 50 mM NaCl, 25 mM NaCl, and control or no saline water (NS). Pots were well irrigated with fresh water every day up to 10 days after sowing (DAS) of seeds for proper establishment and vigorous growth of seedlings. Imposition of salinity stress treatment was started at 11 DAS and continued up to 40 DAS (edible stage). Saline water (100 mM NaCl, 50 mM NaCl and 25 mM NaCl) and fresh water were applied to respective pots once a day. At 40 DAS the leaves of Amaranthus tricolor were harvested. All the parameters were measured in six samples.
Click to Show/Hide
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor Function |
At Moderate salinity stress (MSS) and Severe salinity stress (SSS) conditions, leaf color parameters and pigments, vitamins, phenolic acids, flavonoids and antioxidant capacity of A. tricolor leaves were very high compared to control condition. Hence, salt-stressed A. tricolor leaves had a good source of natural antioxidants compared to plant grown in normal cultivation practices.
Click to Show/Hide
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor | Part | Location | NP Content | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
No saline water (Control)
|
Leaves | Bangabandhu |
NP Content: 0.35 ± 0.01 µg/g fresh weight
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
25 mM NaCl (Low salinity stress)
|
Leaves | Bangabandhu |
NP Content: 0.36 ± 0.01 µg/g fresh weight
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
50 mM NaCl (Moderate salinity stress)
|
Leaves | Bangabandhu |
NP Content: 0.43 ± 0.01 µg/g fresh weight
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
100 mM NaCl (Severe salinity stress)
|
Leaves | Bangabandhu |
NP Content: 0.45 ± 0.01 µg/g fresh weight
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Species Name: Brassica oleracea L., var. italica, cv. Waltham 29 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor Name: Methyl Jasmonate Treatment; UV Radiation Treatment | [2] | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Experiment Detail |
Briefly, broccoli seeds (0.5 g per replication) were sanitized for 15 min in sodium hypochlorite (1.5%, v/v), rinsed with Milli-Q water and soaked with aeration overnight in darkness and at room temperature. After pouring off the soaking water, the seeds were spread evenly on standard 200 square cell plug trays (21.38 × 11.05 × 1.75) containing Canadian Sphagnum peat moss previously moistened. Sprouts were grown in a culture room with controlled temperature (25 ℃ ) and a photoperiod regime with cycles of 16 h light and 8 h darkness. Water (control) or a phytohormone solution were atomized every 12 h throughout the experiment.Six trays with broccoli sprouts seeds were prepared for this study, and were assigned for (A) Control (no UV or phytohormone application), (B) UVA treatment, (C) UVB treatment, (D) MJ treatment, (E) UVA + MJ treatment, and (F) UVB + MJ treatment.MJ treatments (D, E and F) were conducted based on Perez-Balibrea et al. with slight adjustments. Briefly, methyl jasmonate (MJ) was dissolved in 0.2% ethanol to obtain a 25 µM solution and applied every 12 h by exogenous spraying 65 mL of 25 µM MJ solution from sowing day until the end of the experiment (8th day after sowing). Due to the volatility of MJ and to avoid the fact that treatment to one tray may result in application to neighboring trays, the MJ solution was applied using physical separation. Control sprouts (A) and sprouts treated with UVA or UVB alone (B and C) were irrigated with the same frequency using 65 mL of Milli-Q water containing 0.04% ethanol.On the 7th day after sowing, UV (B and C) and UV + MJ (E and F) treatments were carried out in special UVA and UVB chambers based on Moreira-Rodriguez et al. with slight adjustments. Chambers used for treatments B and E were equipped with two 40 W UVA lamps (Sylvania F40W T12 BL350, Ledvance LLC., Wilmington, MA, USA), while chambers for treatments C and F consisted of two 40 W UVB lamps (Philips TL 40W/12 RS, Philips, Ljubljana, Slovenia). Trays with broccoli sprouts were placed 30 cm below the irradiation source. All UV treatments consisted of a single exposure for 120 min. The irradiation intensities were determined prior to the experiment as 9.47 and 7.16 W/m2 for UVA and UVB, respectively, using a PMA 2200 radiometer equipped with PMA 2110 UVA and PMA 2106 UVB sensors (Solar Light, Glenside, PA, USA) measuring in the spectral range from 320-400 nm and 280-320 nm, respectively.After UV treatments, trays were returned to culture room and the proper irrigation with water or MJ solution continued for an additional (acclimatization) period of 24 h. Sprouts of all six trays were harvested at the 8th day after sowing, immediately flash-frozen in liquid nitrogen, placed at -80 ℃ , freeze-dried (Labconco, Kansas City, MO, USA), and then ground to a fine powder. Samples were stored at -80 ℃ until further analysis.
Click to Show/Hide
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor Function |
Simple pre-harvest treatments such as UV radiation, applied alone or in combination with exogenous MJ, can be used as an effective emerging technology that allows the accumulation of specific phytochemicals in broccoli sprouts. Furthermore, results demonstrated that the profile of glucosinolates accumulated in stressed broccoli sprouts could be tailored towards the over-production of most indole glucosinolates by applying 25 µM MJ alone or preferably in combination with a 120 min exposure to UVA or UVB radiation (9.47 and 7.16 W/m2, respectively) 24 h prior harvest. Specifically, a synergistic effect in the accumulation of NGBS was achieved by combining UV and MJ stresses. On the other hand, the production of aliphatic or specific indole glucosinolates can be triggered by UVB supplementation alone. MJ treatments may be applied if an increase in gallic acid, its derivative GAH II, specific sinapic acid derivatives (e.g., 5-SQA) and ferulic acid derivatives (e.g., 1,2-diFG) is desired. However, such increases would be at the expense of the following compounds: GAH I, GTA, diGH, 3-O-H-K, 1-O-S-beta-d-g, sinapoyl malate, sinapic acid, K-3-O-S-so-7-O-g, 1,2-diSG, 1-S-2-FG, the majoritarian isomer of 1,2,2-triSG and 1,2-diS-1-FG; as they were significantly reduced after treatments with MJ. Application of UVA alone may be recommended to accumulate GAH I, 1-O-S-beta-d-g, sinapic acid, gallic acid, K-3-O-S-so-7-O-g, 1-S-2-FG and the second isomer of 1,2,2-triSG. Finally, a single 120 min exposure to UVA radiation should be applied to increase xanthophyll and chlorophyll content in broccoli sprouts.
Click to Show/Hide
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor | Part | Location | NP Content | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
25 µ Methyl Jasmonate
|
Sprouts | NA |
NP Content: 154.2 ± 1.5 mg/kg dry weight
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Control (no UV or phytohormone application)
|
Sprouts | NA |
NP Content: 295.2 ± 34.1 mg/kg dry weight
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
UVA treatment (Irradiation intensities: 9.47 W/m2)
|
Sprouts | NA |
NP Content: 363.4 ± 15.1 mg/kg dry weight
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
UVB treatment (Irradiation intensities: 7.16 W/m2)
|
Sprouts | NA |
NP Content: 265.2 ± 24.6 mg/kg dry weight
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
UVA + 25 µ Methyl Jasmonate
|
Sprouts | NA |
NP Content: 153.2 ± 1.0 mg/kg dry weight
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
UVB + 25 µ Methyl Jasmonate
|
Sprouts | NA |
NP Content: 163.0 ± 2.4 mg/kg dry weight
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||