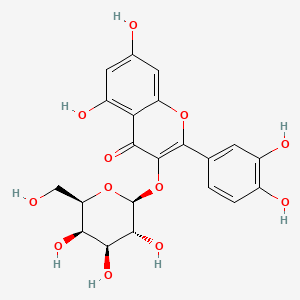
| The Content Variation of Natural Product Induced by Different Factor(s) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Species Name: Amaranthus tricolor genotype VA13 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor Name: NaCl Treatment | [1] | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Experiment Detail |
On the basis of previous studies, an antioxidant enriched high yield potential genotype (Accession VA13) was selected for this investigation. This genotype was grown in pots of a rain shelter open field of Bangabandhu Sheikh Mujibur Rahman Agricultural University, Bangladesh (AEZ-28, 24° 23′ north latitude, 90° 08′ east longitude, 8.4 m.s.l.). The seeds were sown in plastic pots (15 cm in height and 40 cm length and 30 cm width) in a randomized complete block design (RCBD) with three replications. N: P2O5:K2O were applied @92:48:60 kg/ha as a split dose. First, in pot soil, @46:48:60 kg ha 1 N: P2O5:K2O and second, at 7 days after sowing (DAS) @46:0:0 kg/ha N: P2O5:K2O. The genotype was grouped into three sets and subjected to four salinity stress treatments that are, 100 mM NaCl, 50 mM NaCl, 25 mM NaCl, and control or no saline water (NS). Pots were well irrigated with fresh water every day up to 10 days after sowing (DAS) of seeds for proper establishment and vigorous growth of seedlings. Imposition of salinity stress treatment was started at 11 DAS and continued up to 40 DAS (edible stage). Saline water (100 mM NaCl, 50 mM NaCl and 25 mM NaCl) and fresh water were applied to respective pots once a day. At 40 DAS the leaves of Amaranthus tricolor were harvested. All the parameters were measured in six samples.
Click to Show/Hide
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor Function |
At Moderate salinity stress (MSS) and Severe salinity stress (SSS) conditions, leaf color parameters and pigments, vitamins, phenolic acids, flavonoids and antioxidant capacity of A. tricolor leaves were very high compared to control condition. Hence, salt-stressed A. tricolor leaves had a good source of natural antioxidants compared to plant grown in normal cultivation practices.
Click to Show/Hide
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor | Part | Location | NP Content | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
No saline water (Control)
|
Leaves | Bangabandhu |
NP Content: 1.35 ± 0.02 µg/g fresh weight
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
25 mM NaCl (Low salinity stress)
|
Leaves | Bangabandhu |
NP Content: 1.33 ± 0.01 µg/g fresh weight
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
50 mM NaCl (Moderate salinity stress)
|
Leaves | Bangabandhu |
NP Content: 2.43 ± 0.01 µg/g fresh weight
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
100 mM NaCl (Severe salinity stress)
|
Leaves | Bangabandhu |
NP Content: 2.44 ± 0.02 µg/g fresh weight
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Species Name: Chelidonium majus | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor Name: Locality Variation; Harvest Time Variation | [2] | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Experiment Detail |
Aerial parts from five populations of Chelidonium majus were collected from the wild at the flowering stage (hereafter referred to as 'wild') for chemical analysis and biological activity testing during May 2019. Ten randomly selected plantlets were also collected from the same five populations in 2019 and planted in an organically certified experimental field of IES (57° 19′ 11.7″ N 25° 19′ 18.8″ E, 115 m altitude). The plot size was 0.8 m2, and the plant spacing was 0.2 × 0.5 m. A year later, aerial parts were collected during the flowering stage from the same populations in the experimental field (hereafter referred to as 'cultivated').
Click to Show/Hide
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor Function |
The total content of alkaloids in aqueous ethanol extracts prepared from cultivated C. majus specimens was higher than that observed in extracts prepared from wild-grown plant material. Chelidonine, sanguinarine, and chelerythrine were the main contributors to the total increase in alkaloid content. The cultivation of C. majus did not significantly affect the total content of flavonol glycosides. The observed differences in the phytochemical compositions of the C. majus extracts resulted in significant increases in the cytotoxic activities of the preparations.
Click to Show/Hide
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor | Part | Location | NP Content | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Locality: wild + Harvesting time: 2019
|
Aerial parts | Latvia |
NP Content: 220.2 ± 269.9 µg/g
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Locality: experimental field + Harvesting time: 2020
|
Aerial parts | Latvia |
NP Content: 195.9 ± 114.0 µg/g
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||