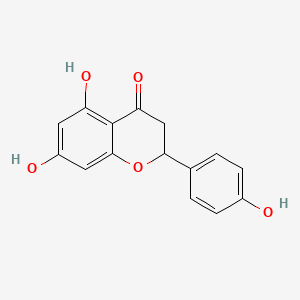
| The Content Variation of Natural Product Induced by Different Factor(s) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Species Name: Chrysanthemum morifolium Ram. cv. 'Gaya yellow' | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor Name: Photosynthetic Active Radiation Treatment | [1] | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Experiment Detail |
After acclimation, the plants were grown independently in different light treatment chambers at 20 ± 0.2 ℃ and 65 ± 2% humidity until the harvest date (35 days after light treatment). The white fluorescent light (70 ± 5 µmol/m2/s) was maintained for 12 h, and then each of the blue, green, red, and white lights was irradiated at 70 ± 5 µmol/m2/s for 4 h using LED arrays (DR LED Networks Co., Seoul, Republic of Korea). The spectral energy distribution of four different LED arrays was measured from 300 to 800 nm with a spectroradiometer (International Light, RPS-900, U.S.). Their maximum spectral wavelengths were 463 (blue), 518 (green), and 632 nm (red); the white LEDs had a broad spectrum. Irradiance was measured using a quantum sensor (LI-COR, LI-191, Lincoln, NE, U.S.). Water was supplied daily with top irrigation and a nutrient solution (Hoagland, pH = 5.9 ± 0.2, electrical conductivity = 1.2 dS/m) every 4 days until harvest.
Click to Show/Hide
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor Function |
A quantitation and principal component analysis biplot demonstrated that luteolin-7-O-glucoside (2), luteolin-7-O-glucuronide (3), and quercetagetin-trimethyl ether (8) were the highest polyphenols yielded under green light, and dicaffeoylquinic acid isomer (4), dicaffeoylquinic acid isomer (5), naringenin (7), and apigenin-7-O-glucuronide (6) were greatest under red light. Chlorogenic acid (1) and 1,2,6-trihydroxy-7,8-dimethoxy-3-methylanthraquinone (9) were produced in similar concentrations under both light types. The white and blue light appeared inefficient for polyphenol production.
Click to Show/Hide
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor | Part | Location | NP Content | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Under blue light (spectral wavelengths: 463 nm)
|
leaf | Korea |
NP Content: 72.2 ± 3.9 mg/kg
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Under green light (spectral wavelengths: 518 nm)
|
leaf | Korea |
NP Content: 90.7 ± 2.7 mg/kg
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Under red light (spectral wavelengths: 632 nm)
|
leaf | Korea |
NP Content: 97.4 ± 5.9 mg/kg
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Under white light (spectral wavelengths: broad spectrum)
|
leaf | Korea |
NP Content: 89.7 ± 0.8 mg/kg
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Species Name: Lentil var. Tina | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor Name: H2O2 Treatment; Mannitol Treatment; NaCl Treatment; High Temperature Treatment; Low Temperature Treatment | [2] | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Experiment Detail |
Seeds were sterilized in 1% (v/v) sodium hypochloride (Sigma-Aldrich, USA) for 10 min, then drained and washed with distilled water until they reached neutral pH. They were placed in distilled water and soaked for 6 h at 25 ℃ . Seeds were dark germinated for 8 days in a growth chamber (SANYO MLR-350H) on Petri dishes (125 mm) lined with absorbent paper. Seedlings were watered with 5 ml of Milli-Q water daily. Sprout (8-day-old) samples were gently collected, weighed (fresh mass), rapidly frozen and kept in polyethylene bags at -20 ℃ . For each treatment, three replicates were performed.Elicitation conditions were selected in previous screening studies. For the experiments, temperature (4 ℃ and 40 ℃ - TC and TH, respectively), H2O2 (20 mM and 200 mM - Ox1 and Ox2, respectively), mannitol (200 mM and 600 mM - Os1 and Os2, respectively) and NaCl (100 mM and 300 mM - S-Os1 and S-Os2, respectively) were selected as abiotic elicitors. All solutions were freshly prepared before each application. Mannitol (Os1, Os2), NaCl (S-O1, S-O2) and H2O2 (Ox1) treatments were applied by watering daily (not soaking) 2-day-old sprouts with 5 ml of test solution. For Ox2 (200 mM H2O2) treatment 2-day-old seedlings were only once watered with 5 ml of 200 mM H2O2 and then cultivated under standard conditions. For temperature conditioning treatment, 2-day-old sprouts were incubated at 4 ℃ and 40 ℃ (TC and TH, respectively) for 1 h and then cultivated under standard conditions. Sprout (8-day-old) samples were gently collected, weighed (fresh mass), rapidly frozen and kept in polyethylene bags at -20 ℃ .
Click to Show/Hide
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor Function |
Application of abiotic elicitors (environmental shocks) was an effective method for improvement of sprout pro-health potential via an increase of phenolic contents and subsequent elevation of antioxidant potential. Innovative application of elicitors on 2-day-old sprouts (not seed) allowed the elimination of the unfavorable influence of the factors employed on germination yield and biomass production. Assuming that the optimal germination conditions are those which most effectively increase the antioxidant potential without any negative influence on biomass accumulation and nutritional quality the elicitation with 20 mM H2O2 for the future applications is recommended.
Click to Show/Hide
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor | Part | Location | NP Content | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Normal condition
|
Sprouts | NA |
NP Content: 0.00005 ± 0.0001 mg/g flour
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Induction with 20 mM H2O2
|
Sprouts | NA |
NP Content: 0.00267 ± 0.00062 mg/g flour
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Induction with 200 mM H2O2
|
Sprouts | NA |
NP Content: 0.00174 ± 0.00064 mg/g flour
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Induction with 200 mM mannitol
|
Sprouts | NA |
NP Content: 0.00053 ± 0.00006 mg/g flour
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Induction with 600 mM mannitol
|
Sprouts | NA |
NP Content: 0.00171 ± 0.00017 mg/g flour
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Induction with 100 mM NaCl
|
Sprouts | NA |
NP Content: 0.00023 ± 0.00014 mg/g flour
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Induction with 300 mM NaCl
|
Sprouts | NA |
NP Content: 0.00074 ± 0.00006 mg/g flour
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Induction at 4 ℃
|
Sprouts | NA |
NP Content: 0.00149 ± 0.00003 mg/g flour
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Induction at 40 ℃
|
Sprouts | NA |
NP Content: 0.0006 ± 0.00022 mg/g flour
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Species Name: Streptomyces sp. BT01 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor Name: ISP-2 medium | [3] | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Experiment Detail |
Streptomyces sp. BT01 was isolated from the root tissues of Boesenbergia rotunda (L.) Mansf. A spore suspension of Streptomyces sp. BT01 was prepared in distilled water from cultures grown on ISP-4 medium at 30 ℃ for 10 days. The suspension, 108 spores per 100 ml of liquid medium, was added to ISP-2 broth in each 500-ml Erlenmeyer flask. Cultures were kept on a shaker at 120 rpm at 30 ℃ for 48 h and used as seed stocks. For large production of culture filtrates, the strain BT01 was grown in a modified 3000 ml glass container containing 1500 ml of ISP-2 broth, and incubated in an orbital shaker for 5 days in the same condition.
Click to Show/Hide
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor | Part | Location | NP Content | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
ISP-2 medium (30℃ + 5 days)
|
Roots | NA |
NP Content: 8 mg
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Species Name: Thymus daenensis | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor Name: NaCl Treatment | [4] | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Experiment Detail |
The seeds were germinated in seedling plastic tray filled up with a mixture of peat moss and coco peat (1:1). The trays were placed in a glass house at the University of Massachusetts, Amherst, USA, in September 2015. After two weeks, seedlings were transplanted into plastic pots (30 cm diameter and 30 cm height, three seedlings per pot). The pots growth media consisted of 2:1:1 ratio of sieved field soil, sand, and leaf mold, respectively. The medium in each pot was supplemented with 5.8 mg P, 3.3 mg N, and 13.8 mg K to warrant the plant growth; the electrical conductivity was 0.3 mS/cm (Beckman EC meter instrument. cedar grove, New jersey, USA); the pH was 8.04. Plants were maintained at the temperature of 18-25 ℃ ; glass house conditions were set as follows: photoperiod, 16 h; relative humidity, 60-70%; light intensity, 180 µmol m 2 s-1. The source of light was a high pressure sodium lamp in addition to the day light in order to attain 16 h of light per day. Fourty days after planting (establishment period), the uniform sized plants were treated with four different salinity levels including: 0, 30, 60, and 90 mM NaCl. The irrigation with saline solution (250 mL in each pots) was performed every two days . Total amount of NaCl per pot during experiment was 8.75 L. To avoid osmotic shock the salinity treatment started with 15 mM NaCl, and was progressively increased (every two days) to reach the maximum salinity level in each treatment. To prevent water and nutrient leaching, a plastic dish was inserted under each pot and the leached water was given back to pots.
Click to Show/Hide
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor Function |
Salinity has negative impact on plant production and stimulates several physiological and biochemical modifications in thyme species. Here, a major decline in dry matter, relative water content (RWC), photosynthetic pigment contents, K+, Ca+ as well as increase in Na+ and EL was observed. Our work showed that T. daenensis was similar to T.vulgaris in terms of tolerance to high levels of salinity stress and both species were moderately tolerant to severe salt stress. It should be noted that T. daenensis, as an Iranian endemic species, competes with commercial species like T. vulgaris as a rich source of phenolic compounds. Furthermore, the relative salt tolerance of both species could be related to the exclusion of Na+ from the vascular system in order to protect tissues from salt toxicity and also to the increase of phenolic content and radical scavenging activity. Thus, the plant behavior under salinity stress may be used to boost the production of bioactive compounds to be used on an industrial level for the manufacture of nutraceuticals, functional foods and cosmetics.
Click to Show/Hide
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor | Part | Location | NP Content | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
0 mM NaCl
|
Leaves | Isfahan, Iran |
NP Content: 1.43 ± 0.17 %
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
30 mM NaCl
|
Leaves | Isfahan, Iran |
NP Content: 1.42 ± 0.16 %
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
60 mM NaCl
|
Leaves | Isfahan, Iran |
NP Content: 1.72 ± 0.18 %
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
90 mM NaCl
|
Leaves | Isfahan, Iran |
NP Content: 1.22 ± 0.03 %
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Species Name: Thymus vulgaris | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor Name: NaCl Treatment | [4] | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Experiment Detail |
The seeds were germinated in seedling plastic tray filled up with a mixture of peat moss and coco peat (1:1). The trays were placed in a glass house at the University of Massachusetts, Amherst, USA, in September 2015. After two weeks, seedlings were transplanted into plastic pots (30 cm diameter and 30 cm height, three seedlings per pot). The pots growth media consisted of 2:1:1 ratio of sieved field soil, sand, and leaf mold, respectively. The medium in each pot was supplemented with 5.8 mg P, 3.3 mg N, and 13.8 mg K to warrant the plant growth; the electrical conductivity was 0.3 mS/cm (Beckman EC meter instrument. cedar grove, New jersey, USA); the pH was 8.04. Plants were maintained at the temperature of 18-25 ℃ ; glass house conditions were set as follows: photoperiod, 16 h; relative humidity, 60-70%; light intensity, 180 µmol m 2 s-1. The source of light was a high pressure sodium lamp in addition to the day light in order to attain 16 h of light per day. Fourty days after planting (establishment period), the uniform sized plants were treated with four different salinity levels including: 0, 30, 60, and 90 mM NaCl. The irrigation with saline solution (250 mL in each pots) was performed every two days . Total amount of NaCl per pot during experiment was 8.75 L. To avoid osmotic shock the salinity treatment started with 15 mM NaCl, and was progressively increased (every two days) to reach the maximum salinity level in each treatment. To prevent water and nutrient leaching, a plastic dish was inserted under each pot and the leached water was given back to pots.
Click to Show/Hide
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor Function |
Salinity has negative impact on plant production and stimulates several physiological and biochemical modifications in thyme species. Here, a major decline in dry matter, relative water content (RWC), photosynthetic pigment contents, K+, Ca+ as well as increase in Na+ and EL was observed. Our work showed that T. daenensis was similar to T.vulgaris in terms of tolerance to high levels of salinity stress and both species were moderately tolerant to severe salt stress. It should be noted that T. daenensis, as an Iranian endemic species, competes with commercial species like T. vulgaris as a rich source of phenolic compounds. Furthermore, the relative salt tolerance of both species could be related to the exclusion of Na+ from the vascular system in order to protect tissues from salt toxicity and also to the increase of phenolic content and radical scavenging activity. Thus, the plant behavior under salinity stress may be used to boost the production of bioactive compounds to be used on an industrial level for the manufacture of nutraceuticals, functional foods and cosmetics.
Click to Show/Hide
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor | Part | Location | NP Content | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
0 mM NaCl
|
Leaves | Isfahan, Iran |
NP Content: 0.44 ± 0.13 %
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
30 mM NaCl
|
Leaves | Isfahan, Iran |
NP Content: 0.49 ± 0.29 %
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
60 mM NaCl
|
Leaves | Isfahan, Iran |
NP Content: 0.65 ± 0.21 %
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
90 mM NaCl
|
Leaves | Isfahan, Iran |
NP Content: 0.86 ± 0.16 %
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||