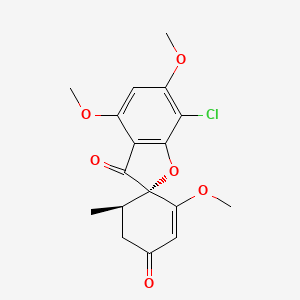
| The Content Variation of Natural Product Induced by Different Factor(s) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Species Name: cubensis sp. SNB-GCI02 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor Name: PDA medium | [1] | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Experiment Detail |
Isolation of endophytes: The leaves of 24 plant species were collected in the Amazon Rainforest biome, in Cayenne and Roura, French Guiana. After collection, the plant material was washed with sterile water and surface sterilised by sequential immersion in 70% aqueous ethanol (3 min), followed by 5% aqueous sodium hypochlorite (5 min) and finally by 70% aqueous ethanol (1 min). After these procedures, the leaves were rinsed with sterilised water. The residual sterilised water was incubated in Petri dishes to ensure that all the epiphytic microorganisms were eliminated. The surface-sterilised leaves were cut into 64 small pieces which were placed in 2 ml Eppendorf tubes containing 1 ml of potato dextrose agar medium (PDA, Fluka Analytical, Germany) at 26 ℃. Each individual hyphal tip of emerging fungi was removed and placed on a sterile PDA culture medium in 10 cm Petri dishes. The leaf fragments were cultured for a maximum of 5 months. All strains, including bacteria, were cultivated on solid PDA medium at 26 ℃ for 15 days, on 3 Petri dishes of 14 cm diameter (150 cm2). On the large scale, the microorganisms were cultivated under the identical conditions with 130 Petri dishes of 14 cm diameter (2 m2).
Click to Show/Hide
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor | Part | Location | NP Content | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
PDA medium (26℃ + 15 days)
|
Leaves | Cayenne and Roura, French Guiana |
NP Content: 4.2 mg
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Species Name: Xylaria flabelliformis isolate G536 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor Name: Rice Medium | [2] | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Experiment Detail |
The fungus Xylaria sp. PSU-G12 was isolated from a branch of G. hombroniana, collected from Songkhla province, Thailand, in the year 2005. The Xylaria sp. fungus PSU-G12 was grown on potato dextrose agar (PDA) at 25o C for 5 days. Three pieces (0.5 × 0.5 cm2 ) of mycelial agar plugs were inoculated into 500 mL Erlenmeyer flasks containing 300 mL potato dextrose broth (PDB) at room temperature for 3 weeks.
Click to Show/Hide
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor | Part | Location | NP Content | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Rice Medium (22 degrees Celsius + 14-21Days)
|
surface sterilized twigs | Pfafftown, NC, USA |
NP Content: 1.06 mg
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Species Name: Xylaria sp. G12 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor Name: PDB Medium | [3] | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Experiment Detail |
The endophytic fungus Coniothyrium sp., internal strain No. zw86, was isolated from the plant Salsola oppostifolia, growing on Gomera, in the Canary Islands. It was cultivated on 12 l of 5% w/v biomalt solid agar medium at room temperature for 28 days.
Click to Show/Hide
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor | Part | Location | NP Content | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
PDB Medium (25 degrees Celsius + 21Days)
|
branch | Songkhla province, Thailand |
NP Content: 1 mg
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||