| The Content Variation of Natural Product Induced by Different Factor(s) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Species Name: Aspergillus flocculosus strain CBS 112784 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor Name: Rice medium | [1] | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Experiment Detail |
Fungal material: The endophytic fungus was isolated from the fresh stems of Markhamia platycalyx family (Bignoniaceae) collected in October 2010 from Al-Zohriya gardens (Al-Zamalek, Giza, Egypt). Small-scale fermentation: A small-scale fermentation was carried out in two Erlenmeyer flasks (1 L each) on rice medium, which was prepared with 100 g of rice powder and approximately 100 mL of demineralized water just enough to cover the rice layer. The rice media was autoclaved prior to fungal inoculation. A 15-day fungal inoculum grown on petri dish was inoculated on the sterile rice medium and was allowed to grow at room temperature under static condition for 30 days. Medium-scale 30-day rice culture fermentation: A medium scale fermentation was performed in 10 Erlenmeyer flasks (1 L each) on rice solid medium for 30 days under same condition applied to small scale culture.
Click to Show/Hide
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor | Part | Location | NP Content | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Medium-scale fermentation: Rice medium (25℃ + 30 days)
|
Fresh stems | Al-Zamalek, Giza, Egypt |
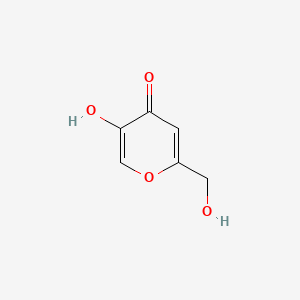
NP Content: 4.8 mg
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| References | ||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 1 | Isolation of anticancer and anti-trypanosome secondary metabolites from the endophytic fungus Aspergillus flocculus via bioactivity guided isolation and MS based metabolomics | |||||||||||||||||||||||