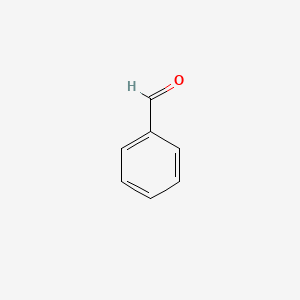
| The Content Variation of Natural Product Induced by Different Factor(s) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Species Name: Arabidopsis thaliana | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor Name: T-DNA Knock-Out Treatment | [1] | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Experiment Detail |
In vitro cultivation of Arabidopsis wildtype and mutant plants: Seeds were sterilized according to standard lab routines (EtOH, NaOCl/NaOH) prior to aseptical (in vitro) cultivation in 500 ml screw cap jars on MS medium (4.3 g/l; 50 ml/jar) containing Bacto- and Phytoagar (1:2; 6 g/l) and 30 g/l sucrose. Ten seeds were pipetted into each jar and plants grown for 6 weeks until flowering at a temperature of 20 ℃ under a 16/8 h day/ night regime using fluorescent tubes (Osram Lumilux Plus Eco 36 W). Both Arabidopsis thaliana wildtype plants of ecotype Columbia-0 (Col) and 4 Col-derived T-DNA knock-out mutants (homozygous lines) showing deficiencies in the GLS biosynthesis pathway were used in this study (five parallels for wildtype and mutants): TGG1 (Atg526000; Salk_130469), TGG2 (At5g25980; Salk_038730), Cyp83A1 (At4g13770) and Cyp83B1 (At4g31500; Salk_028573). Greenhouse-cultivation of Arabidopsis ecotypes: The following Arabidopsis ecotypes were used in the study: Columbia (Col), Cape Verde Islands (Cvi), Landsberg erecta (Ler) and Wassilewskija (Ws). Single plants were greenhouse-cultivated on fertilized soil (P-Jord; Emmaljunga Torvmull AB) in plug trays (9 × 6 cells) at a temperature of 20 ℃ (three parallels for each ecotype). Due to the 6-weeks growth period (November/December 2003), the plants were cultivated under a 16/8 h day/night regime using metal halide lamps (Osram HQI-T 400 W) placed 130 cm above the trays. Depending on the ecotypical plant development, whole plants were sampled after 3-4 weeks right before bolting for in vivo studies, while investigations of single plant organs (leaf, stem, inflorescence) were carried out after 5-6 weeks of cultivation.
Click to Show/Hide
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor Function |
Metabolites from methionine, leucine and phenylalanine-derived glucosinolates were most abundant (4-methylthiobutyl, 4-methylpentyl, 2-phenylethyl). In addition, 24 monoterpenes, 26 sesquiterpenes and 12 aromatic structures, predominantly observed in inflorescenses, are described. Excluding the vast group of straight chain aliphatic structures, a total of 102 volatile compounds were detected, of which 59 are reported in Arabidopsis thaliana for the first time, thus emphasizing the sensitivity and applicability of solid-phase microextraction for volatile profiling of plant secondary metabolites.
Click to Show/Hide
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor | Part | Location | NP Content | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Stem: A. thaliana wildtype detected in vivo
|
Stems | Norway |
NP Content: 0.09 %
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Species Name: Artemisia absinthium | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor Name: Chemotype Comparison | [2] | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Experiment Detail |
Ten different plants of wormwood were collected in March 1997 from each one of the following four wild populations in the Spanish Pyrenees: Tallo de Aulet (prov. Huesca) and Pont de Suert, Sort and Farga de Moles (prov. Lleida). In three of the four populations studied, there was another chemotype, with 25-65% of cis-epoxyocimene and 15-50% of chrysanthenyl acetate. This chemotype, called chemotype B, was less frequent in the Pyrenees than the chemotype A, appearing only in 17% of the samples (two samples in TallO de Aulet and in Pont de Suert and three samples in Farga de Moles).
Click to Show/Hide
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor Function |
Two chemotypes were detected; a cis-epoxyocimene type (with more than 50% of this compound) which was predominant in all the populations, and a cis-epoxyocimene + chrysanthenyl acetate type (with 25-65% of cis-epoxyocimene and 15-50% of chrysanthenyl acetate). The distribution of these chemotypes had no relation with the altitude of the samples.
Click to Show/Hide
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor | Part | Location | NP Content | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Chemotype (cis-epoxyocimene type)
|
Leaves | Spain |
NP Content: <0.03 %
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Chemotype (cis-epoxyocimene + chrysanthenyl acetate type)
|
Leaves | Spain |
NP Content: 0.05 %
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Species Name: Artemisia ferganensis | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor Name: Developmental Stage Variation | [3] | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Experiment Detail |
The leaves of aerial parts were collected in Heshuo county of Xinjiang province in China in July 2003 (a vegetative stage), June 2003 (a budding stage); and August 2003 (a flowering stage), respectively.
Click to Show/Hide
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor Function |
Only 23 constituents were present at the budding stage, while 24 and 26 at the flowering and vegetative stages, respectively. p-Cymene and gamma-terpinene were not detected at the vegetative stage of the plant. During the budding stage, butyric, beta-caryophyllene, geranyl acetate and cis-jasmone could not be detected. Benzaldehyde was observed only at the vegetative stage. Variations were also observed in quantity. In all cases the analyzed oils were characterized by the high concentration of alpha-thujone, ranging in amount from 37.0% at the vegetative stage to 54.8% at the budding stage. The concentration of alpha-thujone at the flowering stage (49.0%) was lower than the budding stage, but higher than the vegetative stage. The concentration of cis-chrysanthenyl acetate varied between 23.5% and 7.2%, respectively, at the vegetative and budding stages. At the vegetative stage the concentration of 1,8-cineole was observed to be the lowest. It was highest at the budding stage, representing 10.4%, then decreased gradually to 8.8% at the flowering stage. The concentration of beta-thujone was relatively low at the vegetative stage, representing 8.6%, and then increased to 10.5% at the budding stage. When flowers appeared it was found to vary a little. Finally, the concentration of sabinyl acetate ranged from 10.2% (vegetative stage) to 6.5% (flowering stage).
Click to Show/Hide
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor | Part | Location | NP Content | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Vegetative stage
|
Leaves | Heshuo, Xinjiang, China |
NP Content: 0.4 %
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Species Name: Artemisia verlotiorum | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor Name: Developmental Stage Variation | [4] | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Experiment Detail |
Plant material of A. verlotiorum was harvested near Marseille (France) in May (before blooming) and November (full flowering) 2000.
Click to Show/Hide
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor Function |
For the oil from the vegetative plants, 50 compounds, representing 99.8% of the oil were characterized. Fifty-nine compounds, representing 99.6% of the oil were identified in the oil from flowering plants. In both cases, the constituents were mainly oxygenated monoterpenes (74% and 88%). The composition of each oil showed only a few differences, as the main components were alpha-thujone (55% and 44%), 1,8-cineole (5% and 15%), beta-caryophyllene (13% and 7%) and beta-thujone (5% and 11%), in the oils of the vegetative plant and flowering plant, respectively. The proportions of the oxygenated compounds seemed to increase during flowering.
Click to Show/Hide
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor | Part | Location | NP Content | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Aerial part: full flowering stage
|
Aerial parts | Marseille, France |
NP Content: <0.1 %
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Species Name: phaseolorum sp. PR4 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor Name: PDB medium | [5] | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Experiment Detail |
PR4 was isolated as an endophyte from the rhizome of Picrorhiza kurroa. Picrorhiza kurroa Royle ex. Benth (Plantaginaceae) is a perennial herb endemic to the north western alpine Himalayas. The endophyte PR4 was grown on PDA and in PDB at 26 ℃ for 15 days with constant shaking at 200 rpm in the latter case.
Click to Show/Hide
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Mechanism |
The two candidate NR-PKSs (PKS_3671 and PKS_4063) show differences in their domain organizations. PKS_3671 possesses two ACP-domains. Apart from that, only PKS_3671 contains a SAT-domain . These domains provide the first building block in the polyketide assembly, which usually is different from the extender unit malonyl-CoA (also known as the 'starter unit effect'). The ACA-synthesis however is believed to involve merely malonyl-CoA molecules. Even though the ACA-producing PKSs MdpG, ACAS, EncA, AptA and ClaG contain SAT-domains, an amino acid sequence alignment of these domains revealed that they all lack the active-site cysteine in the GXCXG motif and therefore most likely have no acyl transferase activity. Instead, all malonate building blocks are assumed to be loaded by the MAT. Under this aspect, the SAT-domain of PKS_3671 (that includes the correct GXCXG motif) likely incorporates a starter unit different from malonyl-CoA indicating that this enzyme is not involved in the biosynthesis of ACA. Therefore, the ACA-synthesizing PKS in C. asteris would rather be PKS_4063 that misses the SAT-domain .In the monodictyphenone and cladofulvin pathways, the cluster-encoded gene products MdpH and ClaH are crucial enzymes pushing the biosynthesis towards emodin. These EthD-domain-containing enzymes are suggested to catalyze the decarboxylation of ACA (3) into atrochrysone (4). Surprisingly, no such EthD-domain is encoded in the whole C. asteris genome. On the other hand, four genes directly attached to the putative ACA-synthase-coding gene pks_4063 show high similarity to genes of non-investigated PKS clusters in other fungi , which indicates an involvement in tailoring reactions of the respective polyketide pathways. According to InterProScan and BLASTp analyses, the genes sky_4060-62 encode a dehydratase and two dehydrogenases potentially catalyzing the multistep conversion of ACA (3) into emodin (1). Gene sky_4059 codes for a monooxygenase that putatively can connect two emodin molecules to the final product skyrin (2) in the style of the monooxygenase ClaM involved in the dimerization of the bisanthraquinone cladofulvin. Thus, the presence of these genes in the gene cluster gives further support to the hypothesis that PKS_4063 is the ACA-synthase in C. asteris. Mutational studies will be done in order to confirm these assumptions after a gene transfer system for this strain has been developed.
Click to Show/Hide
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor | Part | Location | NP Content | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
PDB medium (26℃ + 4 days)
|
Rhizomes | Himalayas |
NP Content: 0.15 % Relative area
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Species Name: Fritillaria imperialis | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor Name: Cultivar Comparison | [6] | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Experiment Detail |
Plants of the F. imperialis cultivars Premier (very strong foxy odor) and Lutea (strong foxy odor), the F. imperialis subspecies Inodora (no odor), a cross between F. imperialis Lutea × Inodora (F1 generation, faint foxy odor) were grown from bulbs during the spring and early summer in clay soil near Midlum (Province of Friesland, The Netherlands). Bulbs, newly grown from these plants, were harvested in mid-June and stored, after removal of soil, at ambient temperature until analysis, which occurred in October and November.
Click to Show/Hide
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor Function |
GC-O revealed that the foxy odor was caused by a single component, identified as 3-methyl-2-butene-1-thiol on the basis of smell in GC-O analyses (two GC columns), mass spectra, and retention times. The abundance of 3-methyl-2-butene-1-thiol is consistent with the intensity of foxy Fritillaria odor in the F. imperialis cultivars: Premier > Lutea >> Lutea × Inodora, where the latter did not show a detectable peak in GC-MS.
Click to Show/Hide
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor | Part | Location | NP Content | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Fritillaria imperialis cv. Inodora (no odor)
|
Flowers | Netherlends |
NP Content: 1.2 %
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Fritillaria imperialis cv. Lutea × Inodora (faint foxy odor)
|
Flowers | Netherlends |
NP Content: 2.5 %
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Fritillaria imperialis cv. Lutea (strong foxy odor)
|
Flowers | Netherlends |
NP Content: 1.2 %
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Fritillaria imperialis cv. Premier (very strong foxy odor)
|
Flowers | Netherlends |
NP Content: 1.8 %
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Species Name: Melaleuca quinquenervia | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor Name: Chemotype Comparison | [7] | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Experiment Detail |
Plant material: Leaves of M. quinquenervia were collected from January to October during the ten year period (1992-2001) on mainly East part of Madagascar island and in particular on Toamasina, Mananjary, Manakara, Farafangana, Vangaindrano, Moramanga, and Ambatondrazaka locations. Olfactory chemotype selection: During the years 1992-1995, since the price for (E)-nerolidol and viridiflorol chemotypes was very strong, leaf collection was done on trees chosen after olfactory selection by a chief harvester who compared the olfactory threshold of crumple leaves with our clearly identified oil samples. After 1996, collection was done at random and no particular attention in leaf harvesting was taken.
Click to Show/Hide
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor Function |
Niaouli essential oils from Madagascar were classified into three chemotypes using Principal Component Analysis (PCA): a cineole chemotype (49-62%), a viridiflorol chemotype (21-36%) and an (E)-nerolidol chemotype (56-95%). The 1,8-cineole chemotype is widespread, representing 92% of the samples investigated if the leaf collection is done at random.
Click to Show/Hide
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor | Part | Location | NP Content | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Chemotype (Cineole type)
|
Leaves | Madagascar |
NP Content: 0.19 %
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Chemotype (Viridiflorol type)
|
Leaves | Madagascar |
NP Content: 1.16 %
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Chemotype (E-nerolidol type)
|
Leaves | Madagascar |
NP Content: 0.59 %
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Species Name: Melaleuca quinquenervia (Cav.) S.T. Blake | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor Name: Chemotype Comparison | [8] | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Experiment Detail |
One hundred grams of mature leaves were collected from 2 to 10 widely spaced trees per site and sent to Sydney for analysis as soon as possible after collection. Samples usually arrived in the laboratory within 48 h of collection. The majority of the sampling was done between December 1998 and October 1999. Seasonal trends in oil yields and composition are confounded in the data on geographic variation, but these were considered minor in the context of this study.
Click to Show/Hide
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor Function |
Chemotype 1 is comprised of E-nerolidol (74-95%) and linalool (14-30%) and is found from Sydney, north along the east coast of Australia to Selection Flat, New South Wales, with an isolated occurrence near Maryborough, Queensland. Two divisions occur in this chemotype which are based on the presence or absence of significant proportions of linalool (14-40%). Chemotype 2 contains 1,8-cineole (10-75%), viridiflorol (13-66%), alpha-terpineol (0.5-14%) and beta-caryophyllene (0.5-28%) in varying proportions and order of dominance in the oils. It is found throughout the distribution of the species, from Sydney to Papua New Guinea and New Caledonia. Within chemotype 2 there appears to be a continuous spread of oil composition without formation of any further discrete divisions as in chemotype 1. Analyses have shown that M. quinquenervia trees that occur at latitudes south of 25d S have high oil yields (1-3% w/w%, fresh leaves) and comprise chemotypes 1 and 2. North of 25d S, however, chemotype 1 does not occur and oil yields amongst the Australian populations are uniformly low (0.1-0.2%).
Click to Show/Hide
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor | Part | Location | NP Content | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Chemotype (E-nerolidol in large concentration type)
|
Leaves | Australia and Papua New Guinea |
NP Content: 0.7 %
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Chemotype (either 1,8-cineole or viridiflorol in highest proportion type)
|
Leaves | Australia and Papua New Guinea |
NP Content: 0.2 %
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Species Name: Salvia aucheri | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor Name: Variety Comparison | [9] | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Experiment Detail |
S. aucheri var. aucheri was collected in Karaman: Ermenek to Mutt Road on July 19,1995; Salvia aucheri var. canescens was collected in Karaman: Ermenek, Tekecati Valley on July 19,1995.
Click to Show/Hide
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor Function |
Eighty components were characterized in the Salvia aucheri var. aucheri oil, with camphor (21.1%), 1, 8-cineole (20.3%), borneol (7.8%), spathulenol (6.3%) and camphene (5.3%) as major constituents. 1, 8-Cineole (25.2%), camphor (17.9%), borneol (10.6%), alpha-pinene (5.4%) and camphene (5.3%) were identified as major constituents among the 88 components characterized in the oil of Salvia aucheri var. canescens.
Click to Show/Hide
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor | Part | Location | NP Content | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Salvia aucheri var. canescens
|
Aerial parts | Karaman, Turkey |
NP Content: <0.01 %
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Species Name: Salvia euphratica | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor Name: Variety Comparison | [10] | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Experiment Detail |
Aerial parts of both varieties(Salvia euphratica Montbret et Aucher ex Benth. var. euphratica and Salvia euphratica Montbret et Aucher ex Benth. var. leiocalycina) were collected in Malatya, Turkey in June 1999.
Click to Show/Hide
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor Function |
Ninety-five compounds in var. euphratica and 94 compounds in var. leiocalycina were characterized representing 93% and 95% of the total components detected, respectively, with 1,8-cineole (13.8% and 15.2%) and myrtenyl acetate (15.9% and 13.9%) as main constituents.
Click to Show/Hide
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor | Part | Location | NP Content | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Salvia euphratica var. euphratica
|
Flowering aerial parts | Turkey |
NP Content: 0.1 %
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Salvia euphratica var. leiocalycina
|
Flowering aerial parts | Turkey |
NP Content: 0.1 %
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Species Name: Stachys pilifera | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor Name: Locality Variation | [11] | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Experiment Detail |
Plant material and isolation procedure: Aerial parts of the plant were collected from two regions, from Kazeroon in southern Iran and Shahr-e-kord in western Iran at the time of flowering in June 2002.
Click to Show/Hide
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor Function |
The main components of the oil of S. pilifera collected from Kazeroon, in southern Iran, were spathulenol (15.8%), cis-chrysanthenol (15.3%), beta-caryophyllene (8.4%) and cis-chrysanthenyl acetate (6.9%), while for the plant collected from Shahr-e-kord, in western Iran, they were cis-chrysanthenyl acetate (21.8%), linalool (18.9%), terpinen-4-ol (11.9%) and cis-chrysanthenol (9.2%).
Click to Show/Hide
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor | Part | Location | NP Content | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Locality: Kazeroon, southern Iran
|
Aerial parts | Iran |
NP Content: <0.05 %
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Locality: Shahr-e-kord, western Iran
|
Aerial parts | Iran |
NP Content: <0.05 %
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Species Name: Teucrium chamaedrys | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor Name: Locality Variation | [12] | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Experiment Detail |
The aerial parts of T. chamaedrys were collected at the flowering stage in June 2004 near Corti, Corsica, France and near Oristano, Sardinia, Italy
Click to Show/Hide
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor Function |
The Corsican and Sardinian oils of T. chamaedrys investigated in this study were qualitatively similar but they differed by the amount of their major components. The major components were beta-caryophyllene (29.0% and 27.4%, respectively) and germacrene D (19.4% and 13.5%, respectively), followed by alpha-humulene (6.8%) and delta-cadinene (5.4%) in the Corsican oil and by caryophyllene oxide (12.3%) and alpha-humulene (6.5%) in the Sardinian oil. These quantitative differences are also noticeable on the amounts of the different class compounds. Especially, the monoterpene hydrocarbons amounted for 10.3% and 4.1% in Sardinian and Corsican oils respectively and the oxygenated sesquiterpenes amounted for 18.9% and only 7.4% in both oils, respectively. Both oils were qualitatively rather similar in comparison with those reported in the literature from various geographic regions. However, among the 87 components identified in this study, 47 minor components (< 0.6%) reported were identified for the first time in T. chamaedrys oil. This study confirms the quantitative variability of the major components according to the plant origin.
Click to Show/Hide
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor | Part | Location | NP Content | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Locality: Corti, Corsica, France
|
Aerial parts | France |
NP Content: <0.05 %
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Locality: Oristano, Sardinia, Italy
|
Aerial parts | Italy |
NP Content: <0.05 %
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Species Name: Thymus pseudopulegioides | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor Name: Locality Variation | [13] | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Experiment Detail |
Plant materials were collected from the following localities in north western Turkey. A = Trabzon: Caykara, Soganli dag on July 28, 1994; B = Bayburt: Caykara, Mohakambo yaylasi on July 25, 1994; C = Trabzon: Koprubasi, Vizara yaylasi on July 20, 1994.
Click to Show/Hide
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor Function |
One hundred and four compounds were identified representing 97.5-99.5% of the total components detected in thymol/carvacrol (50.14/10.67%), thymol/linalool (23.14/20.24%) and linalool/alpha-terpinyl acetate/geraniol (21.55/16.70/11.17%) rich oils.
Click to Show/Hide
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor | Part | Location | NP Content | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Locality: Soganli dag, Caykara, Trabzon, Eskisehir, Turkey
|
Aerial parts | Eskisehir, Turkey |
NP Content: <0.1 %
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Species Name: Vitis vinifera cv. Pinot noir | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor Name: Drought Stress Treatment | [14] | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Experiment Detail |
3-year old single shoot V. vinifera plants (cultivar Pinot noir 18 Gm grafted on Kober 5BB, 51 plants) potted in 3L pots in a sandy loam soil were used. All plants were well watered (200 mL per day) at the beginning of the experiment (04.06.2010; DAY 0; 5 plants) and water was supplied to all control plants once every day (250 mL per day), whereas water supply of stressed plants was stopped. Physiological measurements and sampling of leaves took place on 07.06.2010 (DAY 3; 5 control, 5 stressed plants), 10.06.2010 (DAY 6; 5 control, 5 stressed plants) and 12.06.2010 (DAY 8; 5 control, 10 stressed plants). Due to very hot weather conditions in June 2010 the experiment was stopped after 8 days and 12 available control plants were used to restart the drought treatment with 6 control and 6 stressed plants on 11.06.2010 and all plants were measured on 15.06.2010 (DAY 5). The mean leaf temperatures at midday were: 25 ℃ (04.06.2010; DAY 0), 31.9 ℃ (07.06.2010; DAY 3), 30.8 ℃ (15.06.2010; DAY 5), 35.8 ℃ (10.06.2010; DAY 6) and 35.7 ℃ (12.06.2010; DAY 8). The mean PAR radiation per day (measured from 6:00 am till 7:00 pm) was 144.1 µmol m-2 s-1. Each plant was used only once for physiological measurements and sampling of leaves.On every day of the experiment (day 0, 3, 5, 6, 8) the pot weight and the volumetric soil moisture content (ThetaProbe ML2x and handheld data logger Moisture Meter HH2, Delta-T Devices, Cambridge, United Kingdom) was recorded. The water potential (PWSC Model 3000, Soilmoisture Equipment Corporation, Santa Barbara, USA) was determined for the 6th leaf (representing the insertion level of the shoot from the basis) of every plant and measurement day. Chlorophyll fluorescence and gas exchange parameters of light adapted leaves were determined with the 4th and 5th leaf, whereas dark adaptation was performed only with the 5th leaf. Immediately after these non-invasive measurements, the 5th leaf was harvested, frozen in liquid nitrogen and further used for the measurement of polyphenols, selected primary metabolites and volatiles (VOCs).
Click to Show/Hide
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor Function |
The content of different groups of primary and secondary metabolites is significantly influenced by severe drought stress in grapevine leaves. The content of the majority of the metabolites (around 60% of primary metabolites, around 85% of polyphenols and about 40% of the detected and identified VOCs) increased upon drought stress treatment. Among these especially the primary metabolites citric acid and glyceric acid were strongly influenced by the short as well as the prolonged drought stress treatment, whereas all polyphenols were only induced upon the prolonged drought stress treatment.
Click to Show/Hide
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor | Part | Location | NP Content | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Normal condition
|
Leaves | Vienna, Austria |
NP Content: 27437.7 ± 7574.5 peak areas
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Dry 3-5 days
|
Leaves | Vienna, Austria |
NP Content: 42386.9 ± 11150.1 peak areas
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Dry 6-8 days
|
Leaves | Vienna, Austria |
NP Content: 68084.3 ± 35606.0 peak areas
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||