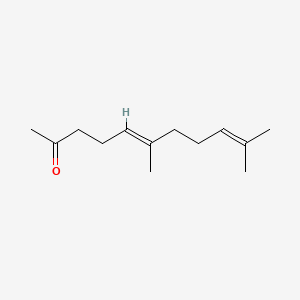
| The Content Variation of Natural Product Induced by Different Factor(s) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Species Name: Arabidopsis thaliana | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor Name: T-DNA Knock-Out Treatment | [1] | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Experiment Detail |
In vitro cultivation of Arabidopsis wildtype and mutant plants: Seeds were sterilized according to standard lab routines (EtOH, NaOCl/NaOH) prior to aseptical (in vitro) cultivation in 500 ml screw cap jars on MS medium (4.3 g/l; 50 ml/jar) containing Bacto- and Phytoagar (1:2; 6 g/l) and 30 g/l sucrose. Ten seeds were pipetted into each jar and plants grown for 6 weeks until flowering at a temperature of 20 ℃ under a 16/8 h day/ night regime using fluorescent tubes (Osram Lumilux Plus Eco 36 W). Both Arabidopsis thaliana wildtype plants of ecotype Columbia-0 (Col) and 4 Col-derived T-DNA knock-out mutants (homozygous lines) showing deficiencies in the GLS biosynthesis pathway were used in this study (five parallels for wildtype and mutants): TGG1 (Atg526000; Salk_130469), TGG2 (At5g25980; Salk_038730), Cyp83A1 (At4g13770) and Cyp83B1 (At4g31500; Salk_028573). Greenhouse-cultivation of Arabidopsis ecotypes: The following Arabidopsis ecotypes were used in the study: Columbia (Col), Cape Verde Islands (Cvi), Landsberg erecta (Ler) and Wassilewskija (Ws). Single plants were greenhouse-cultivated on fertilized soil (P-Jord; Emmaljunga Torvmull AB) in plug trays (9 × 6 cells) at a temperature of 20 ℃ (three parallels for each ecotype). Due to the 6-weeks growth period (November/December 2003), the plants were cultivated under a 16/8 h day/night regime using metal halide lamps (Osram HQI-T 400 W) placed 130 cm above the trays. Depending on the ecotypical plant development, whole plants were sampled after 3-4 weeks right before bolting for in vivo studies, while investigations of single plant organs (leaf, stem, inflorescence) were carried out after 5-6 weeks of cultivation.
Click to Show/Hide
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor Function |
Metabolites from methionine, leucine and phenylalanine-derived glucosinolates were most abundant (4-methylthiobutyl, 4-methylpentyl, 2-phenylethyl). In addition, 24 monoterpenes, 26 sesquiterpenes and 12 aromatic structures, predominantly observed in inflorescenses, are described. Excluding the vast group of straight chain aliphatic structures, a total of 102 volatile compounds were detected, of which 59 are reported in Arabidopsis thaliana for the first time, thus emphasizing the sensitivity and applicability of solid-phase microextraction for volatile profiling of plant secondary metabolites.
Click to Show/Hide
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor | Part | Location | NP Content | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Inflorescences: A. thaliana wildtype detected in vivo
|
Flowers | Norway |
NP Content: 9.03 %
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Leaves: A. thaliana wildtype detected in vivo
|
Leaves | Norway |
NP Content: 13.59 %
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Stem: A. thaliana wildtype detected in vivo
|
Stems | Norway |
NP Content: 4.85 %
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Arabidopsis thaliana wildtype
|
Whole plant | Norway |
NP Content: 1.26 %
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
A. thaliana T-DNA knock-out mutant Cyp83A1: (At4g13770)
|
Whole plant | Norway |
NP Content: 0.85 %
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
A. thaliana T-DNA knock-out mutant Cyp83B: (At4g31500; Salk_028573)
|
Whole plant | Norway |
NP Content: 0.59 %
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
A. thaliana T-DNA knock-out mutant TGG1: (Atg526000; Salk_130469)
|
Whole plant | Norway |
NP Content: 0.66 %
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
A. thaliana T-DNA knock-out mutant TGG2: (At5g25980; Salk_038730)
|
Whole plant | Norway |
NP Content: 0.46 %
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Species Name: Callistemon salignus | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor Name: Locality Variation | [2] | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Experiment Detail |
Fresh leaf samples of C. salignus were collected on the campus of University of Zululand, KwaDlangezwa and Empangeni (Both in KwaZulu-Natal Province) , South Africa.
Click to Show/Hide
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor Function |
1,8-Cineole (63.4%), alpha-pinene (17.8%) and E-(beta)-ocimene (6.7%) were the major constituents identified in the KwaDlangezwa sample (Sample A). The Empangeni sample (Sample B) contained only 1,8-cineole (85.4%) and alpha-pinene (6.2%) as the main compounds present in the oil.
Click to Show/Hide
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor | Part | Location | NP Content | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Locality: KwaDlangezwa, KwaZulu-Natal Province, South Africa
|
Leaves | South Africa |
NP Content: 0.2 %
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Species Name: Dalbergia odorifera | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor Name: NaCl Treatment; Acetic Acid Treatment; H2O2 Treatment | [3] | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Experiment Detail |
A 5-year-old D. odorifera plantation located in Xiashi Arboretum (22° 60′ N, 106° 53′ E), Pingxiang City, Guangxi Zhuang Autonomous Region (GZAR) was selected for this study. Three chemicals used were: 0.1 mol /L hydrogen peroxide (H2O2), 1.0 mol/L (pH=2.4) acetic acid (CH3COOH) and 1.0 mol/L sodium chloride (NaCl). Distilled water was injected as a control. Sixty trees of similar size, all without heartwood were selected for a single-tree plot experiment with 15 replicates for each treatment. A tiny drill was used to detect which trees have or don't have heartwood. In May 2017, an injection hole of 1 cm diameter and 5 cm deep was drilled at a downward angle of 45 ℃ at 1.3 m from the ground on each tree. An amount of 50 ml solution was injected into each tree over 8 h period with a 5 ml syringe, and the hole was sealed with a cork after injection. Five trees per treatment were harvested at one, three and six months after injection treatment. At the same time, three trees with heartwood were selected to collect natural heartwood.
Click to Show/Hide
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor Function |
Hydrogen peroxide-induced Jiang Xiang was closest to natural heartwood, and the essential oil components met the standards for high-quality Jiang Xiang, while the induction effects of acetic acid and sodium chloride were unsatisfactory. Thus, this study indicates that hydrogen peroxide has the potential to induce Jiang Xiang production in Dalbergia odorifera.
Click to Show/Hide
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor | Part | Location | NP Content | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
0.1 mol/L H2O2(Six months after treatment)
|
heartwood | Pingxiang City, Guangxi Zhuang Autonomous Region, China |
NP Content: 0.05 %
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
1.0 mol/L NaCl (Six months after treatment)
|
heartwood | Pingxiang City, Guangxi Zhuang Autonomous Region, China |
NP Content: 0.08 %
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Control (Six months after treatment)
|
heartwood | Pingxiang City, Guangxi Zhuang Autonomous Region, China |
NP Content: 0.01 %
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Natural heartwood
|
heartwood | Pingxiang City, Guangxi Zhuang Autonomous Region, China |
NP Content: 0.01 %
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Species Name: Fragaria vesca | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor Name: Cultivar Comparison | [4] | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Experiment Detail |
Whole leaves and inflorescences of two wild strawberry cultivars ('Rugia' and 'Baron von Solemacher') harvested in 2008 during the agrotechnical experiment performed by Department of Vegetable and Medicinal Plants, University of Life Sciences in Lublin, were used as a material for determinations. Samples were collected before noon at sunny and dry days at the beginning of wild strawberry's flowering stage. Material was dried up to 35 ℃ in shadow and air just after the harvest.
Click to Show/Hide
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor Function |
Depending on a cultivar, air-dry inflorescences from wild strawberry contain from 0.21% ('Baron von Solemacher' cv.) to 0.30% ('Rugia' cv.), whereas leaves contains from 0.46% ('Baron von Solemacher' cv.) to 0.62% ('Rugia' cv.) of essential oils. GC/MS analysis of essential oils achieved from studied materials revealed presence of 70 (including 59 identified) compounds in leaves of 'Rugia' cv. and 58 (including 50 identified) compounds in leaves of 'Baron von Solemacher' cv. Essential oils from inflorescences of 'Rugia' cv. contained 52 (including 47 identified), while 'Baron von Solemacher' cv. contained 54 (including 46 identified) compounds. The chromatographic analyses by GC-MS revealed that myrthenol, nonal, linalool and phthalide dibuthyl dominated in essential oils obtained from leaves, while myrthenol, citronelol, linalool and geraniol - from those of inflorescences. There were qualitative differences between oil components at both studied materials and differentiation between both cultivars, as well.
Click to Show/Hide
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor | Part | Location | NP Content | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Inflorescence: Fragaria vesca cv. Baron von Solemacher
|
Inflorescence | Poland |
NP Content: <0.05 %
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Inflorescence: Fragaria vesca cv. Rugia
|
Inflorescence | Poland |
NP Content: <0.05 %
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Species Name: Tanacetum larvatum | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor Name: Locality Variation | [5] | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Experiment Detail |
Aerial parts of T. larvatum were collected in July and August during a five-year period, starting in 2001, in Montenegro on several locations: Planinica (Sample a), Visitor (Sample b) and Sinjajevina (Sample c).
Click to Show/Hide
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor Function |
Sixty-four components were identified, representing 83.1%, 96.6% and 89.4% of the total oils content in the Planinica [Sample a], Visitor [Sample b] and Sinjajevina [Sample c], respectively. The major constituent in Samples a and b , was oxygenated monoterpene, trans-sabinyl acetate (38.1% and 55.8% respectively). Monoterpene hydrocarbons, beta-pinene (13.5%) and santolinatriene (30.6%), were found to be the dominant components in Sample c. The toxic trans-sabinyl acetate was present only in traces in this sample. trans-Chrysanthenyl acetate, as one of major components in feverfew essential oil, has not been previously identified in the investigated essential oils.
Click to Show/Hide
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor | Part | Location | NP Content | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Locality: Sinjajevina, Montenegro
|
Aerial parts | Montenegro |
NP Content: 0.3 %
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Locality: Visitor, Montenegro
|
Aerial parts | Montenegro |
NP Content: 0.3 %
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Species Name: Vitis vinifera cv. Pinot noir | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor Name: Drought Stress Treatment | [6] | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Experiment Detail |
3-year old single shoot V. vinifera plants (cultivar Pinot noir 18 Gm grafted on Kober 5BB, 51 plants) potted in 3L pots in a sandy loam soil were used. All plants were well watered (200 mL per day) at the beginning of the experiment (04.06.2010; DAY 0; 5 plants) and water was supplied to all control plants once every day (250 mL per day), whereas water supply of stressed plants was stopped. Physiological measurements and sampling of leaves took place on 07.06.2010 (DAY 3; 5 control, 5 stressed plants), 10.06.2010 (DAY 6; 5 control, 5 stressed plants) and 12.06.2010 (DAY 8; 5 control, 10 stressed plants). Due to very hot weather conditions in June 2010 the experiment was stopped after 8 days and 12 available control plants were used to restart the drought treatment with 6 control and 6 stressed plants on 11.06.2010 and all plants were measured on 15.06.2010 (DAY 5). The mean leaf temperatures at midday were: 25 ℃ (04.06.2010; DAY 0), 31.9 ℃ (07.06.2010; DAY 3), 30.8 ℃ (15.06.2010; DAY 5), 35.8 ℃ (10.06.2010; DAY 6) and 35.7 ℃ (12.06.2010; DAY 8). The mean PAR radiation per day (measured from 6:00 am till 7:00 pm) was 144.1 µmol m-2 s-1. Each plant was used only once for physiological measurements and sampling of leaves.On every day of the experiment (day 0, 3, 5, 6, 8) the pot weight and the volumetric soil moisture content (ThetaProbe ML2x and handheld data logger Moisture Meter HH2, Delta-T Devices, Cambridge, United Kingdom) was recorded. The water potential (PWSC Model 3000, Soilmoisture Equipment Corporation, Santa Barbara, USA) was determined for the 6th leaf (representing the insertion level of the shoot from the basis) of every plant and measurement day. Chlorophyll fluorescence and gas exchange parameters of light adapted leaves were determined with the 4th and 5th leaf, whereas dark adaptation was performed only with the 5th leaf. Immediately after these non-invasive measurements, the 5th leaf was harvested, frozen in liquid nitrogen and further used for the measurement of polyphenols, selected primary metabolites and volatiles (VOCs).
Click to Show/Hide
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor Function |
The content of different groups of primary and secondary metabolites is significantly influenced by severe drought stress in grapevine leaves. The content of the majority of the metabolites (around 60% of primary metabolites, around 85% of polyphenols and about 40% of the detected and identified VOCs) increased upon drought stress treatment. Among these especially the primary metabolites citric acid and glyceric acid were strongly influenced by the short as well as the prolonged drought stress treatment, whereas all polyphenols were only induced upon the prolonged drought stress treatment.
Click to Show/Hide
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor | Part | Location | NP Content | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Normal condition
|
Leaves | Vienna, Austria |
NP Content: 53111.2 ± 18274.8 peak areas
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Dry 3-5 days
|
Leaves | Vienna, Austria |
NP Content: 73444.3 ± 23198.9 peak areas
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Dry 6-8 days
|
Leaves | Vienna, Austria |
NP Content: 108890.3 ± 104305.1 peak areas
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||