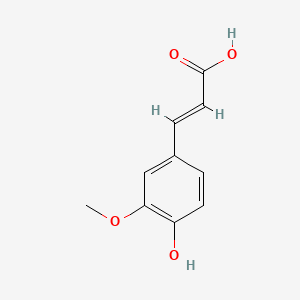
| The Content Variation of Natural Product Induced by Different Factor(s) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Species Name: Amaranthus tricolor genotype VA13 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor Name: NaCl Treatment | [1] | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Experiment Detail |
On the basis of previous studies, an antioxidant enriched high yield potential genotype (Accession VA13) was selected for this investigation. This genotype was grown in pots of a rain shelter open field of Bangabandhu Sheikh Mujibur Rahman Agricultural University, Bangladesh (AEZ-28, 24° 23′ north latitude, 90° 08′ east longitude, 8.4 m.s.l.). The seeds were sown in plastic pots (15 cm in height and 40 cm length and 30 cm width) in a randomized complete block design (RCBD) with three replications. N: P2O5:K2O were applied @92:48:60 kg/ha as a split dose. First, in pot soil, @46:48:60 kg ha 1 N: P2O5:K2O and second, at 7 days after sowing (DAS) @46:0:0 kg/ha N: P2O5:K2O. The genotype was grouped into three sets and subjected to four salinity stress treatments that are, 100 mM NaCl, 50 mM NaCl, 25 mM NaCl, and control or no saline water (NS). Pots were well irrigated with fresh water every day up to 10 days after sowing (DAS) of seeds for proper establishment and vigorous growth of seedlings. Imposition of salinity stress treatment was started at 11 DAS and continued up to 40 DAS (edible stage). Saline water (100 mM NaCl, 50 mM NaCl and 25 mM NaCl) and fresh water were applied to respective pots once a day. At 40 DAS the leaves of Amaranthus tricolor were harvested. All the parameters were measured in six samples.
Click to Show/Hide
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor Function |
At Moderate salinity stress (MSS) and Severe salinity stress (SSS) conditions, leaf color parameters and pigments, vitamins, phenolic acids, flavonoids and antioxidant capacity of A. tricolor leaves were very high compared to control condition. Hence, salt-stressed A. tricolor leaves had a good source of natural antioxidants compared to plant grown in normal cultivation practices.
Click to Show/Hide
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor | Part | Location | NP Content | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
No saline water (Control)
|
Leaves | Bangabandhu |
NP Content: 1.20 ± 0.02 µg/g fresh weight
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
25 mM NaCl (Low salinity stress)
|
Leaves | Bangabandhu |
NP Content: 1.16 ± 0.02 µg/g fresh weight
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
50 mM NaCl (Moderate salinity stress)
|
Leaves | Bangabandhu |
NP Content: 2.05 ± 0.04 µg/g fresh weight
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
100 mM NaCl (Severe salinity stress)
|
Leaves | Bangabandhu |
NP Content: 3.19 ± 0.05 µg/g fresh weight
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Species Name: Clausena lansium | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor Name: Developmental Stage Variation | [2] | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Experiment Detail |
Clausena lansium (Lour.) Skeels leaves of four developmental stages, namely, (i) leaf buds, (ii) young leaves, (iii) mature leaves, and (iv) old leaves, were collected from three 13-year-old trees grown in wampee resources nursery of Institute of Fruit Tree Research, Guangdong Academy of Agricultural Sciences in Guangzhou, China.
Click to Show/Hide
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor Function |
Increase in bound flavonoids, quercetin, and cellular antioxidant activity was observed in bound and free fractions at different stages of leaf development. Predominantly, quercetin and ferulic acid contents were high in free and bound fractions of old leaves. In addition, phenolic components depicted highly significant positive association (p < 0.05) with antioxidant activity.
Click to Show/Hide
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor | Part | Location | NP Content | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Leaf buds
|
Leaves | Guangzhou, Guangdong, China |
NP Content: 1.31 ± 0.01 mg/100g
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Young leaves
|
Leaves | Guangzhou, Guangdong, China |
NP Content: 1.49 ± 0.01 mg/100g
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Young leaves
|
Leaves | Guangzhou, Guangdong, China |
NP Content: 1.29 ± 0.03 mg/100g
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Mature leaves
|
Leaves | Guangzhou, Guangdong, China |
NP Content: 1.66 ± 0.03 mg/100g
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Mature leaves
|
Leaves | Guangzhou, Guangdong, China |
NP Content: 1.28 ± 0.02 mg/100g
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Old leaves
|
Leaves | Guangzhou, Guangdong, China |
NP Content: 1.75 ± 0.01 mg/100g
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Old leaves
|
Leaves | Guangzhou, Guangdong, China |
NP Content: 1.82 ± 0.19 mg/100g
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Species Name: Cucumis sativus L. cv. Jincun 2 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor Name: Low Temperature Treatment; AMF Inoculation | [3] | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Experiment Detail |
AMF inocula [Funneliformis mosseae (T.H. Nicolson & Gerd.) C. Walker & A. Schubler] consisting of spores, soil, hyphae and infected clove (Trifolium repens L.) root fragment from a stock culture of F. mosseae.The inoculated dosage was 10 g of inocula per pot containing approximately 720 spores calculated by microscopy before experiment. There was 2100 infective propagules/g in the inoculum as determined by MPN assay . Seeds were surface sterilized by immersion in 70% ethanol for 5 min, rinsed four times with distilled water, and placed on wet filter paper in Petri dishes at 28 ℃ for germination. After 3 days, the germinate seeds were transplanted into 13 cm × 13 cm plastic pots containing 0.88 kg organized soil substrate (organic manure, soil and decomposed straw = 1:2:1). Half of the pots (AM plants) were inoculated with 10 g of F. mosseae per pot. Non-AM plants received the same weight of autoclaved inocula. The inocula were placed adjacent to each seeding root. The organic substrate was collected from the greenhouse of Institute of Vegetables and Flowers, CAAS and sterilized for 4 h at 160 ℃ , with the chemical properties as follows: pH 7.26, 11.1% organic matter, 150 mg/kg available phosphorus, 451 mg/kg available nitrogen and 518 mg/kg available potassium. The experimental pots were placed in solar greenhouse at an average temperature of 28 ℃ /20 ℃ (day/night) with photon flux density of 600 µmol m-2 s-1 and 85% relative humidity.The seedlings uniformed in size were transferred to a growth chamber subjected to different temperature conditions at 20 days after inoculation. The experimental design consisted of four treatments crossing two mycorrhizal inoculations levels (non-AMF and F. mosseae) with two temperature levels with photon flux density of 100 µmol m-2 s-1 (25 ℃ /15 ℃ , 15 ℃ /10 ℃ , day/night). (1) Normal temperature (NT): 10 g of sterilized inoculua, 25 ℃ /15 ℃ (day/night); (2) AMF-inoculation (AMF): 10 g of inocula, 25 ℃ /15 ℃ (day/night); (3) low temperature (LT): 10 g of sterilized inocula, 15 ℃ /10 ℃ (day/night); (4) AMF-inoculation under low temperature (AMF + LT): 10 g of inocula, 15 ℃ /10 ℃ (day/night).The experimental design was a completely randomized block design and thirty plants were arranged in each replication. On 45 days after inoculation, phenolic compounds contents, enzymes and gene transcription were determined.
Click to Show/Hide
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor Function |
AMF-inoculated cucumber seedlings had significant higher fresh weight and dry weight than non-AMF inoculated control plants under both normal (25/15 ℃ ) and low temperature (15/10 ℃ ) treatment. Under chilling stress, AMF inoculation significantly improved the content of related secondary metabolites including phenols, flavonoids, lignin, DPPH activity and phenolic compounds compared with the non-AMF control. Furthermore, large increments were observed in a number of enzymatic activities related to secondary metabolism and antioxidant system in AMF-inoculated seedlings under low temperature, such as glucose-6-phosphate dehydrogenase (G6PDH), shikimate dehydrogenase (SKDH), phenylalanine ammonia-lyase (PAL), cinnamyl alcohol dehydrogenase (CAD), polyphenol oxidase (PPO), guaiacol peroxidase (G-POD), caffeic acid peroxidase (CA-POD) and chlorogenic acid peroxidase (CGA-POD). As well, the expression of stress-related marker genes was enhanced in AMF-inoculated seedlings in comparison with the non-AMF control. Furthermore, AMF symbiosis decreased hydrogen peroxide (H2O2) content under low temperature.
Click to Show/Hide
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Mechanism |
Consistent with the changes of enzyme activities, the relative transcriptional level of genes related to secondary metabolism increased significantly in response to AM fungi inoculation in cucumber roots . Moreover, under chilling stress, the expression levels of WRKY30, PR-1, C4H, CCOMT, CAD, G6PDH, PAL, LPO, and POD genes in the AMF-inoculated seedling were 2.46, 2.90, 1.84, 2.47, 1.96, 2.52, 1.89 and 1.93 folds respectively compared with those under low temperature alone.
Click to Show/Hide
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor | Part | Location | NP Content | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Normal temperature: 10 g of sterilized inoculua (Funneliformis mosseae), 25/15 ℃ (day/night)
|
Leaves | NA |
NP Content: 0.86 µg/g fresh weight
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Normal temperature: 10 g of sterilized inoculua (Funneliformis mosseae), 25/15 ℃ (day/night)
|
Leaves | NA |
NP Content: 10.25 µg/g fresh weight
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
AMF-inoculation: 10 g of inocula (F. mosseae), 25/15 ℃ (day/night)
|
Leaves | NA |
NP Content: 1.85 µg/g fresh weight
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
AMF-inoculation: 10 g of inocula (F. mosseae), 25/15 ℃ (day/night)
|
Leaves | NA |
NP Content: 26.56 µg/g fresh weight
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Low temperature: 10 g of sterilized inocula (F. mosseae), 15/10 ℃ (day/night)
|
Leaves | NA |
NP Content: 1.34 µg/g fresh weight
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Low temperature: 10 g of sterilized inocula (F. mosseae), 15/10 ℃ (day/night)
|
Leaves | NA |
NP Content: 20.14 µg/g fresh weight
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
AMF-inoculation under low temperature: 10 g of inocula (F. mosseae), 15/10 ℃ (day/night)
|
Leaves | NA |
NP Content: 2.18 µg/g fresh weight
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
AMF-inoculation under low temperature: 10 g of inocula (F. mosseae), 15/10 ℃ (day/night)
|
Leaves | NA |
NP Content: 34.56 µg/g fresh weight
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Species Name: Fragaria × ananassa Duch. | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor Name: Nitrogen Treatment; AMF Inoculation | [4] | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Experiment Detail |
The experiment was conducted in a 'shade'-type greenhouse with 30% shade at the Instituto de Investigaciones Agropecuarias y Forestales (IIAF), Universidad Michoacana de San Nicolas de Hidalgo (UMSNH), Morelia, Michoacan, Mexico. Maximum and minimum temperatures in the greenhouse varied between 28 and 32 ℃ and between 8 and 18 ℃ respectively. Plants of the strawberry cultivar 'Aromas' were used that had previously been grown in a sterilised (95 ℃ water/steam, 40 min) substrate of coconut fibre/perlite (1:3 v/v) under greenhouse conditions. Before the experiment was established, the absence of AMF in the roots was verified by the ink and vinegar technique, modifying the duration of immersion in KOH and ink/vinegar solution (7 and 5 min respectively). Before planting, roots were disinfected by submerging them for 20 s in 20 g/L sodium hypochlorite solution and rinsing them in water. The inoculum was prepared with spores of Glomus intraradices cultivated in liquid medium (3.5 × 106 spores/L, 90% viability; Premier Tech Biotechnologies Company, Quebec, Canada), which was diluted with fitagel (Sigma P-8169, Saint Louis, MO, USA) solution at 50 g/L to obtain a final concentration of about 5 × 104 spores/L. The viability of spores was determined according to the method of An and Hendrix. Eighteen days after setting up the experiment, each plant received 2 mL of inoculum applied directly to the recently formed roots. One month later, after staining, the percentage of root colonisation was determined by the gridline intersect method. The experiment was organised as a full factorial, completely randomised design with two factors: inoculation (two levels: mycorrhizal and non-mycorrhizal plants) and N concentration in the nutrient solution (three levels: 3, 6 and 18 mmol/L). The six treatments were replicated four times, producing 24 experimental units with ten plants each. Every second day, all plants were irrigated up to substrate saturation. Nitrogen was supplied as NO and the cation/anion ratio was kept constant by varying the concentration of SO. When N was below 18 mmol/L, the cation concentrations were maintained as follows: K+, 3; Ca2+, 3.5; Mg2+, 1.5 mmol/L. They were increased in the 18 mmol/L N treatment: K+, 6.5; Ca2+, 7.5; Mg2+, 3.25 mmol/L. In all nutrient solutions the concentration of phosphorus (P) was 0.3 mmol/L. The other nutrients in the solutions were: H3BO3, 20; CuSO4. 5H2O, 0.5; Fe-EDTA (Ethylenediaminetetraacetic acid iron (III) sodium salt), 15; MnSO4.H2O, 12; (NH4)6Mo7O24 . 4H2O, 0.05; ZnSO4 . 7H2O, 3 µmol/L. The pH was adjusted to 5.5 at every application date.
Click to Show/Hide
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor Function |
Mycorrhization did not modify the weight, diameter or length of strawberry fruits but had a negative effect on most colour parameters. Moreover, fruits of mycorrhizal plants had higher K and Cu concentrations and showed greater accumulation of most phenolic compounds.
Click to Show/Hide
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor | Part | Location | NP Content | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Nitrogen concentration (mmol/L): 3
|
Mature fruits | Morelia, Michoacan, Mexico |
NP Content: 0.011 g/kg dry matter
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Nitrogen concentration (mmol/L): 6
|
Mature fruits | Morelia, Michoacan, Mexico |
NP Content: 0.01 g/kg dry matter
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Nitrogen concentration (mmol/L): 18
|
Mature fruits | Morelia, Michoacan, Mexico |
NP Content: 0.016 g/kg dry matter
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Glomus intraradices inoculation
|
Mature fruits | Morelia, Michoacan, Mexico |
NP Content: 0.01 g/kg dry matter
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Non-AMF inoculation
|
Mature fruits | Morelia, Michoacan, Mexico |
NP Content: 0.014 g/kg dry matter
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Nitrogen concentration (mmol/L): 3 + G. intraradices inoculation
|
Mature fruits | Morelia, Michoacan, Mexico |
NP Content: 0.011 g/kg dry matter
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Nitrogen concentration (mmol/L): 3 + Non-AMF inoculation
|
Mature fruits | Morelia, Michoacan, Mexico |
NP Content: 0.011 g/kg dry matter
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Nitrogen concentration (mmol/L): 6 + G. intraradices inoculation
|
Mature fruits | Morelia, Michoacan, Mexico |
NP Content: 0.006 g/kg dry matter
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Nitrogen concentration (mmol/L): 6 + Non-AMF inoculation
|
Mature fruits | Morelia, Michoacan, Mexico |
NP Content: 0.014 g/kg dry matter
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Nitrogen concentration (mmol/L): 18 + G. intraradices inoculation
|
Mature fruits | Morelia, Michoacan, Mexico |
NP Content: 0.013 g/kg dry matter
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Nitrogen concentration (mmol/L): 18 + Non-AMF inoculation
|
Mature fruits | Morelia, Michoacan, Mexico |
NP Content: 0.018 g/kg dry matter
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Species Name: Lentil var. Tina | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor Name: H2O2 Treatment; Mannitol Treatment; NaCl Treatment; High Temperature Treatment; Low Temperature Treatment | [5] | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Experiment Detail |
Seeds were sterilized in 1% (v/v) sodium hypochloride (Sigma-Aldrich, USA) for 10 min, then drained and washed with distilled water until they reached neutral pH. They were placed in distilled water and soaked for 6 h at 25 ℃ . Seeds were dark germinated for 8 days in a growth chamber (SANYO MLR-350H) on Petri dishes (125 mm) lined with absorbent paper. Seedlings were watered with 5 ml of Milli-Q water daily. Sprout (8-day-old) samples were gently collected, weighed (fresh mass), rapidly frozen and kept in polyethylene bags at -20 ℃ . For each treatment, three replicates were performed.Elicitation conditions were selected in previous screening studies. For the experiments, temperature (4 ℃ and 40 ℃ - TC and TH, respectively), H2O2 (20 mM and 200 mM - Ox1 and Ox2, respectively), mannitol (200 mM and 600 mM - Os1 and Os2, respectively) and NaCl (100 mM and 300 mM - S-Os1 and S-Os2, respectively) were selected as abiotic elicitors. All solutions were freshly prepared before each application. Mannitol (Os1, Os2), NaCl (S-O1, S-O2) and H2O2 (Ox1) treatments were applied by watering daily (not soaking) 2-day-old sprouts with 5 ml of test solution. For Ox2 (200 mM H2O2) treatment 2-day-old seedlings were only once watered with 5 ml of 200 mM H2O2 and then cultivated under standard conditions. For temperature conditioning treatment, 2-day-old sprouts were incubated at 4 ℃ and 40 ℃ (TC and TH, respectively) for 1 h and then cultivated under standard conditions. Sprout (8-day-old) samples were gently collected, weighed (fresh mass), rapidly frozen and kept in polyethylene bags at -20 ℃ .
Click to Show/Hide
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor Function |
Application of abiotic elicitors (environmental shocks) was an effective method for improvement of sprout pro-health potential via an increase of phenolic contents and subsequent elevation of antioxidant potential. Innovative application of elicitors on 2-day-old sprouts (not seed) allowed the elimination of the unfavorable influence of the factors employed on germination yield and biomass production. Assuming that the optimal germination conditions are those which most effectively increase the antioxidant potential without any negative influence on biomass accumulation and nutritional quality the elicitation with 20 mM H2O2 for the future applications is recommended.
Click to Show/Hide
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor | Part | Location | NP Content | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Normal condition
|
Sprouts | NA |
NP Content: 0.00108 ± 0.0001 mg/g flour
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Induction with 20 mM H2O2
|
Sprouts | NA |
NP Content: 0.00294 ± 0.0009 mg/g flour
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Induction with 200 mM H2O2
|
Sprouts | NA |
NP Content: 0.00209 ± 0.0009 mg/g flour
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Induction with 200 mM mannitol
|
Sprouts | NA |
NP Content: 0.00186 ± 0.0001 mg/g flour
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Induction with 600 mM mannitol
|
Sprouts | NA |
NP Content: 0.0023 ± 0.0002 mg/g flour
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Induction with 100 mM NaCl
|
Sprouts | NA |
NP Content: 0.00098 ± 0.0001 mg/g flour
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Induction with 300 mM NaCl
|
Sprouts | NA |
NP Content: 0.00286 ± 0.0001 mg/g flour
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Induction at 4 ℃
|
Sprouts | NA |
NP Content: 0.00116 ± 0 mg/g flour
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Induction at 40 ℃
|
Sprouts | NA |
NP Content: 0.00141 ± 0.0002 mg/g flour
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Species Name: Vitis vinifera cv. Pinot noir | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor Name: Drought Stress Treatment | [6] | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Experiment Detail |
3-year old single shoot V. vinifera plants (cultivar Pinot noir 18 Gm grafted on Kober 5BB, 51 plants) potted in 3L pots in a sandy loam soil were used. All plants were well watered (200 mL per day) at the beginning of the experiment (04.06.2010; DAY 0; 5 plants) and water was supplied to all control plants once every day (250 mL per day), whereas water supply of stressed plants was stopped. Physiological measurements and sampling of leaves took place on 07.06.2010 (DAY 3; 5 control, 5 stressed plants), 10.06.2010 (DAY 6; 5 control, 5 stressed plants) and 12.06.2010 (DAY 8; 5 control, 10 stressed plants). Due to very hot weather conditions in June 2010 the experiment was stopped after 8 days and 12 available control plants were used to restart the drought treatment with 6 control and 6 stressed plants on 11.06.2010 and all plants were measured on 15.06.2010 (DAY 5). The mean leaf temperatures at midday were: 25 ℃ (04.06.2010; DAY 0), 31.9 ℃ (07.06.2010; DAY 3), 30.8 ℃ (15.06.2010; DAY 5), 35.8 ℃ (10.06.2010; DAY 6) and 35.7 ℃ (12.06.2010; DAY 8). The mean PAR radiation per day (measured from 6:00 am till 7:00 pm) was 144.1 µmol m-2 s-1. Each plant was used only once for physiological measurements and sampling of leaves.On every day of the experiment (day 0, 3, 5, 6, 8) the pot weight and the volumetric soil moisture content (ThetaProbe ML2x and handheld data logger Moisture Meter HH2, Delta-T Devices, Cambridge, United Kingdom) was recorded. The water potential (PWSC Model 3000, Soilmoisture Equipment Corporation, Santa Barbara, USA) was determined for the 6th leaf (representing the insertion level of the shoot from the basis) of every plant and measurement day. Chlorophyll fluorescence and gas exchange parameters of light adapted leaves were determined with the 4th and 5th leaf, whereas dark adaptation was performed only with the 5th leaf. Immediately after these non-invasive measurements, the 5th leaf was harvested, frozen in liquid nitrogen and further used for the measurement of polyphenols, selected primary metabolites and volatiles (VOCs).
Click to Show/Hide
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor Function |
The content of different groups of primary and secondary metabolites is significantly influenced by severe drought stress in grapevine leaves. The content of the majority of the metabolites (around 60% of primary metabolites, around 85% of polyphenols and about 40% of the detected and identified VOCs) increased upon drought stress treatment. Among these especially the primary metabolites citric acid and glyceric acid were strongly influenced by the short as well as the prolonged drought stress treatment, whereas all polyphenols were only induced upon the prolonged drought stress treatment.
Click to Show/Hide
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor | Part | Location | NP Content | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Normal condition
|
Leaves | Vienna, Austria |
NP Content: 0.12 ± 0.05 µg/g dry weight
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Dry 3-5 days
|
Leaves | Vienna, Austria |
NP Content: 0.12 ± 0.10 µg/g dry weight
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Dry 6-8 days
|
Leaves | Vienna, Austria |
NP Content: 0.21 ± 0.12 µg/g dry weight
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||