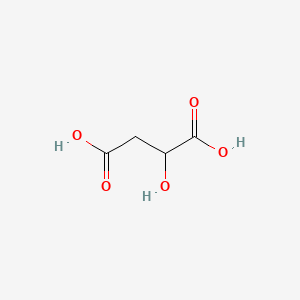
| The Content Variation of Natural Product Induced by Different Factor(s) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Species Name: Brassica juncea (var. RLC-1) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor Name: CdCl2 Treatment; Earthworms Treatment | [1] | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Experiment Detail |
The experiments were conducted under controlled conditions using plastic pots having lower diameter of 7.8 cm, upper diameter of 13.5 cm and 12 cm in height. The soil was collected from the top layer (0-20 cm) from the Botanical Garden of the university. Soil was air dried crushed and sieved through 2 mm filter autoclaved at 121 ℃ for 2 h. The soil was autoclaved to exclude soil pathogens and other microorganisms if any. The autoclaved soil was poured in pots and kept in the growth chamber. The pots were filled with 500 g uncontaminated soil and partially decayed compost (cow manure) (2:1) and was used as growing medium. The cow dung was added into the soil for better performance of earthworms. A subsample of the study soil before mixing with compost was analyzed for its physicochemical characteristics. The soil used for the experiment was sandy loam soil having pH 7.8 , EC (Electrical conductivity) (µS/cm) =184.25 , TDS (Total Dissolved Solids) (mg/kg) = 130 , N (Nitrogen) (mg/kg) = 103 , P (Phosphorus) (mg/kg) = 10.6 , K (Potassium) (mg/kg) = 0.343 , %OC = 0.894, Cd (mg/kg) = ND (not detected by AAS).The Cd treatment was given by using anhydrous CdCl2 (Minimum assay: 95.0%) procured from Hi-Media laboratories. The CdCl2 anhydrous was added to the soil to make different concentrations of Cd 0.50 mM, 0.75 mM, 1.00 mM, and 1.25 mM (i.e. 56 mg/Kg , 84 mg/Kg , 112 mg/Kg and 140 mg/Kg respectively). The various treatments given are as shown below:(1)C0 (Control): (Cadmium absence);(2)C1: (0.5 mM Cd);(3)C2: (0.75 mM Cd);(4)C3: (1.00 mM Cd);(5)C4: (1.25 mM Cd).Each Cd treatment was given in soils without as well as with earthworms (WTE = without, WE = with earthworms). Earthworms (3 earthworms per pot) were inoculated after seven days of Cd treatment and incubated for 7 d in soil with earthworms. The seeds after surface sterilization were sown in soil containing different concentration of Cd and earthworms in plastic pots. These pots were kept in seed germinator under controlled conditions i.e. 25 ℃ temperature and 16:8 h dark: light photoperiod (1700 lx) for 15 d. Seedlings were harvested after 15 d followed by washing with distilled water. The growth and biochemical analysis was done on these seedlings.
Click to Show/Hide
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor Function |
Increased Cd uptake in plants in presence of earthworms enhances the total antioxidative capacity, metal chelating compounds and content of other antioxidants in plants grown under metal polluted soils. Earthworms can improve plant growth by improving nutrient availability to plants through their vermicasting activity. Their role in modifying soil pH and increasing metal phytoavailability made their use ideal in phytoremediation of polluted soils. Increased uptake and accumulation of Cd in plants activates the antioxidative system of plants takes place by addition of earthworms to soil.
Click to Show/Hide
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Mechanism |
The gene expression for the key enzymes involved in organic acid metabolism was studied to understand the role of earthworms in organic acid metabolism in plants under Cd metal stress. It was observed that in comparison to control (C0) seedlings the expression of CS, SUCLG1, SDH and FH was enhanced 1.72, 1.58, 1.65 and 1.88 folds in seedlings given C4 treatment with 1.25 mM dose of Cd respectively . However, after supplementation of earthworms to Cd treated soils given C4 treatment resulted in further enhancement in expression of CS (2.53 fold), SUCLG1 (2.35 fold), SDH (2.13 fold) and FH (3.06 fold) .
Click to Show/Hide
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor | Part | Location | NP Content | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
0.5 mM CdCl2 + without earthworms
|
NA | Ludhiana, India. |
NP Content: 1.319 ± 0.039 mg/g
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
0.75 mM CdCl2 + without earthworms
|
NA | Ludhiana, India. |
NP Content: 1.676 ± 0.083 mg/g
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
1.00 mM CdCl2 + without earthworms
|
NA | Ludhiana, India. |
NP Content: 2.024 ± 0.073 mg/g
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
1.25 mM CdCl2 + without earthworms
|
NA | Ludhiana, India. |
NP Content: 2.366 ± 0.083 mg/g
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
0 mM CdCl2 + with earthworms
|
NA | Ludhiana, India. |
NP Content: 1.324 ± 0.053 mg/g
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
0 mM CdCl2 + without earthworms
|
NA | Ludhiana, India. |
NP Content: 1.263 ± 0.012 mg/g
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
0.5 mM CdCl2 + with earthworms
|
NA | Ludhiana, India. |
NP Content: 1.451 ± 0.117 mg/g
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
0.75 mM CdCl2 + with earthworms
|
NA | Ludhiana, India. |
NP Content: 1.975 ± 0.128 mg/g
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
1.00 mM CdCl2 + with earthworms
|
NA | Ludhiana, India. |
NP Content: 2.402 ± 0.053 mg/g
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
1.25 mM CdCl2 + with earthworms
|
NA | Ludhiana, India. |
NP Content: 2.782 ± 0.039 mg/g
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor Name: 24-epibrassinolide Treatment; Imidacloprid Treatment | [2] | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Experiment Detail |
Seeds of B. juncea (cv. RLC-1) were given pre-sowing treatment with 24-epibrassinolide (EBR) solutions (0 and 100 nM EBR/L) for 8 h. Petri-plates were lined with Whatman1 filter paper and were supplemented with different imidacloprid (IMI) concentrations (0, 150, 200, and 250 mg IMI/L). The EBR treated seeds were rinsed with distilled water and grown in Petri-plates supplemented with IMI solutions (three petri-plates for each treatment). The Petri-plates were kept in seed germinator (temperature = 25 ℃ , photoperiod = 16 h, light intensity = 175 µmol m -2 s-1) and the seedlings were harvested 10 days after sowing for further analysis.
Click to Show/Hide
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor Function |
Seed soaking with 24-epibrassinolide recovers the impaired growth of B. juncea seedlings under imidacloprid stress by modulating the expression of genes encoding key enzymes including chlorophyllase, citrate synthase, succinyl Co-A ligase, succinate dehydrogenase, fumarate hydratase, malate synthase, phytoene synthase, chalcone synthase, and phenylalanine ammonialyase.
Click to Show/Hide
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Mechanism |
In the present study, as compared to control seedlings, the expression of gene CHLASE (encoding chlorophyllase) was observed to increase by 2.66-fold under IMI toxicity, but seed soaking with EBR significantly reduced the expression of CHLASE to 1.07-fold in the seedlings under IMI toxicity . Data analysis using two-way ANOVA and Tukey's HSD showed significant difference for CHLASE expression in B. juncea seedlings (FIMI p < 0.01, FEBR p < 0.01, FIMI * EBR p < 0.001). MLR analysis of the fold change in CHLASE expression also revealed the increased expression of gene with IMI toxicity and EBR application (positive betaIMI-value), whereas interaction between IMI and EBR was observed to be negative .Further, in comparison to control seedlings, the expression of PSY (encoding phytoene synthase) and CHS (encoding chalcone synthase) was significantly enhanced by 5.22 and 4.54-folds respectively in the seedlings raised from EBR treated as well as untreated seeds grown under IMI stress . Significant differences in expression PSY (FIMI p < 0.001, FEBR P<0.05) and CHS (FIMI * EBR p < 0.001) were observed after analyzing the data using two-way ANOVA and Tukey's HSD. MLR analysis of fold change in gene expression also revealed the role of EBR in modulation of gene expression of PSY and CHS. Concentrations of IMI as well as EBR were regressed positively on the fold change in gene expression of PSY and CHS, thus revealing enhanced expressions of these genes under both the treatments. Moreover, interaction between IMI and EBR was positive for PSY expression, whereas negative interaction was observed for the expression of CHS .In the present study, the expression of PAL was also observed to enhance significantly by 6.68-fold in the seedlings raised from EBR treated seeds and grown under IMI stress . After analyzing the data using two-way ANOVA and Tukey's HSD, significant difference in the expression of PAL was observed (FIMI p < 0.01, FEBR p < 0.01, FIMI * EBR P<0.05). MLR analysis of the fold change in gene expression also confirmed the role of EBR in increasing the PAL gene expression under IMI pesticide stress. Positive beta-regression coefficients were observed for IMI, EBR, and IMI * EBR .The expression of genes encoding the key enzymes involved in organic acid metabolism was also studied to understand the role of EBR in organic acid metabolism under IMI pesticide stress. It was observed that as compared to control seedlings, the expression of CS (encoding citrate synthase, 2.35-fold), SUCLG1 (encoding succinyl-Co-A ligase, 1.57-fold), SDH (encoding succinate dehydrogenase, 2.01-fold), FH (encoding fumarate hydratase, 1.57-fold), and MS (encoding malate synthase, 1.91-fold) were increased in B. juncea seedlings raised from untreated seeds and grown under IMI pesticide toxicity . However, seed soaking with 100 nM EBR and germinating them under IMI toxicity resulted in further enhancement in expression of CS (2.61-fold), SUCLGD1 (4.18-fold), SDH (2.55-fold), FH (3.73-fold), and MS (4.03-fold). Data analysis using two-way ANOVA and Tukey's HSD showed significant differences in the expression of CS (FEBR p < 0.01, FIMI * EBR p < 0.01), SUCLG1 (FEBR p < 0.001, FIMI * EBR P<0.05), SDH (FEBR p < 0.01), FH (FEBR p < 0.001), and MS (FEBR p < 0.001). MLR analysis showed that gene expression in seedlings under IMI stress as well as after the EBR seed treatment was increased as indicated by positive beta-regression coefficients. Whereas, negative interactions were noticed between IMI and EBR treatments for the expression of all genes studied related to organic acid metabolism.
Click to Show/Hide
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor | Part | Location | NP Content | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
0 nM 24-epibrassinolide + 0 mg/L Imidacloprid
|
Fresh seedlings | NA |
NP Content: 1.39 ± 0.06 mg/g dry weight
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
100 nM 24-epibrassinolide + 0 mg/L Imidacloprid
|
Fresh seedlings | NA |
NP Content: 1.47 ± 0.08 mg/g dry weight
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
0 nM 24-epibrassinolide + 150 mg/L Imidacloprid
|
Fresh seedlings | NA |
NP Content: 2.81 ± 0.31 mg/g dry weight
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
100 nM 24-epibrassinolide + 150 mg/L Imidacloprid
|
Fresh seedlings | NA |
NP Content: 5.94 ± 1.77 mg/g dry weight
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
0 nM 24-epibrassinolide + 200 mg/L Imidacloprid
|
Fresh seedlings | NA |
NP Content: 2.82 ± 0.05 mg/g dry weight
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
100 nM 24-epibrassinolide + 200 mg/L Imidacloprid
|
Fresh seedlings | NA |
NP Content: 5.91 ± 1.03 mg/g dry weight
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
0 nM 24-epibrassinolide + 250 mg/L Imidacloprid
|
Fresh seedlings | NA |
NP Content: 3.29 ± 0.07 mg/g dry weight
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
100 nM 24-epibrassinolide + 250 mg/L Imidacloprid
|
Fresh seedlings | NA |
NP Content: 4.59 ± 0.46 mg/g dry weight
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Species Name: Fragaria × ananassa Duch. | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor Name: Phosphate Treatment; AMF Inoculation; Nutrient Treatment | [3] | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Experiment Detail |
Seedlings of Fragaria × ananassa Duch var. Elyana F1 with continuous flowering habit, were transplanted in plastic pots (400 mL) in a soil (Brill Ortopack, Agrochimica, Bolzano, Italy; pH 5.5-6.5) previously sterilized by flowing steam (101℃ for 1 h), and kept in a greenhouse for rooting. After 1 month, they were transplanted in new plastic pots (900 mL) in a 2/1 v/v mixture of sterile soil (the same used in the first transplant) and sand (sterilized in oven at 180℃ for 3 h), and inoculated or not with one of three different AMF in combination with one of three different bacterial strains . Plants were initially irrigated 3 times per week. When the plants began to produce fruits, they were transplanted again in plastic pots of 3 L capacity. Starting from 1 week after the last transplant, they were irrigated daily: once per week with a Long Ashton (LA) nutrient solution , and with tap water on the other days. In particular, half of the control plants (C) were watered with LA 32 µM phosphate, while the remaining controls (C-P) and all the inoculated plants were fed with LA 16 µM phosphate until harvest. Strawberry plants were maintained in greenhouse for 16 weeks.The arbuscular mycorrhizal fungus Rhizophagus irregularis (Ri) (DAOM197-198) was provided by INRA (Recorbet and Bernaud, Dijon). Funneliformis mosseae (Fm) (BEG12) was provided by the European Bank of Glomales (Dijon). Septoglomus viscosum (Sv), collected from an Italian soil (Tuscany, Italy), was produced and provided by Mybasol S.r.l. (Alessandria, Italy). The three inocula, prepared as a mixture of soil, mycorrhizal roots, hyphae, and spores, were mixed (11% v/v) with the plant growth medium.Pseudomonas fluorescens strain Pf4 (Pf4) was isolated from a forest soil located in Sassello (Savona, Italy) and characterized by Berta et al. . Pseudomonas sp. 5Vm1K (5Vm) was isolated from the rhizosphere of blueberry plants grown in a larch woodland (Bellino, Cuneo, Italy) and characterized as described by Bona et al. . P. fluorescens strain 19Fv1t (19Fv) was provided by Mybasol s.r.l (Alessandria, Italy) and characterized by Bona et al. . Bacterial 16S rDNA sequences were deposited in the NCBI database GenBank with the accession numbers KF234076, KF233995, KF752592 for Pf4, 5Vm, and 19Fv, respectively.Bacterial cells were grown on tryptic soy agar (TSA) at 28℃ for 48 h and suspended in 0.1M MgSO4. Bacterial density (600 nm) was adjusted to 109 CFU/mL. Each plant was inoculated with 8 mL of bacterial suspension, except uninoculated ones that were irrigated with the same quantity of MgSO4. After 1 week the plants were inoculated again.
Click to Show/Hide
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor Function |
In general, AMF mostly affected the parameters associated with the vegetative portion of the plant, while plant growth promoting bacteria (PGPB) were especially relevant for fruit yield and quality. The plant physiological status was differentially affected by inoculations, resulting in enhanced root and shoot biomass. Inoculation with Pf4 bacterial strain increased flower and fruit production per plant and malic acid content in fruits, while decreased the pH value, regardless of the used fungus. Inoculations affected fruit nutritional quality, increasing sugar and anthocyanin concentrations, and modulated pH, malic acid, volatile compounds and elements.
Click to Show/Hide
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor | Part | Location | NP Content | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
32 µM phosphate (P) in Long Ashton (LA) nutrient solution
|
Fruits | NA |
NP Content: 0.00078 ± 0.00009 g/kg
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
16 µM P in LA nutrient solution
|
Fruits | NA |
NP Content: 0.0006 ± 0.0001 g/kg
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
(Funneliformis mosseae BEG12 (Fm) and Pseudomonas fluorescens 19Fv1t (19Fv) inoculation) + (16 µM P in LA)
|
Fruits | NA |
NP Content: 0.00063 ± 0.00012 g/kg
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
(Fm and Pseudomonas sp. 5Vm1K (5Vm) inoculation) + (16 µM P in LA)
|
Fruits | NA |
NP Content: 0.00058 ± 0.00007 g/kg
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
(Fm and Pseudomonas fluorescens Pf4 (Pf4) inoculation) + (16 µM P in LA)
|
Fruits | NA |
NP Content: 0.00079 ± 0.00009 g/kg
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
(Septoglomus viscosum (Sv) and (19Fv) inoculation) + (16 µM P in LA)
|
Fruits | NA |
NP Content: 0.00088 ± 0.00008 g/kg
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
(Sv and 5Vm inoculation) + (16 µM P in LA)
|
Fruits | NA |
NP Content: 0.00077 ± 0.00009 g/kg
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
(Sv and Pf4 inoculation) + (16 µM P in LA)
|
Fruits | NA |
NP Content: 0.00082 ± 0.00008 g/kg
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
(Rhizophagus irregularis DAOM197-198 (Ri) and 19Fv inoculation) + (16 µM P in LA)
|
Fruits | NA |
NP Content: 0.00053 ± 0.00009 g/kg
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
(Ri and 5Vm inoculation) + (16 µM P in LA)
|
Fruits | NA |
NP Content: 0.0005 ± 0.00008 g/kg
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
(Ri and Pf4 inoculation) + (16 µM P in LA)
|
Fruits | NA |
NP Content: 0.00079 ± 0.00008 g/kg
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Species Name: Vitis vinifera cv. Pinot noir | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor Name: Drought Stress Treatment | [4] | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Experiment Detail |
3-year old single shoot V. vinifera plants (cultivar Pinot noir 18 Gm grafted on Kober 5BB, 51 plants) potted in 3L pots in a sandy loam soil were used. All plants were well watered (200 mL per day) at the beginning of the experiment (04.06.2010; DAY 0; 5 plants) and water was supplied to all control plants once every day (250 mL per day), whereas water supply of stressed plants was stopped. Physiological measurements and sampling of leaves took place on 07.06.2010 (DAY 3; 5 control, 5 stressed plants), 10.06.2010 (DAY 6; 5 control, 5 stressed plants) and 12.06.2010 (DAY 8; 5 control, 10 stressed plants). Due to very hot weather conditions in June 2010 the experiment was stopped after 8 days and 12 available control plants were used to restart the drought treatment with 6 control and 6 stressed plants on 11.06.2010 and all plants were measured on 15.06.2010 (DAY 5). The mean leaf temperatures at midday were: 25 ℃ (04.06.2010; DAY 0), 31.9 ℃ (07.06.2010; DAY 3), 30.8 ℃ (15.06.2010; DAY 5), 35.8 ℃ (10.06.2010; DAY 6) and 35.7 ℃ (12.06.2010; DAY 8). The mean PAR radiation per day (measured from 6:00 am till 7:00 pm) was 144.1 µmol m-2 s-1. Each plant was used only once for physiological measurements and sampling of leaves.On every day of the experiment (day 0, 3, 5, 6, 8) the pot weight and the volumetric soil moisture content (ThetaProbe ML2x and handheld data logger Moisture Meter HH2, Delta-T Devices, Cambridge, United Kingdom) was recorded. The water potential (PWSC Model 3000, Soilmoisture Equipment Corporation, Santa Barbara, USA) was determined for the 6th leaf (representing the insertion level of the shoot from the basis) of every plant and measurement day. Chlorophyll fluorescence and gas exchange parameters of light adapted leaves were determined with the 4th and 5th leaf, whereas dark adaptation was performed only with the 5th leaf. Immediately after these non-invasive measurements, the 5th leaf was harvested, frozen in liquid nitrogen and further used for the measurement of polyphenols, selected primary metabolites and volatiles (VOCs).
Click to Show/Hide
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor Function |
The content of different groups of primary and secondary metabolites is significantly influenced by severe drought stress in grapevine leaves. The content of the majority of the metabolites (around 60% of primary metabolites, around 85% of polyphenols and about 40% of the detected and identified VOCs) increased upon drought stress treatment. Among these especially the primary metabolites citric acid and glyceric acid were strongly influenced by the short as well as the prolonged drought stress treatment, whereas all polyphenols were only induced upon the prolonged drought stress treatment.
Click to Show/Hide
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor | Part | Location | NP Content | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Normal condition
|
Leaves | Vienna, Austria |
NP Content: 2051.3 ± 363.7 µg/g dry weight
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Dry 3-5 days
|
Leaves | Vienna, Austria |
NP Content: 2153.0 ± 357.5 µg/g dry weight
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Dry 6-8 days
|
Leaves | Vienna, Austria |
NP Content: 1190.1 ± 542.0 µg/g dry weight
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||