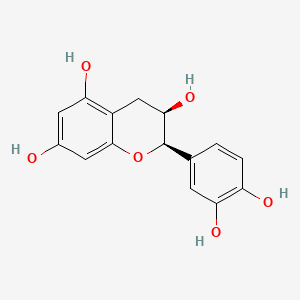
| The Content Variation of Natural Product Induced by Different Factor(s) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Species Name: Brassica juncea (var. RLC-1) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor Name: CdCl2 Treatment; Earthworms Treatment | [1] | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Experiment Detail |
The experiments were conducted under controlled conditions using plastic pots having lower diameter of 7.8 cm, upper diameter of 13.5 cm and 12 cm in height. The soil was collected from the top layer (0-20 cm) from the Botanical Garden of the university. Soil was air dried crushed and sieved through 2 mm filter autoclaved at 121 ℃ for 2 h. The soil was autoclaved to exclude soil pathogens and other microorganisms if any. The autoclaved soil was poured in pots and kept in the growth chamber. The pots were filled with 500 g uncontaminated soil and partially decayed compost (cow manure) (2:1) and was used as growing medium. The cow dung was added into the soil for better performance of earthworms. A subsample of the study soil before mixing with compost was analyzed for its physicochemical characteristics. The soil used for the experiment was sandy loam soil having pH 7.8 , EC (Electrical conductivity) (µS/cm) =184.25 , TDS (Total Dissolved Solids) (mg/kg) = 130 , N (Nitrogen) (mg/kg) = 103 , P (Phosphorus) (mg/kg) = 10.6 , K (Potassium) (mg/kg) = 0.343 , %OC = 0.894, Cd (mg/kg) = ND (not detected by AAS).The Cd treatment was given by using anhydrous CdCl2 (Minimum assay: 95.0%) procured from Hi-Media laboratories. The CdCl2 anhydrous was added to the soil to make different concentrations of Cd 0.50 mM, 0.75 mM, 1.00 mM, and 1.25 mM (i.e. 56 mg/Kg , 84 mg/Kg , 112 mg/Kg and 140 mg/Kg respectively). The various treatments given are as shown below:(1)C0 (Control): (Cadmium absence);(2)C1: (0.5 mM Cd);(3)C2: (0.75 mM Cd);(4)C3: (1.00 mM Cd);(5)C4: (1.25 mM Cd).Each Cd treatment was given in soils without as well as with earthworms (WTE = without, WE = with earthworms). Earthworms (3 earthworms per pot) were inoculated after seven days of Cd treatment and incubated for 7 d in soil with earthworms. The seeds after surface sterilization were sown in soil containing different concentration of Cd and earthworms in plastic pots. These pots were kept in seed germinator under controlled conditions i.e. 25 ℃ temperature and 16:8 h dark: light photoperiod (1700 lx) for 15 d. Seedlings were harvested after 15 d followed by washing with distilled water. The growth and biochemical analysis was done on these seedlings.
Click to Show/Hide
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor Function |
Increased Cd uptake in plants in presence of earthworms enhances the total antioxidative capacity, metal chelating compounds and content of other antioxidants in plants grown under metal polluted soils. Earthworms can improve plant growth by improving nutrient availability to plants through their vermicasting activity. Their role in modifying soil pH and increasing metal phytoavailability made their use ideal in phytoremediation of polluted soils. Increased uptake and accumulation of Cd in plants activates the antioxidative system of plants takes place by addition of earthworms to soil.
Click to Show/Hide
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Mechanism |
The gene expression for the key enzymes involved in organic acid metabolism was studied to understand the role of earthworms in organic acid metabolism in plants under Cd metal stress. It was observed that in comparison to control (C0) seedlings the expression of CS, SUCLG1, SDH and FH was enhanced 1.72, 1.58, 1.65 and 1.88 folds in seedlings given C4 treatment with 1.25 mM dose of Cd respectively . However, after supplementation of earthworms to Cd treated soils given C4 treatment resulted in further enhancement in expression of CS (2.53 fold), SUCLG1 (2.35 fold), SDH (2.13 fold) and FH (3.06 fold) .
Click to Show/Hide
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor | Part | Location | NP Content | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
0.75 mM CdCl2 + without earthworms
|
NA | Ludhiana, India. |
NP Content: 0.000947 ± 0.000004 mg/g
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
1.00 mM CdCl2 + without earthworms
|
NA | Ludhiana, India. |
NP Content: 0.000667 ± 0.000026 mg/g
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
1.25 mM CdCl2 + without earthworms
|
NA | Ludhiana, India. |
NP Content: 0.000996 ± 0.000061 mg/g
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
0.5 mM CdCl2 + with earthworms
|
NA | Ludhiana, India. |
NP Content: 0.002338 ± 0.000379 mg/g
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
0.75 mM CdCl2 + with earthworms
|
NA | Ludhiana, India. |
NP Content: 0.001106 ± 0.000034 mg/g
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
1.25 mM CdCl2 + with earthworms
|
NA | Ludhiana, India. |
NP Content: 0.001148 ± 0.000067 mg/g
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Species Name: Crocus sativus L. (saffron) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor Name: AMF Inoculation; Harvest Time Variation | [2] | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Experiment Detail |
AMF Inoculation in Pot : Saffron corms with horizontal diameters of 1.3 to 2.8 cm were sown in pots (4 L; 1 corm per pot) in the last ten days of August 2016. Pots were filled with sterile quartz sand (3 L per pot) on a layer of sterilized expanded clay (1 L per pot). Corms were treated with two inocula (MycAgro Lab, Breteniere, FR), one composed of a single fungus Rhizophagus intraradices (Ri) and one of R. intraradices and Funneliformis mosseae (Ri + Fm). Ten grams of each inoculum were placed under each corm in order to guarantee the contact between the inoculum and the roots and therefore to favor the symbiosis between AMF and roots. Saffron corms used as controls were not inoculated (AMF-). Corms were not treated against fungal pathogens. A randomized block design was used with a total of 48 pots displayed in two experimental plot units (24 pots per unit) and three treatments (8 pots per treatment). Cultivation lasted for one cycle (August 2016-April 2017) in a heated glasshouse of the Department of Agricultural Forest and Food Sciences (DISAFA) of the University of Torino (Italy, 45° 06′ 23.21″ N Lat, 7° 57′ 82.8″ E Long; 293 m a.s.l.), with an average temperature of 22 ℃ during the day and 16 ℃ in the night. Irrigation water (pH 7.4, EC 505 µS cm) was added weekly (250 mL per pot) with a drip system. The corms were fertilized by fertigation (VIGORFLOR, AL.FE. srl, MN, Italy) every two weeks starting from the emergence of the spate, in quantities of 1.5 g/L of water. No flowering occurred because of the small size of the corms.AMF Inoculation in Open Field : Saffron corms with horizontal diameters of 2.5 to 3.5 cm were planted in the last ten days of August 2016 in two Alpine experimental sites located in the municipality of Morgex (45° 45′ 35″ N; 7° 02′ 37.3″ E; 1000 m a.s.l.) and Saint Cristophe (45° 45′ 06″ N; 7° 20′ 37″ E; 700 m a.s.l.) in Italy and cultivation lasted for two cycles (2016-2017 and 2017-2018). Both sites were cultivated with saffron for at least the previous three years. Before starting the experiment both fields were milled. To assess the effects of AMF inocula on saffron cultivation and production, the same treatments used in the pot trial were applied (Ri, Ri + Fm or AMF-). A randomized block design was used, with three experimental plot units (blocks). Each plot unit consisted of 56 corms, planted in a 1.44 m2 area (39 corms m-2). Inter-row planting distance was of 7 cm, while between-row distance was 25 cm. Plots were separated from each other with at least 4 m distance. Before planting, 10 g of inoculum was placed under the corms to ensure contact between plant and the treatment. Irrigation was provided when needed and hand weeding control was conducted during cultivation, while no preplanting fertilization, tillage, or treatments against pathogens were applied. The two Alpine sites were characterized by semicontinental climate, with a long and cold winter . In general, both sites had a sandy-loam texture according to the USDA classification and similar chemical characteristics.
Click to Show/Hide
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor Function |
The inoculum composed by R. intraradices and F. mosseae was particularly effective in increasing flower production and saffron yield, while R. intraradices alone increased the content of some bioactive compounds-picrocrocin, quercitrin, crocin II-as well as antioxidant activity.
Click to Show/Hide
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor | Part | Location | NP Content | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Harvesting time: 2016-2017
|
Powdered saffrons | Italy |
NP Content: 6 mg/100g dry weight
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Harvesting time: 2017-2018
|
Powdered saffrons | Italy |
NP Content: 8 mg/100g dry weight
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Rhizophagus intraradices and Funneliformis mosseae inoculation
|
Powdered saffrons | Italy |
NP Content: 6 mg/100g dry weight
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Rhizophagus intraradices inoculation
|
Powdered saffrons | Italy |
NP Content: 6 mg/100g dry weight
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Non-AMF inoculation (Control)
|
Powdered saffrons | Italy |
NP Content: 9 mg/100g dry weight
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Species Name: Vitis vinifera cv. Pinot noir | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor Name: Drought Stress Treatment | [3] | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Experiment Detail |
3-year old single shoot V. vinifera plants (cultivar Pinot noir 18 Gm grafted on Kober 5BB, 51 plants) potted in 3L pots in a sandy loam soil were used. All plants were well watered (200 mL per day) at the beginning of the experiment (04.06.2010; DAY 0; 5 plants) and water was supplied to all control plants once every day (250 mL per day), whereas water supply of stressed plants was stopped. Physiological measurements and sampling of leaves took place on 07.06.2010 (DAY 3; 5 control, 5 stressed plants), 10.06.2010 (DAY 6; 5 control, 5 stressed plants) and 12.06.2010 (DAY 8; 5 control, 10 stressed plants). Due to very hot weather conditions in June 2010 the experiment was stopped after 8 days and 12 available control plants were used to restart the drought treatment with 6 control and 6 stressed plants on 11.06.2010 and all plants were measured on 15.06.2010 (DAY 5). The mean leaf temperatures at midday were: 25 ℃ (04.06.2010; DAY 0), 31.9 ℃ (07.06.2010; DAY 3), 30.8 ℃ (15.06.2010; DAY 5), 35.8 ℃ (10.06.2010; DAY 6) and 35.7 ℃ (12.06.2010; DAY 8). The mean PAR radiation per day (measured from 6:00 am till 7:00 pm) was 144.1 µmol m-2 s-1. Each plant was used only once for physiological measurements and sampling of leaves.On every day of the experiment (day 0, 3, 5, 6, 8) the pot weight and the volumetric soil moisture content (ThetaProbe ML2x and handheld data logger Moisture Meter HH2, Delta-T Devices, Cambridge, United Kingdom) was recorded. The water potential (PWSC Model 3000, Soilmoisture Equipment Corporation, Santa Barbara, USA) was determined for the 6th leaf (representing the insertion level of the shoot from the basis) of every plant and measurement day. Chlorophyll fluorescence and gas exchange parameters of light adapted leaves were determined with the 4th and 5th leaf, whereas dark adaptation was performed only with the 5th leaf. Immediately after these non-invasive measurements, the 5th leaf was harvested, frozen in liquid nitrogen and further used for the measurement of polyphenols, selected primary metabolites and volatiles (VOCs).
Click to Show/Hide
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor Function |
The content of different groups of primary and secondary metabolites is significantly influenced by severe drought stress in grapevine leaves. The content of the majority of the metabolites (around 60% of primary metabolites, around 85% of polyphenols and about 40% of the detected and identified VOCs) increased upon drought stress treatment. Among these especially the primary metabolites citric acid and glyceric acid were strongly influenced by the short as well as the prolonged drought stress treatment, whereas all polyphenols were only induced upon the prolonged drought stress treatment.
Click to Show/Hide
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor | Part | Location | NP Content | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Normal condition
|
Leaves | Vienna, Austria |
NP Content: 0.5 ± 0.4 µg/g dry weight
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Dry 3-5 days
|
Leaves | Vienna, Austria |
NP Content: 0.7 ± 0.5 µg/g dry weight
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Dry 6-8 days
|
Leaves | Vienna, Austria |
NP Content: 3.3 ± 5.9 µg/g dry weight
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||