| The Content Variation of Natural Product Induced by Different Factor(s) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Species Name: Asparagus aethiopicus L. (A. sprengeri Regel) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor Name: NaCl Treatment; Seaweed extracts Treatment; 5-aminolevulinic acid Treatment; Harvest Time Variation | [1] | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Experiment Detail |
They were grown in 14 cm plastic pots containing soil mixture of peat:sand (1:1) and maintained in a controlled greenhouse conditions in a private nursery located on Alexandria-Cairo Desert Road. The soil was fertilized with 2 g l -1 Crystalon (19% N: 19% P: 19% K, Chema Industries, Egypt). The plants were maintained at natural light conditions and the temperature ranged between 15 and 28 ℃ . Plants were watered every two days with 2000 and 4000 ppm NaCl solution. The plants subjected to saline conditions (watered with 2000 and 4000 ppm NaCl solution) were treated with 7 ml/L weekly application of Seaweed extracts (SWE) (Ascophyllum nodosum, Stella Maris, Acadian Seaplants, Canada) as foliar spray until drop-off two weeks ahead of saline conditions. A foliar spray of 5-aminolevulinic acid (Sigma-Aldrich, Germany) at (3, 5 and 10 ppm) was applied weekly until drop-off to all plants during saline conditions and untreated plants were considered as controls. Experiments continued for 6 weeks in two consecutive seasons of 2016 and 2017 and the plants were distributed in three blocks and each treatment was represented by 5 replicates with a total number of 270 plants. The NaCl was added to the tank gradually and the electrical conductivity (EC) was measured using EC meter.
Click to Show/Hide
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor Function |
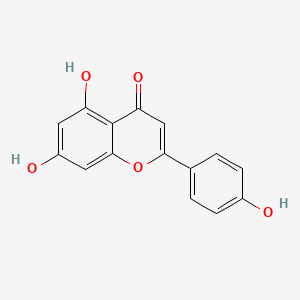
There were significant increases in branch length and number of branches per plant, fresh and the dry weight in Seaweed extracts (SWE) + 5-aminolevulinic acid (ALA) treated plants under saline irrigation conditions compared to control. These morphological improvements associated with several physiological changes in treated plants including increased accumulation of specific phenols (robinin, rutin, apigein, chlorogenic acid and caffeic acid) and increased antioxidant activities of leaf extracts. There were also increased the chlorophyll composition and the accumulation of sugars and proline. Improved transpiration and photosynthetic rates as well as stomatal conductance were also detected in treated plants. The expression of several genes responsible for water management, flavonoid accumulation and antioxidants accumulation was enhanced in SWE + ALA treated plants.
Click to Show/Hide
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Mechanism |
Under normal and saline irrigation conditions, there were changes in genes expression following SWE and ALA applications.The transcription levels of ANN1 and ANN2 increased significantly in SWE + ALA treatments compared to control under non-saline and saline conditions . However, transcription levels of MYB44 showed no significant variations among treatments. PIP1, P5CS1 and CHS relative expressions was higher in plants treated with SWE + ALA under normal and saline irrigation conditions . The transcription levels of the redox responsive genes of APX1 and GPX3 were significantly higher in SWE + ALA treated plants compared to control treatments . Increasing ALA amount from 3 to 10 ppm had no significant effects on APX1 transcription levels under non-saline conditions. In addition, the application of SWE or ALA only had no significant effects on GPX3 transcription levels under non-saline conditions.
Click to Show/Hide
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor | Part | Location | NP Content | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Harvesting time: January-2016 + 640 ppm NaCl + 0 mL/L Seaweed extracts + 0 ppm 5-aminolevulinic acid
|
Leaves | Alexandria, Egypt |
NP Content: 0.521 ± 0.3 mg/g dry weight
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Harvesting time: January-2016 + 640 ppm NaCl + 7 mL/L Seaweed extracts + 0 ppm 5-aminolevulinic acid
|
Leaves | Alexandria, Egypt |
NP Content: 0.559 ± 0.2 mg/g dry weight
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Harvesting time: January-2016 + 640 ppm NaCl + 3 mL/L Seaweed extracts + 0 ppm 5-aminolevulinic acid
|
Leaves | Alexandria, Egypt |
NP Content: 0.553 ± 0.4 mg/g dry weight
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Harvesting time: January-2016 + 640 ppm NaCl + 7 mL/L Seaweed extracts + 3 ppm 5-aminolevulinic acid
|
Leaves | Alexandria, Egypt |
NP Content: 0.564 ± 0.1 mg/g dry weight
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Harvesting time: January-2016 + 640 ppm NaCl + 7 mL/L Seaweed extracts + 5 ppm 5-aminolevulinic acid
|
Leaves | Alexandria, Egypt |
NP Content: 0.568 ± 0.2 mg/g dry weight
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Harvesting time: January-2016 + 640 ppm NaCl + 7 mL/L Seaweed extracts + 10 ppm 5-aminolevulinic acid
|
Leaves | Alexandria, Egypt |
NP Content: 0.604 ± 0.4 mg/g dry weight
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Harvesting time: January-2016 + 2000 ppm NaCl + 0 mL/L Seaweed extracts + 0 ppm 5-aminolevulinic acid
|
Leaves | Alexandria, Egypt |
NP Content: 0.565 ± 0.5 mg/g dry weight
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Harvesting time: January-2016 + 2000 ppm NaCl + 7 mL/L Seaweed extracts + 0 ppm 5-aminolevulinic acid
|
Leaves | Alexandria, Egypt |
NP Content: 0.595 ± 0.3 mg/g dry weight
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Harvesting time: January-2016 + 2000 ppm NaCl + 0 mL/L Seaweed extracts + 3 ppm 5-aminolevulinic acid
|
Leaves | Alexandria, Egypt |
NP Content: 0.593 ± 0.4 mg/g dry weight
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Harvesting time: January-2016 + 2000 ppm NaCl + 7 mL/L Seaweed extracts + 3 ppm 5-aminolevulinic acid
|
Leaves | Alexandria, Egypt |
NP Content: 0.597 ± 0.2 mg/g dry weight
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Harvesting time: January-2016 + 2000 ppm NaCl + 7 mL/L Seaweed extracts + 5 ppm 5-aminolevulinic acid
|
Leaves | Alexandria, Egypt |
NP Content: 0.606 ± 0.3 mg/g dry weight
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Harvesting time: January-2016 + 2000 ppm NaCl + 7 mL/L Seaweed extracts + 10 ppm 5-aminolevulinic acid
|
Leaves | Alexandria, Egypt |
NP Content: 0.647 ± 0.5 mg/g dry weight
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Harvesting time: January-2016 + 4000 ppm NaCl + 0 mL/L Seaweed extracts + 0 ppm 5-aminolevulinic acid
|
Leaves | Alexandria, Egypt |
NP Content: 0.608 ± 0.4 mg/g dry weight
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Harvesting time: January-2016 + 4000 ppm NaCl + 7 mL/L Seaweed extracts + 0 ppm 5-aminolevulinic acid
|
Leaves | Alexandria, Egypt |
NP Content: 0.65 ± 0.1 mg/g dry weight
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Harvesting time: January-2016 + 4000 ppm NaCl + 0 mL/L Seaweed extracts + 3 ppm 5-aminolevulinic acid
|
Leaves | Alexandria, Egypt |
NP Content: 0.645 ± 0.3 mg/g dry weight
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Harvesting time: January-2016 + 4000 ppm NaCl + 7 mL/L Seaweed extracts + 3 ppm 5-aminolevulinic acid
|
Leaves | Alexandria, Egypt |
NP Content: 0.652 ± 0.2 mg/g dry weight
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Harvesting time: January-2016 + 4000 ppm NaCl + 7 mL/L Seaweed extracts + 5 ppm 5-aminolevulinic acid
|
Leaves | Alexandria, Egypt |
NP Content: 0.661 ± 0.3 mg/g dry weight
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Harvesting time: January-2016 + 4000 ppm NaCl + 7 mL/L Seaweed extracts + 10 ppm 5-aminolevulinic acid
|
Leaves | Alexandria, Egypt |
NP Content: 0.705 ± 0.2 mg/g dry weight
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Harvesting time: January-2017 + 640 ppm NaCl + 0 mL/L Seaweed extracts + 0 ppm 5-aminolevulinic acid
|
Leaves | Alexandria, Egypt |
NP Content: 0.501 ± 0.2 mg/g dry weight
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Harvesting time: January-2017 + 640 ppm NaCl + 7 mL/L Seaweed extracts + 0 ppm 5-aminolevulinic acid
|
Leaves | Alexandria, Egypt |
NP Content: 0.538 ± 0.2 mg/g dry weight
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Harvesting time: January-2017 + 640 ppm NaCl + 3 mL/L Seaweed extracts + 0 ppm 5-aminolevulinic acid
|
Leaves | Alexandria, Egypt |
NP Content: 0.533 ± 0.6 mg/g dry weight
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Harvesting time: January-2017 + 640 ppm NaCl + 7 mL/L Seaweed extracts + 3 ppm 5-aminolevulinic acid
|
Leaves | Alexandria, Egypt |
NP Content: 0.545 ± 0.3 mg/g dry weight
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Harvesting time: January-2017 + 640 ppm NaCl + 7 mL/L Seaweed extracts + 5 ppm 5-aminolevulinic acid
|
Leaves | Alexandria, Egypt |
NP Content: 0.553 ± 0.1 mg/g dry weight
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Harvesting time: January-2017 + 640 ppm NaCl + 7 mL/L Seaweed extracts + 10 ppm 5-aminolevulinic acid
|
Leaves | Alexandria, Egypt |
NP Content: 0.587 ± 0.5 mg/g dry weight
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Harvesting time: January-2017 + 2000 ppm NaCl + 0 mL/L Seaweed extracts + 0 ppm 5-aminolevulinic acid
|
Leaves | Alexandria, Egypt |
NP Content: 0.541 ± 0.7 mg/g dry weight
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Harvesting time: January-2017 + 2000 ppm NaCl + 7 mL/L Seaweed extracts + 0 ppm 5-aminolevulinic acid
|
Leaves | Alexandria, Egypt |
NP Content: 0.578 ± 0.4 mg/g dry weight
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Harvesting time: January-2017 + 2000 ppm NaCl + 0 mL/L Seaweed extracts + 3 ppm 5-aminolevulinic acid
|
Leaves | Alexandria, Egypt |
NP Content: 0.573 ± 0.3 mg/g dry weight
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Harvesting time: January-2017 + 2000 ppm NaCl + 7 mL/L Seaweed extracts + 3 ppm 5-aminolevulinic acid
|
Leaves | Alexandria, Egypt |
NP Content: 0.582 ± 0.2 mg/g dry weight
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Harvesting time: January-2017 + 2000 ppm NaCl + 7 mL/L Seaweed extracts + 5 ppm 5-aminolevulinic acid
|
Leaves | Alexandria, Egypt |
NP Content: 0.584 ± 0.2 mg/g dry weight
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Harvesting time: January-2017 + 2000 ppm NaCl + 7 mL/L Seaweed extracts + 10 ppm 5-aminolevulinic acid
|
Leaves | Alexandria, Egypt |
NP Content: 0.637 ± 0.3 mg/g dry weight
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Harvesting time: January-2017 + 4000 ppm NaCl + 0 mL/L Seaweed extracts + 0 ppm 5-aminolevulinic acid
|
Leaves | Alexandria, Egypt |
NP Content: 0.596 ± 0.6 mg/g dry weight
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Harvesting time: January-2017 + 4000 ppm NaCl + 7 mL/L Seaweed extracts + 0 ppm 5-aminolevulinic acid
|
Leaves | Alexandria, Egypt |
NP Content: 0.635 ± 0.3 mg/g dry weight
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Harvesting time: January-2017 + 4000 ppm NaCl + 0 mL/L Seaweed extracts + 3 ppm 5-aminolevulinic acid
|
Leaves | Alexandria, Egypt |
NP Content: 0.626 ± 0.4 mg/g dry weight
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Harvesting time: January-2017 + 4000 ppm NaCl + 7 mL/L Seaweed extracts + 3 ppm 5-aminolevulinic acid
|
Leaves | Alexandria, Egypt |
NP Content: 0.638 ± 0.1 mg/g dry weight
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Harvesting time: January-2017 + 4000 ppm NaCl + 7 mL/L Seaweed extracts + 5 ppm 5-aminolevulinic acid
|
Leaves | Alexandria, Egypt |
NP Content: 0.632 ± 0.0 mg/g dry weight
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Harvesting time: January-2017 + 4000 ppm NaCl + 7 mL/L Seaweed extracts + 10 ppm 5-aminolevulinic acid
|
Leaves | Alexandria, Egypt |
NP Content: 0.683 ± 0.3 mg/g dry weight
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Species Name: Dracocephalum kotschyi Boiss | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor Name: SiO2 NPs Treatment | [2] | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Experiment Detail |
Seeds were treated with sulfuric acid (98%, 10 min) and then surface sterilized with 70% ethanol (v/v) for 1 min and sodium hypochlorite solution (10%, at 10 min). After sterilization, seeds were germinated on MS media (Murashige and Skoog, 1962) containing 7 g/L agar (Duchefa, Netherlands). Cultures were maintained under 16/8 h light/dark. Explants were taken from 4-week-old leaves for inoculation with bacteria strain.ATCC15834 strain of A. rhizogenes was supplied by microbial unit of the National Research Center for Genetic Engineering and Biotechnology, Tehran-Iran. Bacterial cells cultivated on LB (Luria-Bertani) culture medium (Bertani, 1952) on rotary shaker (at 26 ℃ and 180 rpm for 48 h) in the darkened state.The leaves were wounded and inoculated with bacterial suspension for 5 min and transferred to MS media containing 7 g/L agar in darkness at 25 ℃ . After 48 h treated Explants were cultured on the 1/2 MS media containing cefotaxime (500 mg/L) and indole-3-butyric acid (IBA) (2 mg/L). Hairy roots emerged at wounded sites, after 4-weeks of incubation, and then each hairy root line was isolated from explants tissue and was subcultured weekly in new media (1/2 MS hormone-free media) with appropriate antibiotic. The concentration of cefotaxime was decreased gradually and eliminated from the culture medium after 8 subcultures and axenic root cultures were obtained. Then hairy root lines were transferred to the 250 mL Erlenmeyer flasks containing 30 mL hormone- free 1/2 MS liquid medium and incubated on a rotary shaker (120 rpm) at 25 ℃ and subcultured every two week. Hairy root line, which showed sufficient growth in 1/2 MS liquid medium, was selected for further investigations.The genomic DNA was extracted from transformed hairy root lines and plant intact roots with CTAB method . Gene-specific primers from rol B were used for amplification of the 780-bp segment in PCR analysis. The primers sequences were, F:5'-ATGGATCCCAAATTGCTATTCCCCCACGA-3'and R:5'-TTAGGCTTCTTTCATTCGGTTTACTGCAGC-3'. Thirty-five PCR cycles were performed with 5 min initial denaturation at 94 ℃ , annealing steps at 60 ℃ for 80 s, extension at 72 ℃ for 90 s, and final extension step of 72 ℃ for 10 min. The amplimer were analyzed by 1% agarose gel electrophoresis.To investigate the effects of SiO2 NPs, various concentrations (0, 25, 50, 100 and 200 mg/L) of this elicitor were added to the hairy roots culture medium (1/2 MS + 3% sucrose, pH = 5.7) at the end of log phase of growth stages (21-days-old cultures). Hairy roots were incubated with elicitor for 24 and 48 h of exposure time. Hairy roots were harvested 7 days after elicitation and dried on sterile filter paper to remove excess surface moisture and were weighed before freezing by liquid nitrogen and stored at -80℃ until used to measure growth, biochemical and phytochemical analysis.
Click to Show/Hide
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor Function |
The effect of silicon dioxide nanoparticles on production of phenolic compounds and expression rate of pal and ras genes involved in rosmarinic acid biosynthesis pathway has been investigated in D. kotschyi. SiO2 nanoparticles, used as an abiotic elicitor in our study, has appropriate optical, electrical and catalysts properties and has many applications in various industries as well as agriculture. This study clearly suggested that, in the presence of this nanoparticle, induction, production and accumulation of valuable compounds and corresponding antioxidant activity increased in hairy roots.
Click to Show/Hide
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Mechanism |
According to the results, expression levels of the pal and ras genes were influenced by elicitor concentration and exposure time. The elicitation by SiO2 NP of 100 mg/L after 48 h of exposure time dramatically increased pal expression compared to the control. Briefly, with increasing SiO2 NP concentrations after 48 h of exposure time, the expression level of pal was also significantly induced . Similarly, ras expression was significantly raised at 48 h after treatment by increasing SiO2 NP concentration and enhanced to the greatest extent in 50 mg/L concentration. After 24 h of exposure time, the minimum level of ras expression was observed in the 200 mg/L SiO2 . Amplification products of real-time PCR were assessed with 1.8% agarose gel which was corresponded to the predicted size.
Click to Show/Hide
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor | Part | Location | NP Content | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Normal condition
|
Frozen hairy roots | Iran |
NP Content: 193.12 µg/g fresh weight
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
25 mg/L SiO2 NPs + Exposure time: 24 h
|
Frozen hairy roots | Iran |
NP Content: 19.05 µg/g fresh weight
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
50 mg/L SiO2 NPs + Exposure time: 24 h
|
Frozen hairy roots | Iran |
NP Content: 38.13 µg/g fresh weight
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
100 mg/L SiO2 NPs + Exposure time: 24 h
|
Frozen hairy roots | Iran |
NP Content: 34.35 µg/g fresh weight
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
200 mg/L SiO2 NPs + Exposure time: 24 h
|
Frozen hairy roots | Iran |
NP Content: 1.11 µg/g fresh weight
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
25 mg/L SiO2 NPs + Exposure time: 48 h
|
Frozen hairy roots | Iran |
NP Content: 15.96 µg/g fresh weight
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
50 mg/L SiO2 NPs + Exposure time: 48 h
|
Frozen hairy roots | Iran |
NP Content: 7.47 µg/g fresh weight
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
100 mg/L SiO2 NPs + Exposure time: 48 h
|
Frozen hairy roots | Iran |
NP Content: 27.35 µg/g fresh weight
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
200 mg/L SiO2 NPs + Exposure time: 48 h
|
Frozen hairy roots | Iran |
NP Content: 8.54 µg/g fresh weight
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Species Name: Saponaria officinalis | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor Name: Titanium Dioxide Nanoparticles Treatment | [3] | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Experiment Detail |
Different concentrations of TiO2 NPs (0, 10, 20, 30, and 50) were prepared for hairy root treatments. 0.5 g of .S. officinalis hairy roots were transferred to 250 mL Erlenmeyer flasks containing 15 mL of liquid MS culture medium with three replicates. Then, they were placed in an incubator shaker at 110 rpm and 25 ℃ in dark conditions. On the 22nd day, the liquid MS culture media containing different concentrations of nano titanium dioxide was added to Erlenmeyer flasks. 24 and 48 h after treatment, the hairy roots were taken out and transferred to the MS culture medium lacking elicitor.
Click to Show/Hide
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor Function |
The highest rate of total phenol (9.79 mg GLA/g FW) and total flavonoid contents (1.06 mg QE/g FW) were obtained in the treated hairy roots with 50 and 30 mg/L of nano elicitor in 24 and 48 h of treatments, respectively. The maximum level of most polyphenols, such as rosmarinic acid, cinnamic acid, and rutin, was produced in 24 h of treatment. The use of TiO2 NP for 48 h with 50 mg/L concentration showed the highest production level of SO6 protein.
Click to Show/Hide
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor | Part | Location | NP Content | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Nano-TiO2 concentration (mg/L): 0 + Exposure time: 24h
|
hairy roots | NA |
NP Content: 10.97 mg/kg fresh weight
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Nano-TiO2 concentration (mg/L): 25 + Exposure time: 24h
|
hairy roots | NA |
NP Content: 8.59 mg/kg fresh weight
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Nano-TiO2 concentration (mg/L): 50 + Exposure time: 24h
|
hairy roots | NA |
NP Content: 10.9 mg/kg fresh weight
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Nano-TiO2 concentration (mg/L): 100 + Exposure time: 24h
|
hairy roots | NA |
NP Content: 10.35 mg/kg fresh weight
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Nano-TiO2 concentration (mg/L): 200 + Exposure time: 24h
|
hairy roots | NA |
NP Content: 11.48 mg/kg fresh weight
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Nano-TiO2 concentration (mg/L): 0 + Exposure time: 48h
|
hairy roots | NA |
NP Content: 10.97 mg/kg fresh weight
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Nano-TiO2 concentration (mg/L): 25 + Exposure time: 48h
|
hairy roots | NA |
NP Content: 8.59 mg/kg fresh weight
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Nano-TiO2 concentration (mg/L): 50 + Exposure time: 48h
|
hairy roots | NA |
NP Content: 5.95 mg/kg fresh weight
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Nano-TiO2 concentration (mg/L): 100 + Exposure time: 48h
|
hairy roots | NA |
NP Content: 12.96 mg/kg fresh weight
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Nano-TiO2 concentration (mg/L): 200 + Exposure time: 48h
|
hairy roots | NA |
NP Content: 10.72 mg/kg fresh weight
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Species Name: Thymus daenensis | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor Name: NaCl Treatment | [4] | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Experiment Detail |
The seeds were germinated in seedling plastic tray filled up with a mixture of peat moss and coco peat (1:1). The trays were placed in a glass house at the University of Massachusetts, Amherst, USA, in September 2015. After two weeks, seedlings were transplanted into plastic pots (30 cm diameter and 30 cm height, three seedlings per pot). The pots growth media consisted of 2:1:1 ratio of sieved field soil, sand, and leaf mold, respectively. The medium in each pot was supplemented with 5.8 mg P, 3.3 mg N, and 13.8 mg K to warrant the plant growth; the electrical conductivity was 0.3 mS/cm (Beckman EC meter instrument. cedar grove, New jersey, USA); the pH was 8.04. Plants were maintained at the temperature of 18-25 ℃ ; glass house conditions were set as follows: photoperiod, 16 h; relative humidity, 60-70%; light intensity, 180 µmol m 2 s-1. The source of light was a high pressure sodium lamp in addition to the day light in order to attain 16 h of light per day. Fourty days after planting (establishment period), the uniform sized plants were treated with four different salinity levels including: 0, 30, 60, and 90 mM NaCl. The irrigation with saline solution (250 mL in each pots) was performed every two days . Total amount of NaCl per pot during experiment was 8.75 L. To avoid osmotic shock the salinity treatment started with 15 mM NaCl, and was progressively increased (every two days) to reach the maximum salinity level in each treatment. To prevent water and nutrient leaching, a plastic dish was inserted under each pot and the leached water was given back to pots.
Click to Show/Hide
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor Function |
Salinity has negative impact on plant production and stimulates several physiological and biochemical modifications in thyme species. Here, a major decline in dry matter, relative water content (RWC), photosynthetic pigment contents, K+, Ca+ as well as increase in Na+ and EL was observed. Our work showed that T. daenensis was similar to T.vulgaris in terms of tolerance to high levels of salinity stress and both species were moderately tolerant to severe salt stress. It should be noted that T. daenensis, as an Iranian endemic species, competes with commercial species like T. vulgaris as a rich source of phenolic compounds. Furthermore, the relative salt tolerance of both species could be related to the exclusion of Na+ from the vascular system in order to protect tissues from salt toxicity and also to the increase of phenolic content and radical scavenging activity. Thus, the plant behavior under salinity stress may be used to boost the production of bioactive compounds to be used on an industrial level for the manufacture of nutraceuticals, functional foods and cosmetics.
Click to Show/Hide
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor | Part | Location | NP Content | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
0 mM NaCl
|
Leaves | Isfahan, Iran |
NP Content: 1.3 ± 0.1 %
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
30 mM NaCl
|
Leaves | Isfahan, Iran |
NP Content: 1.6 ± 0.3 %
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
60 mM NaCl
|
Leaves | Isfahan, Iran |
NP Content: 1.9 ± 0.2 %
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
90 mM NaCl
|
Leaves | Isfahan, Iran |
NP Content: 2.2 ± 0.1 %
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Species Name: Thymus vulgaris | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor Name: NaCl Treatment | [4] | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Experiment Detail |
The seeds were germinated in seedling plastic tray filled up with a mixture of peat moss and coco peat (1:1). The trays were placed in a glass house at the University of Massachusetts, Amherst, USA, in September 2015. After two weeks, seedlings were transplanted into plastic pots (30 cm diameter and 30 cm height, three seedlings per pot). The pots growth media consisted of 2:1:1 ratio of sieved field soil, sand, and leaf mold, respectively. The medium in each pot was supplemented with 5.8 mg P, 3.3 mg N, and 13.8 mg K to warrant the plant growth; the electrical conductivity was 0.3 mS/cm (Beckman EC meter instrument. cedar grove, New jersey, USA); the pH was 8.04. Plants were maintained at the temperature of 18-25 ℃ ; glass house conditions were set as follows: photoperiod, 16 h; relative humidity, 60-70%; light intensity, 180 µmol m 2 s-1. The source of light was a high pressure sodium lamp in addition to the day light in order to attain 16 h of light per day. Fourty days after planting (establishment period), the uniform sized plants were treated with four different salinity levels including: 0, 30, 60, and 90 mM NaCl. The irrigation with saline solution (250 mL in each pots) was performed every two days . Total amount of NaCl per pot during experiment was 8.75 L. To avoid osmotic shock the salinity treatment started with 15 mM NaCl, and was progressively increased (every two days) to reach the maximum salinity level in each treatment. To prevent water and nutrient leaching, a plastic dish was inserted under each pot and the leached water was given back to pots.
Click to Show/Hide
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor Function |
Salinity has negative impact on plant production and stimulates several physiological and biochemical modifications in thyme species. Here, a major decline in dry matter, relative water content (RWC), photosynthetic pigment contents, K+, Ca+ as well as increase in Na+ and EL was observed. Our work showed that T. daenensis was similar to T.vulgaris in terms of tolerance to high levels of salinity stress and both species were moderately tolerant to severe salt stress. It should be noted that T. daenensis, as an Iranian endemic species, competes with commercial species like T. vulgaris as a rich source of phenolic compounds. Furthermore, the relative salt tolerance of both species could be related to the exclusion of Na+ from the vascular system in order to protect tissues from salt toxicity and also to the increase of phenolic content and radical scavenging activity. Thus, the plant behavior under salinity stress may be used to boost the production of bioactive compounds to be used on an industrial level for the manufacture of nutraceuticals, functional foods and cosmetics.
Click to Show/Hide
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor | Part | Location | NP Content | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
0 mM NaCl
|
Leaves | Isfahan, Iran |
NP Content: 1.6 ± 0.09 %
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
30 mM NaCl
|
Leaves | Isfahan, Iran |
NP Content: 1.7 ± 0.4 %
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
60 mM NaCl
|
Leaves | Isfahan, Iran |
NP Content: 1.9 ± 0.3 %
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
90 mM NaCl
|
Leaves | Isfahan, Iran |
NP Content: 2.1 ± 0.1 %
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||