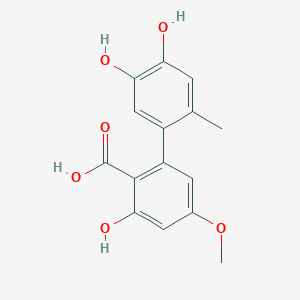
| The Content Variation of Natural Product Induced by Different Factor(s) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Species Name: Alternaria alternata isolate Tche153 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor Name: YES medium | [1] | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Experiment Detail |
Apparently healthy leaves of T. chebula (Family Combretaceae) were collected from Suanluang Rama IX Public Park, Bangkok, Thailand. The endophytic fungus A. alternata Tche-153 was cultivated on PDA agar plates at 25 ℃ for 7 days. Six pieces (@ 0.5 × 0.5 cm) of mycelial agar plugs were inoculated into a 1000-ml Erlenmeyer flask containing 200 ml of yeast extract sucrose (YES) broth and incubated at 25 ℃ under still conditions for 21 days.
Click to Show/Hide
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor | Part | Location | NP Content | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
YES medium (25℃ + 21 days)
|
Leaves | Bangkok, Thailand |
NP Content: 10 mg
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Species Name: Botryosphaeria dothidea strain KJ-1 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor Name: Rice Medium | [2] | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Experiment Detail |
The endophytic fungal strain KJ-1 was isolated from the symptomless tissue of stem bark of M. azedarach L., which was collected at Yangling, Shaanxi province, China, in August 2011. The producing strain was cultured on a plate of potato dextrose agar (PDA) medium at 28 ± 0.5 °C for 5 days. Then one piece (approximately 7 mm2) of mycelium was inoculated aseptically to 50 mL Erlenmeyer flasks each containing 20 mL of PD liquid medium, and the seed liquids were incubated at 28 ± 0.5 °C for 3 days on a rotary shaker at 120 rpm. A suspension (100 µL) of the seed liquid was inoculated aseptically to 500 mL Erlenmeyer flasks each containing 60 g of rice and 90 mL of distilled water. There were 300 flasks in total, and the flask cultures were incubated at 28 ± 0.5 °C for 4 weeks.
Click to Show/Hide
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor | Part | Location | NP Content | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Rice Medium (28 ± 0.5 degrees Celsius + 28Days)
|
tissue of stem bark | Yangling, Shaanxi province, China |
NP Content: 38 mg
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||