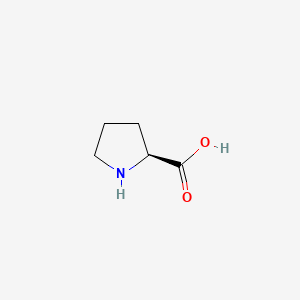
| The Content Variation of Natural Product Induced by Different Factor(s) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Species Name: Asparagus aethiopicus L. (A. sprengeri Regel) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor Name: NaCl Treatment; Seaweed extracts Treatment; 5-aminolevulinic acid Treatment; Harvest Time Variation | [1] | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Experiment Detail |
They were grown in 14 cm plastic pots containing soil mixture of peat:sand (1:1) and maintained in a controlled greenhouse conditions in a private nursery located on Alexandria-Cairo Desert Road. The soil was fertilized with 2 g l -1 Crystalon (19% N: 19% P: 19% K, Chema Industries, Egypt). The plants were maintained at natural light conditions and the temperature ranged between 15 and 28 ℃ . Plants were watered every two days with 2000 and 4000 ppm NaCl solution. The plants subjected to saline conditions (watered with 2000 and 4000 ppm NaCl solution) were treated with 7 ml/L weekly application of Seaweed extracts (SWE) (Ascophyllum nodosum, Stella Maris, Acadian Seaplants, Canada) as foliar spray until drop-off two weeks ahead of saline conditions. A foliar spray of 5-aminolevulinic acid (Sigma-Aldrich, Germany) at (3, 5 and 10 ppm) was applied weekly until drop-off to all plants during saline conditions and untreated plants were considered as controls. Experiments continued for 6 weeks in two consecutive seasons of 2016 and 2017 and the plants were distributed in three blocks and each treatment was represented by 5 replicates with a total number of 270 plants. The NaCl was added to the tank gradually and the electrical conductivity (EC) was measured using EC meter.
Click to Show/Hide
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor Function |
There were significant increases in branch length and number of branches per plant, fresh and the dry weight in Seaweed extracts (SWE) + 5-aminolevulinic acid (ALA) treated plants under saline irrigation conditions compared to control. These morphological improvements associated with several physiological changes in treated plants including increased accumulation of specific phenols (robinin, rutin, apigein, chlorogenic acid and caffeic acid) and increased antioxidant activities of leaf extracts. There were also increased the chlorophyll composition and the accumulation of sugars and proline. Improved transpiration and photosynthetic rates as well as stomatal conductance were also detected in treated plants. The expression of several genes responsible for water management, flavonoid accumulation and antioxidants accumulation was enhanced in SWE + ALA treated plants.
Click to Show/Hide
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Mechanism |
Under normal and saline irrigation conditions, there were changes in genes expression following SWE and ALA applications.The transcription levels of ANN1 and ANN2 increased significantly in SWE + ALA treatments compared to control under non-saline and saline conditions . However, transcription levels of MYB44 showed no significant variations among treatments. PIP1, P5CS1 and CHS relative expressions was higher in plants treated with SWE + ALA under normal and saline irrigation conditions . The transcription levels of the redox responsive genes of APX1 and GPX3 were significantly higher in SWE + ALA treated plants compared to control treatments . Increasing ALA amount from 3 to 10 ppm had no significant effects on APX1 transcription levels under non-saline conditions. In addition, the application of SWE or ALA only had no significant effects on GPX3 transcription levels under non-saline conditions.
Click to Show/Hide
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor | Part | Location | NP Content | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Harvesting time: January-2016 + 640 ppm NaCl + 0 mL/L Seaweed extracts + 0 ppm 5-aminolevulinic acid
|
Leaves | Alexandria, Egypt |
NP Content: 6.21 mg/g dry weight
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Harvesting time: January-2016 + 640 ppm NaCl + 7 mL/L Seaweed extracts + 0 ppm 5-aminolevulinic acid
|
Leaves | Alexandria, Egypt |
NP Content: 6.87 mg/g dry weight
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Harvesting time: January-2016 + 640 ppm NaCl + 3 mL/L Seaweed extracts + 0 ppm 5-aminolevulinic acid
|
Leaves | Alexandria, Egypt |
NP Content: 6.86 mg/g dry weight
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Harvesting time: January-2016 + 640 ppm NaCl + 7 mL/L Seaweed extracts + 3 ppm 5-aminolevulinic acid
|
Leaves | Alexandria, Egypt |
NP Content: 7.71 mg/g dry weight
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Harvesting time: January-2016 + 640 ppm NaCl + 7 mL/L Seaweed extracts + 5 ppm 5-aminolevulinic acid
|
Leaves | Alexandria, Egypt |
NP Content: 7.6 mg/g dry weight
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Harvesting time: January-2016 + 640 ppm NaCl + 7 mL/L Seaweed extracts + 10 ppm 5-aminolevulinic acid
|
Leaves | Alexandria, Egypt |
NP Content: 8.71 mg/g dry weight
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Harvesting time: January-2016 + 2000 ppm NaCl + 0 mL/L Seaweed extracts + 0 ppm 5-aminolevulinic acid
|
Leaves | Alexandria, Egypt |
NP Content: 10.74 mg/g dry weight
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Harvesting time: January-2016 + 2000 ppm NaCl + 7 mL/L Seaweed extracts + 0 ppm 5-aminolevulinic acid
|
Leaves | Alexandria, Egypt |
NP Content: 11.91 mg/g dry weight
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Harvesting time: January-2016 + 2000 ppm NaCl + 0 mL/L Seaweed extracts + 3 ppm 5-aminolevulinic acid
|
Leaves | Alexandria, Egypt |
NP Content: 11.87 mg/g dry weight
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Harvesting time: January-2016 + 2000 ppm NaCl + 7 mL/L Seaweed extracts + 3 ppm 5-aminolevulinic acid
|
Leaves | Alexandria, Egypt |
NP Content: 12.86 mg/g dry weight
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Harvesting time: January-2016 + 2000 ppm NaCl + 7 mL/L Seaweed extracts + 5 ppm 5-aminolevulinic acid
|
Leaves | Alexandria, Egypt |
NP Content: 12.68 mg/g dry weight
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Harvesting time: January-2016 + 2000 ppm NaCl + 7 mL/L Seaweed extracts + 10 ppm 5-aminolevulinic acid
|
Leaves | Alexandria, Egypt |
NP Content: 13.36 mg/g dry weight
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Harvesting time: January-2016 + 4000 ppm NaCl + 0 mL/L Seaweed extracts + 0 ppm 5-aminolevulinic acid
|
Leaves | Alexandria, Egypt |
NP Content: 14.8 mg/g dry weight
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Harvesting time: January-2016 + 4000 ppm NaCl + 7 mL/L Seaweed extracts + 0 ppm 5-aminolevulinic acid
|
Leaves | Alexandria, Egypt |
NP Content: 15.01 mg/g dry weight
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Harvesting time: January-2016 + 4000 ppm NaCl + 0 mL/L Seaweed extracts + 3 ppm 5-aminolevulinic acid
|
Leaves | Alexandria, Egypt |
NP Content: 15.78 mg/g dry weight
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Harvesting time: January-2016 + 4000 ppm NaCl + 7 mL/L Seaweed extracts + 3 ppm 5-aminolevulinic acid
|
Leaves | Alexandria, Egypt |
NP Content: 16.43 mg/g dry weight
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Harvesting time: January-2016 + 4000 ppm NaCl + 7 mL/L Seaweed extracts + 5 ppm 5-aminolevulinic acid
|
Leaves | Alexandria, Egypt |
NP Content: 17.41 mg/g dry weight
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Harvesting time: January-2016 + 4000 ppm NaCl + 7 mL/L Seaweed extracts + 10 ppm 5-aminolevulinic acid
|
Leaves | Alexandria, Egypt |
NP Content: 18.25 mg/g dry weight
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Harvesting time: January-2017 + 640 ppm NaCl + 0 mL/L Seaweed extracts + 0 ppm 5-aminolevulinic acid
|
Leaves | Alexandria, Egypt |
NP Content: 6.01 mg/g dry weight
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Harvesting time: January-2017 + 640 ppm NaCl + 7 mL/L Seaweed extracts + 0 ppm 5-aminolevulinic acid
|
Leaves | Alexandria, Egypt |
NP Content: 6.75 mg/g dry weight
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Harvesting time: January-2017 + 640 ppm NaCl + 3 mL/L Seaweed extracts + 0 ppm 5-aminolevulinic acid
|
Leaves | Alexandria, Egypt |
NP Content: 6.69 mg/g dry weight
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Harvesting time: January-2017 + 640 ppm NaCl + 7 mL/L Seaweed extracts + 3 ppm 5-aminolevulinic acid
|
Leaves | Alexandria, Egypt |
NP Content: 7.67 mg/g dry weight
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Harvesting time: January-2017 + 640 ppm NaCl + 7 mL/L Seaweed extracts + 5 ppm 5-aminolevulinic acid
|
Leaves | Alexandria, Egypt |
NP Content: 7.61 mg/g dry weight
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Harvesting time: January-2017 + 640 ppm NaCl + 7 mL/L Seaweed extracts + 10 ppm 5-aminolevulinic acid
|
Leaves | Alexandria, Egypt |
NP Content: 8.42 mg/g dry weight
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Harvesting time: January-2017 + 2000 ppm NaCl + 0 mL/L Seaweed extracts + 0 ppm 5-aminolevulinic acid
|
Leaves | Alexandria, Egypt |
NP Content: 10.63 mg/g dry weight
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Harvesting time: January-2017 + 2000 ppm NaCl + 7 mL/L Seaweed extracts + 0 ppm 5-aminolevulinic acid
|
Leaves | Alexandria, Egypt |
NP Content: 11.87 mg/g dry weight
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Harvesting time: January-2017 + 2000 ppm NaCl + 0 mL/L Seaweed extracts + 3 ppm 5-aminolevulinic acid
|
Leaves | Alexandria, Egypt |
NP Content: 11.84 mg/g dry weight
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Harvesting time: January-2017 + 2000 ppm NaCl + 7 mL/L Seaweed extracts + 3 ppm 5-aminolevulinic acid
|
Leaves | Alexandria, Egypt |
NP Content: 12.83 mg/g dry weight
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Harvesting time: January-2017 + 2000 ppm NaCl + 7 mL/L Seaweed extracts + 5 ppm 5-aminolevulinic acid
|
Leaves | Alexandria, Egypt |
NP Content: 12.61 mg/g dry weight
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Harvesting time: January-2017 + 2000 ppm NaCl + 7 mL/L Seaweed extracts + 10 ppm 5-aminolevulinic acid
|
Leaves | Alexandria, Egypt |
NP Content: 13.15 mg/g dry weight
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Harvesting time: January-2017 + 4000 ppm NaCl + 0 mL/L Seaweed extracts + 0 ppm 5-aminolevulinic acid
|
Leaves | Alexandria, Egypt |
NP Content: 14.54 mg/g dry weight
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Harvesting time: January-2017 + 4000 ppm NaCl + 7 mL/L Seaweed extracts + 0 ppm 5-aminolevulinic acid
|
Leaves | Alexandria, Egypt |
NP Content: 15.89 mg/g dry weight
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Harvesting time: January-2017 + 4000 ppm NaCl + 0 mL/L Seaweed extracts + 3 ppm 5-aminolevulinic acid
|
Leaves | Alexandria, Egypt |
NP Content: 15.77 mg/g dry weight
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Harvesting time: January-2017 + 4000 ppm NaCl + 7 mL/L Seaweed extracts + 3 ppm 5-aminolevulinic acid
|
Leaves | Alexandria, Egypt |
NP Content: 16.35 mg/g dry weight
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Harvesting time: January-2017 + 4000 ppm NaCl + 7 mL/L Seaweed extracts + 5 ppm 5-aminolevulinic acid
|
Leaves | Alexandria, Egypt |
NP Content: 17.29 mg/g dry weight
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Harvesting time: January-2017 + 4000 ppm NaCl + 7 mL/L Seaweed extracts + 10 ppm 5-aminolevulinic acid
|
Leaves | Alexandria, Egypt |
NP Content: 18.12 mg/g dry weight
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Species Name: Brassica juncea (var. RLC-1) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor Name: K2CrO4 Treatment; Na2SeO4 Treatment | [2] | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Experiment Detail |
The seeds of B. juncea(var. RLC 1) were sterilized with 0.01% mercuric chloride (HgCl2), soaked in distilled water for 2h (h), and germinated in Petri dishes lined with Whatman No. 1 filter paper moistened with 3 ml solutions of sodium selenate (Na2SeO4) (2, 4, and 6µM) and potassium chromate (K2CrO4) (300µM), in unary and binary treatments. The concentrations of the elements were decided on the basis of preliminary experiments by obtaining the most stimulatory concentration of Se and 50% inhibitory concentration (IC50) of Cr. The treatment solutions were prepared in half strength Hoagland's nutrient medium and the experiment was conducted in triplicates with 16hphotoperiodand 25℃ temperature. The seedlings were harvested after 15days for the estimation of various parameters.
Click to Show/Hide
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor Function |
In conclusion, Cr negatively affects the metabolic activities of B. juncea, which were restored by Se application. Se application showed its protective effects on photosynthetic pigments and gene expression studies confirmed the spectrophotometric observations. The Scanning electron microscope (SEM) studies also clearly indicated the positive effects of Se against Cr toxicity on morphology and density of stomata, which further confirmed its imperative role in efficient gas exchange for both photosynthesis and respiration. Exogenous application of Se was also observed to enhance the lipid- and water-soluble antioxidants, which further indicated its role in alleviating oxidative damage. The positive effect of Se in maintaining the osmotic homeostasis as well as activation of thiols in Cr-stressed seedlings also showed its potential to enhance the ability of B. juncea seedlings to combat heavy metal stress effectively. The results, therefore, indicate that Se can be effectively used as a mitigating agent against Cr stress.
Click to Show/Hide
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Mechanism |
Treatments of Se and Cr aided in modulating the gene expression of CHLASE, PSY and CHS. CHLASE showed a significant upregulation of 366.3% in its expression in the Cr treated seedlings with respect to untreated control seedlings. The expression of PSY, however, was observed to be downregulated with Cr application by 28.6%, while CHS expression showed an upregulation by 73.13%. Se application at 4 µM in combination with 300 µM Cr downregulated its expression by 46.92% when compared to Cr-treated seedlings. On the other hand, the same concentration of Se caused an increase of PSY and CHS expression by 425.2 and 209.4%, respectively.
Click to Show/Hide
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor | Part | Location | NP Content | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
0 µ/Kg K2CrO4 + 0 µ/Kg Na2SeO4 (Control)
|
Seedlings | NA |
NP Content: 0.01541 ± 0.00247 mg/g fresh weight
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
0 µ/Kg K2CrO4 + 2 µ/Kg Na2SeO4
|
Seedlings | NA |
NP Content: 0.01986 ± 0.00208 mg/g fresh weight
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
0 µ/Kg K2CrO4 + 4 µ/Kg Na2SeO4
|
Seedlings | NA |
NP Content: 0.02112 ± 0.0011 mg/g fresh weight
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
0 µ/Kg K2CrO4 + 6 µ/Kg Na2SeO4
|
Seedlings | NA |
NP Content: 0.02454 ± 0.00195 mg/g fresh weight
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
300 µ/Kg K2CrO4 + 0 µ/Kg Na2SeO4
|
Seedlings | NA |
NP Content: 0.03927 ± 0.00223 mg/g fresh weight
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
300 µ/Kg K2CrO4 + 2 µ/Kg Na2SeO4
|
Seedlings | NA |
NP Content: 0.04692 ± 0.00213 mg/g fresh weight
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
300 µ/Kg K2CrO4 + 4 µ/Kg Na2SeO4
|
Seedlings | NA |
NP Content: 0.05536 ± 0.00189 mg/g fresh weight
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
300 µ/Kg K2CrO4 + 6 µ/Kg Na2SeO4
|
Seedlings | NA |
NP Content: 0.03824 ± 0.00351 mg/g fresh weight
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor Name: CdCl2 Treatment; Earthworms Treatment | [3] | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Experiment Detail |
To observe the effect of earthworms in Cd-polluted soils, a pot experiment was conducted. The seeds were washed with Tween 20 and surface sterilized before sowing. The pots were filled with soil and cattle dung (partially decomposed) in the ratio of 2:1. The pots were inoculated with earthworms (5 earthworms Kg -1 soil) 7 days prior to sowing, and seeds were then uniformly sown in pots. Plants were allowed to grow under natural conditions and then harvested after 30 and 60 days after sowing (DAS).Different concentrations of Cd (0.50, 0.75, 1.00 and 1.25 mM) were prepared using CdCl2 (anhydrous) obtained from HiMedia. The various concentrations were prepared by dissolving CdCl2 in double-distilled water. The soil was spiked with the different concentrations of Cd and analysed for Cd concentration before experimental set-up using atomic absorption spectrophotometer (AAS). The Cd content was found to be approximately 56 mg (0.5 mM Cd), 84 mg (0.75 mM Cd), 112 mg (1.00 mM Cd) and 140 mg (1.25 mM) in respective concentrations per kilogram of soil. All metal-treated soils were supplemented with fixed number of earthworms. Two sets were maintained which included one without earthworms (WTE) and other supplemented with earthworms (WE).
Click to Show/Hide
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor Function |
Earthworms help to mitigate the toxic effects produced by Cd on plant growth and photosynthetic efficiency along with enhanced phytoremediation capacity when co-inoculated with Cd in soil.
Click to Show/Hide
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Mechanism |
In the present study, the expression of genes CHLASE, PSY, CHS and PAL was enhanced in plants grown in Cd-treated soils supplemented with earthworms. Significant differences in expression of CHLASE, PSY, CHS and PAL were observed after the analysis of data using two-way ANOVA and Tukey's HSD test. The expression of gene CHLASE was enhanced by 1.56-fold in Cd-treated plants which was further enhanced to 3.63-fold when earthworms were co-inoculated along with Cd-treated soils. In comparison to control plants, the expression of PSY and CHS was significantly enhanced by 1.43-fold and 2.07-fold when plants were grown in 1.25 mM Cd treatment. The expression was further enhanced to 3.32-fold (PSY) and 3.37-fold (CHS) when earthworms were supplemented along with Cd treatment (1.25 mM). Similarly, significant increase in the expression of PAL was also observed in Cd-treated plants by 1.64-fold in Cd-treated soil which was enhanced to 2.74-fold in the presence of earthworms.
Click to Show/Hide
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor | Part | Location | NP Content | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
0 mM CdCl2 + with earthworms + 30 DAS
|
NA | Ludhiana, India |
NP Content: 0.10093 ± 0.01241 mg/g
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
0 mM CdCl2 + with earthworms + 60 DAS
|
NA | Ludhiana, India |
NP Content: 0.1124 ± 0.00903 mg/g
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
0.50 mM CdCl2 + without earthworms + 30 DAS
|
NA | Ludhiana, India |
NP Content: 0.2042 ± 0.00775 mg/g
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
0.75 mM CdCl2 + without earthworms + 30 DAS
|
NA | Ludhiana, India |
NP Content: 0.22661 ± 0.00739 mg/g
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
1.00 mM CdCl2 + without earthworms + 30 DAS
|
NA | Ludhiana, India |
NP Content: 0.28126 ± 0.02303 mg/g
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
1.25 mM CdCl2 + without earthworms + 30 DAS
|
NA | Ludhiana, India |
NP Content: 0.38235 ± 0.03222 mg/g
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
0.50 mM CdCl2 + without earthworms + 60 DAS
|
NA | Ludhiana, India |
NP Content: 0.24246 ± 0.01664 mg/g
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
0.75 mM CdCl2 + without earthworms + 60 DAS
|
NA | Ludhiana, India |
NP Content: 0.32825 ± 0.01092 mg/g
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
1.00 mM CdCl2 + without earthworms + 60 DAS
|
NA | Ludhiana, India |
NP Content: 0.40749 ± 0.00775 mg/g
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
1.25 mM CdCl2 + without earthworms + 60 DAS
|
NA | Ludhiana, India |
NP Content: 0.4853 ± 0.01056 mg/g
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
0 mM CdCl2 + without earthworms + 30 DAS
|
NA | Ludhiana, India |
NP Content: 0.14792 ± 0.00841 mg/g
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
0 mM CdCl2 + without earthworms + 60 DAS
|
NA | Ludhiana, India |
NP Content: 0.16923 ± 0.01151 mg/g
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
0.50 mM CdCl2 + with earthworms + 30 DAS
|
NA | Ludhiana, India |
NP Content: 0.14058 ± 0.00748 mg/g
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
0.75 mM CdCl2 + with earthworms + 30 DAS
|
NA | Ludhiana, India |
NP Content: 0.15885 ± 0.02364 mg/g
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
1.00 mM CdCl2 + with earthworms + 30 DAS
|
NA | Ludhiana, India |
NP Content: 0.24355 ± 0.04591 mg/g
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
1.25 mM CdCl2 + with earthworms + 30 DAS
|
NA | Ludhiana, India |
NP Content: 0.36596 ± 0.02303 mg/g
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
0.50 mM CdCl2 + with earthworms + 60 DAS
|
NA | Ludhiana, India |
NP Content: 0.1976 ± 0.01187 mg/g
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
0.75 mM CdCl2 + with earthworms + 60 DAS
|
NA | Ludhiana, India |
NP Content: 0.2892 ± 0.008102 mg/g
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
1.00 mM CdCl2 + with earthworms + 60 DAS
|
NA | Ludhiana, India |
NP Content: 0.36869 ± 0.01496 mg/g
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
1.25 mM CdCl2 + with earthworms + 60 DAS
|
NA | Ludhiana, India |
NP Content: 0.4737 ± 0.01682 mg/g
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Species Name: Brassica juncea L. | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor Name: CdCl2 Treatment; Citric Acid Treatment; Castasterone Treatment | [4] | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Experiment Detail |
Earthen pots (12 in. diameter) were filled with five kg of soil per pot and arranged in a randomized block design (Kothari, 2004) with 5 pots for each treatment. 0 mM Cd(II) was added to the soil in form of anhydrous CdCl2. The seeds of B. juncea var. RLC-1 (procured from Punjab Agricultural University, Ludhiana, India) were surface sterilized with 0.5% (v/v) sodium hypochlorite. These were then soaked in different concentration of castasterone (CS) (0 nM) for 8 h before sowing. After seven days of germination, 0 mM citric acid (CA) was supplemented to the soil. The plants were harvested after 30 days of sowing, and fresh mature top leaves were analysed for biochemical, physiological and molecular studies.
Click to Show/Hide
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor Function |
Castasterone (CS) and Citric Acid (CA) treatments also improved the osmolyte contents of the leaves which enabled the plants to tide over Cd (II) stress. Beta regression and multiple linear regression analysis showed that CS treatment has more positive effect on shoot height, fresh weight and photosynthetic pigments and efficiency than the CA treatment. The most effective concentration enhancing Cd(II) stress tolerance in B. juncea plants was seed soaking treatment with 100 nM CS and soil application of 0.6 mM/kg CA soil.
Click to Show/Hide
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Mechanism |
The present study found that relative expression of CHLASE gene was significantly increased by 3.57 folds under Cd(II) toxicity. Seed pre-sowing treatment of 100 nM CS and soil citric acid application significantly lowered expression of CHLASE in 30 day old B. juncea leaves. MLR analysis also revealed positive effect of Cd(II) and negative effects of CA and CS treatments on fold change of CHLASE expression. Maximum reduction in CHLASE expression of 1.19 fold was observed with binary combination of CS (100 nM) and CA (0.6 mM/kg) in the leaves of Cd(II) stressed B. juncea.Expression of PSY and CHS genes was significantly enhanced by binary combination of CS and CA treatment by 2.85 fold and 5.15 fold, respectively. MLR analysis also revealed that CS, CA and Cd(II) treatments positively regressed on fold change of PSY and CHS expressions.
Click to Show/Hide
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor | Part | Location | NP Content | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
0 mM/kg CdCl2 + 0 mM/kg Citric Acid+ 0 nM Castasterone (Control)
|
Fresh leaves | NA |
NP Content: 2.59 ± 0.16 mg/g dry weight
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
0 mM/kg CdCl2 + 0 mM/kg Citric Acid+ 0.01 nM Castasterone
|
Fresh leaves | NA |
NP Content: 2.69 ± 0.21 mg/g dry weight
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
0 mM/kg CdCl2 + 0 mM/kg Citric Acid+ 1 nM Castasterone
|
Fresh leaves | NA |
NP Content: 2.8 ± 0.08 mg/g dry weight
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
0 mM/kg CdCl2 + 0 mM/kg Citric Acid+ 100 nM Castasterone
|
Fresh leaves | NA |
NP Content: 3.11 ± 0.12 mg/g dry weight
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
0 mM/kg CdCl2 + 0.6 mM/kg Citric Acid+ 0 nM Castasterone
|
Fresh leaves | NA |
NP Content: 2.68 ± 0.08 mg/g dry weight
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
0 mM/kg CdCl2 + 0.6 mM/kg Citric Acid+ 0.01 nM Castasterone
|
Fresh leaves | NA |
NP Content: 2.86 ± 0.23 mg/g dry weight
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
0 mM/kg CdCl2 + 0.6 mM/kg Citric Acid+ 1 nM Castasterone
|
Fresh leaves | NA |
NP Content: 2.88 ± 0.17 mg/g dry weight
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
0 mM/kg CdCl2 + 0.6 mM/kg Citric Acid+ 100 nM Castasterone
|
Fresh leaves | NA |
NP Content: 3.13 ± 0.25 mg/g dry weight
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
0.6 mM/kg CdCl2 + 0 mM/kg Citric Acid+ 0 nM Castasterone
|
Fresh leaves | NA |
NP Content: 3.29 ± 0.36 mg/g dry weight
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
0.6 mM/kg CdCl2 + 0 mM/kg Citric Acid+ 0.01 nM Castasterone
|
Fresh leaves | NA |
NP Content: 4.08 ± 0.28 mg/g dry weight
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
0.6 mM/kg CdCl2 + 0 mM/kg Citric Acid+ 1 nM Castasterone
|
Fresh leaves | NA |
NP Content: 4.45 ± 0.25 mg/g dry weight
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
0.6 mM/kg CdCl2 + 0 mM/kg Citric Acid+ 100 nM Castasterone
|
Fresh leaves | NA |
NP Content: 4.41 ± 0.22 mg/g dry weight
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
0.6 mM/kg CdCl2 + 0.6 mM/kg Citric Acid+ 0 nM Castasterone
|
Fresh leaves | NA |
NP Content: 4.55 ± 0.48 mg/g dry weight
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
0.6 mM/kg CdCl2 + 0.6 mM/kg Citric Acid+ 0.01 nM Castasterone
|
Fresh leaves | NA |
NP Content: 4.73 ± 0.37 mg/g dry weight
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
0.6 mM/kg CdCl2 + 0.6 mM/kg Citric Acid+ 1 nM Castasterone
|
Fresh leaves | NA |
NP Content: 4.92 ± 0.3 mg/g dry weight
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
0.6 mM/kg CdCl2 + 0.6 mM/kg Citric Acid+ 100 nM Castasterone
|
Fresh leaves | NA |
NP Content: 5.3 ± 0.24 mg/g dry weight
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Species Name: Lagenaria siceraria | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor Name: Cultivar Comparison; Developmental Stage Variation | [5] | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Experiment Detail |
The experiment was conducted from July to November 2007 at the experimental station of the University of Abobo-Adjame (Abidjan, Ivory Coast) (latitude between 5° 17′ and 5° 31′ N, longitude between 3° 45′ and 4° 22′ W). During this period, rainfall, mean temperature and humidity varied from 5.33 to 192.28 mm, from 23.3 to 26.4 ℃ and from 86.7 to 96% respectively. Open-pollinated accessions from two edible-seeded L. siceraria (Molina) Standl. cultivars recognisable by their fruit shape (oval or round) were used. Seeds from the round fruit cultivar are characterised by the presence of a cap on the distal side, whereas those from the oval fruit cultivar lack this cap. Differences are also noted between the two cultivars in their rates of seed germination and seedling emergence, the best performances being observed for the round fruit cultivar. However, the visual changes in fruits during their growth as well as at plant whiteness are the same in the two cultivars. Both round and oval fruit cultivars were obtained from the cucurbit germplasm of the university, where they are identified by the alphanumeric codes NI354 and NI260 respectively. Each cultivar was sown on a plot of 20 m × 20 m in 12 holes. Female flowers were tagged after their closure in order to monitor the fruits until the date determined for harvesting. Fruits were harvested at three stages of maturation: (i) 30 days after fruit set (DAFS), at which stage fruits do not grow any more; (ii) 50 DAFS, at which stage the colour of fruits no longer changes; (iii) complete plant whiteness (CPW), indicating the end of plant growth. For each of the three fruit maturation times, five fruits per cultivar were selected. The seeds were extracted from each fruit, washed and dried in the sun for 1 week. After drying, the seeds of all five fruits were grouped and decorticated to obtain seed kernels that were used for analysis.
Click to Show/Hide
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor Function |
The results of this study showed that there were considerable modifications during fruit maturation in the oilseed gourd L. siceraria and that chemical characterisation of the seed kernel is important for controlling the processes of maturation. The two cultivars of L. siceraria studied should be harvested at 50 days after fruit set (DAFS) owing to their high contents of proteins, lipids, energy and minerals at this stage. At 50 DAFS the leaves of L. siceraria were still green and could serve as a source of nutrients for livestock. However, to obtain the best amino acid composition and biological values of proteins, the round and oval berry cultivars should be harvested at 30 DAFS and complete plant whiteness (CPW) respectively. At these stages their proteins could be used as a supplement. The low digestibility of the proteins at this stage could be improved by appropriate technological treatment.
Click to Show/Hide
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor | Part | Location | NP Content | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Lagenaria siceraria cv. Round berry + Harvesting time: 30 days after fruit set
|
Seed kernels | Abidjan, Cote d'lvoire |
NP Content: 11.8 ± 0.1 g/kg dry matter
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Lagenaria siceraria cv. Round berry + Harvesting time: 50 days after fruit set
|
Seed kernels | Abidjan, Cote d'lvoire |
NP Content: 11.4 ± 0.1 g/kg dry matter
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Lagenaria siceraria cv. Round berry + Harvesting time: complete plant whiteness stage
|
Seed kernels | Abidjan, Cote d'lvoire |
NP Content: 11.3 ± 0.4 g/kg dry matter
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Lagenaria siceraria cv. Oval berry + Harvesting time: 30 days after fruit set
|
Seed kernels | Abidjan, Cote d'lvoire |
NP Content: 11.6 ± 0.0 g/kg dry matter
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Lagenaria siceraria cv. Oval berry + Harvesting time: 50 days after fruit set
|
Seed kernels | Abidjan, Cote d'lvoire |
NP Content: 10.6 ± 0.2 g/kg dry matter
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Lagenaria siceraria cv. Oval berry + Harvesting time: complete plant whiteness stage
|
Seed kernels | Abidjan, Cote d'lvoire |
NP Content: 12.2 ± 0.2 g/kg dry matter
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Species Name: Poncirus trifoliata (L.) Raf. | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor Name: Drought Stress Treatment; AMF Inoculation | [6] | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Experiment Detail |
Seven day-old non-AM-infected trifoliate orange [Poncirus trifoliata (L.) Raf.] seedlings were used in this study. Six seedlings were grown in a plastic pot (15 × 20 cm) containing 3.37 kg of autoclaved growth substrate (soil:vermiculite:sphagnum, 5:2:1, v/v/v). The substrate had a pH of 5.9, 1.3% organic matter, 30 mg/kg available phosphorus, 147 mg/kg alkali hydrolysable nitrogen, and 141 mg/kg available potassium. Half of the pots received Glomus versiforme (Karsten) Berch (30 g of inoculum placed 5 cm deep) at transplanting. This inoculum contained approximately 2,233 spores and was provided by the Institute of Plant Nutrition and Resources, Beijing Academy of Agriculture and Forestry Sciences. Control treatment received no AMF inoculum (30 g of autoclaved growth substrate). The AM and non-AM seedlings were placed in a greenhouse without temperature control from March to September 2004.All seedlings were watered daily until differential water treatments were initiated 97 days after transplanting. Pots with WW and DS seedlings were maintained everyday at 75% (corresponding to -0.09 MPa) or 55% (corresponding to -0.40 MPa) relative soil-water content by gravimetry, respectively.The experiment was laid out in randomized complete blocks with two water treatment (WW and DS) and two mycorrhizal treatment (G. versiforme and non-AMF). Each of the four treatments had six replicates.
Click to Show/Hide
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor Function |
AMF colonization enhances osmotic solute accumulation of trifoliate orange seedlings, thus providing better osmotic adjustment in AMF seedlings, which did not correlate with proline but with K+, Ca2+, Mg2+, glucose, fructose and sucrose accumulation.
Click to Show/Hide
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor | Part | Location | NP Content | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Leaf: (Well-watered) + (Glomus versiforme inoculation)
|
Leaves | NA |
NP Content: 0.26 mg/g fresh weight
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Root: (Well-watered) + (Glomus versiforme inoculation)
|
Roots | NA |
NP Content: 0.2 mg/g fresh weight
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Leaf: (Well-watered) + (Non-AMF inoculation)
|
Leaves | NA |
NP Content: 0.46 mg/g fresh weight
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Root: (Well-watered) + (Non-AMF inoculation)
|
Roots | NA |
NP Content: 0.24 mg/g fresh weight
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Leaf: (Drought-stressed) + (Glomus versiforme inoculation)
|
Leaves | NA |
NP Content: 0.37 mg/g fresh weight
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Root: (Drought-stressed) + (Glomus versiforme inoculation)
|
Roots | NA |
NP Content: 0.27 mg/g fresh weight
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Leaf: (Drought-stressed) + (Non-AMF inoculation)
|
Leaves | NA |
NP Content: 0.55 mg/g fresh weight
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Root: (Drought-stressed) + (Non-AMF inoculation)
|
Roots | NA |
NP Content: 0.3 mg/g fresh weight
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||