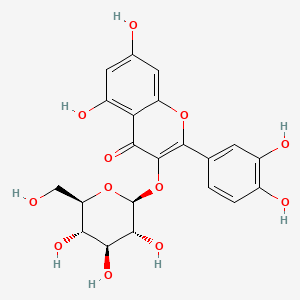
| The Content Variation of Natural Product Induced by Different Factor(s) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Species Name: Amaranthus tricolor genotype VA13 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor Name: NaCl Treatment | [1] | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Experiment Detail |
On the basis of previous studies, an antioxidant enriched high yield potential genotype (Accession VA13) was selected for this investigation. This genotype was grown in pots of a rain shelter open field of Bangabandhu Sheikh Mujibur Rahman Agricultural University, Bangladesh (AEZ-28, 24° 23′ north latitude, 90° 08′ east longitude, 8.4 m.s.l.). The seeds were sown in plastic pots (15 cm in height and 40 cm length and 30 cm width) in a randomized complete block design (RCBD) with three replications. N: P2O5:K2O were applied @92:48:60 kg/ha as a split dose. First, in pot soil, @46:48:60 kg ha 1 N: P2O5:K2O and second, at 7 days after sowing (DAS) @46:0:0 kg/ha N: P2O5:K2O. The genotype was grouped into three sets and subjected to four salinity stress treatments that are, 100 mM NaCl, 50 mM NaCl, 25 mM NaCl, and control or no saline water (NS). Pots were well irrigated with fresh water every day up to 10 days after sowing (DAS) of seeds for proper establishment and vigorous growth of seedlings. Imposition of salinity stress treatment was started at 11 DAS and continued up to 40 DAS (edible stage). Saline water (100 mM NaCl, 50 mM NaCl and 25 mM NaCl) and fresh water were applied to respective pots once a day. At 40 DAS the leaves of Amaranthus tricolor were harvested. All the parameters were measured in six samples.
Click to Show/Hide
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor Function |
At Moderate salinity stress (MSS) and Severe salinity stress (SSS) conditions, leaf color parameters and pigments, vitamins, phenolic acids, flavonoids and antioxidant capacity of A. tricolor leaves were very high compared to control condition. Hence, salt-stressed A. tricolor leaves had a good source of natural antioxidants compared to plant grown in normal cultivation practices.
Click to Show/Hide
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor | Part | Location | NP Content | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
No saline water (Control)
|
Leaves | Bangabandhu |
NP Content: 4.66 ± 0.21 µg/g fresh weight
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
25 mM NaCl (Low salinity stress)
|
Leaves | Bangabandhu |
NP Content: 4.80 ± 0.24 µg/g fresh weight
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
50 mM NaCl (Moderate salinity stress)
|
Leaves | Bangabandhu |
NP Content: 7.23 ± 0.16 µg/g fresh weight
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
100 mM NaCl (Severe salinity stress)
|
Leaves | Bangabandhu |
NP Content: 9.24 ± 0.18 µg/g fresh weight
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Species Name: Crocus sativus L. (saffron) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor Name: AMF Inoculation; Harvest Time Variation | [2] | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Experiment Detail |
AMF Inoculation in Pot : Saffron corms with horizontal diameters of 1.3 to 2.8 cm were sown in pots (4 L; 1 corm per pot) in the last ten days of August 2016. Pots were filled with sterile quartz sand (3 L per pot) on a layer of sterilized expanded clay (1 L per pot). Corms were treated with two inocula (MycAgro Lab, Breteniere, FR), one composed of a single fungus Rhizophagus intraradices (Ri) and one of R. intraradices and Funneliformis mosseae (Ri + Fm). Ten grams of each inoculum were placed under each corm in order to guarantee the contact between the inoculum and the roots and therefore to favor the symbiosis between AMF and roots. Saffron corms used as controls were not inoculated (AMF-). Corms were not treated against fungal pathogens. A randomized block design was used with a total of 48 pots displayed in two experimental plot units (24 pots per unit) and three treatments (8 pots per treatment). Cultivation lasted for one cycle (August 2016-April 2017) in a heated glasshouse of the Department of Agricultural Forest and Food Sciences (DISAFA) of the University of Torino (Italy, 45° 06′ 23.21″ N Lat, 7° 57′ 82.8″ E Long; 293 m a.s.l.), with an average temperature of 22 ℃ during the day and 16 ℃ in the night. Irrigation water (pH 7.4, EC 505 µS cm) was added weekly (250 mL per pot) with a drip system. The corms were fertilized by fertigation (VIGORFLOR, AL.FE. srl, MN, Italy) every two weeks starting from the emergence of the spate, in quantities of 1.5 g/L of water. No flowering occurred because of the small size of the corms.AMF Inoculation in Open Field : Saffron corms with horizontal diameters of 2.5 to 3.5 cm were planted in the last ten days of August 2016 in two Alpine experimental sites located in the municipality of Morgex (45° 45′ 35″ N; 7° 02′ 37.3″ E; 1000 m a.s.l.) and Saint Cristophe (45° 45′ 06″ N; 7° 20′ 37″ E; 700 m a.s.l.) in Italy and cultivation lasted for two cycles (2016-2017 and 2017-2018). Both sites were cultivated with saffron for at least the previous three years. Before starting the experiment both fields were milled. To assess the effects of AMF inocula on saffron cultivation and production, the same treatments used in the pot trial were applied (Ri, Ri + Fm or AMF-). A randomized block design was used, with three experimental plot units (blocks). Each plot unit consisted of 56 corms, planted in a 1.44 m2 area (39 corms m-2). Inter-row planting distance was of 7 cm, while between-row distance was 25 cm. Plots were separated from each other with at least 4 m distance. Before planting, 10 g of inoculum was placed under the corms to ensure contact between plant and the treatment. Irrigation was provided when needed and hand weeding control was conducted during cultivation, while no preplanting fertilization, tillage, or treatments against pathogens were applied. The two Alpine sites were characterized by semicontinental climate, with a long and cold winter . In general, both sites had a sandy-loam texture according to the USDA classification and similar chemical characteristics.
Click to Show/Hide
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor Function |
The inoculum composed by R. intraradices and F. mosseae was particularly effective in increasing flower production and saffron yield, while R. intraradices alone increased the content of some bioactive compounds-picrocrocin, quercitrin, crocin II-as well as antioxidant activity.
Click to Show/Hide
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor | Part | Location | NP Content | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Harvesting time: 2016-2017
|
Powdered saffrons | Italy |
NP Content: 2 mg/100g dry weight
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Harvesting time: 2017-2018
|
Powdered saffrons | Italy |
NP Content: 2 mg/100g dry weight
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Rhizophagus intraradices and Funneliformis mosseae inoculation
|
Powdered saffrons | Italy |
NP Content: 2 mg/100g dry weight
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Rhizophagus intraradices inoculation
|
Powdered saffrons | Italy |
NP Content: 2 mg/100g dry weight
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Non-AMF inoculation (Control)
|
Powdered saffrons | Italy |
NP Content: 2 mg/100g dry weight
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Species Name: Prunus persica Batsch cv. 'Yuhua No. 2' | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor Name: Low Temperature Treatment; Glycine betaine Treatment | [3] | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Experiment Detail |
Peach fruit (Prunus persica Batsch cv. 'Yuhua No. 2') was hand-harvested at commercial maturity (about 9-12N firmness, 10-12% total soluble solids) from a local orchard in Nanjing, China. The fresh weight of 'Yuhua No. 2' peach is about 215g and the dry weight is about 30g. The fruit shape is round and the diameter size is about 72 mm. The peaches were selected in uniform size and color and absence of any damage. The selected peaches were randomly divided into two groups, each with 360 fruits for 3 replicates. According to our previous study, 10 mmol/LGB was selected as the treatment concentration. Peach fruits were immersed in 10 mmol/LGB solution for 10 min to ensure that GB could be equally distributed on the fruits. The control fruits were soaked in sterile deionized water for 10 min. After treatment, all fruits were air dried about 30 min and stored at 0℃ with a relative humidity of 85-90% for 35 days. Mesocarp samples were collected from 18 fruits on the 7th, 14th, 21th, 28th, 35th day and frozen in liquid nitrogen, then stored at -80℃ until biochemical analysis. Another 18 fruits were removed from 0℃ after 7th, 14th, 21th, 28th, 35th day, and held at 20℃ for three days to simulate shelf condition, and then evaluated CI index, firmness and extractable juice. Each treatment was replicated three times and the experiment was conducted twice with similar results.
Click to Show/Hide
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor Function |
Glycine betaine (GB) treatment enhanced chilling tolerance throughout regulating phenolic and sugar metabolisms in peach fruit during cold storage. The alleviation of chilling injury (CI) by GB treatment may be attributed to enhancement of individual of phenolic compounds and sucrose content, and induce the activities of enzymes related to phenolic and sugar metabolisms.
Click to Show/Hide
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor | Part | Location | NP Content | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Cold storage(days): 0
|
Flesh tissues | Nanjing, China |
NP Content: 2.25 ± 0.03 mg/g fresh weight
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Cold storage(days): 7
|
Flesh tissues | Nanjing, China |
NP Content: 3.05 ± 0.04 mg/g fresh weight
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
10 mmol/L Glycine betaine + Cold storage(days): 7
|
Flesh tissues | Nanjing, China |
NP Content: 3.56 ± 0.03 mg/g fresh weight
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Cold storage(days): 21
|
Flesh tissues | Nanjing, China |
NP Content: 2.68 ± 0.03 mg/g fresh weight
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
10 mmol/L Glycine betaine + Cold storage(days): 10
|
Flesh tissues | Nanjing, China |
NP Content: 3.08 ± 0.02 mg/g fresh weight
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Cold storage(days): 35
|
Flesh tissues | Nanjing, China |
NP Content: 1.86 ± 0.06 mg/g fresh weight
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
10 mmol/L Glycine betaine + Cold storage(days): 35
|
Flesh tissues | Nanjing, China |
NP Content: 2.23 ± 0.05 mg/g fresh weight
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Species Name: Rubus idaeus | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor Name: Cultivar Comparison; Organic Fertilization; Traditional Fertilization | [4] | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Experiment Detail |
The experiment was carried out in 2013. Leaves of five raspberry cultivars ('Polana', 'Polka', 'Tulameen', 'Laszka' and 'Glen Ample') were collected at the time of cultivation. Three organic and neighborhood conventional farms were used for experimental purposes. From one cultivar (one field plot), 3-4 plants were chosen, which were analyzed separately. One sample consisted of 10 leaves. The farm was treated as a replication. [organic farm no. 1 Localization: akroczym(52° 26″ N 20° 36″ E), Type of Soil: sandy middle soil IVa and IVb category (15% floatable particles) pH 5.5, Kind of Fertilizer: cow manure, Dose of Fertilizers and Time of Given: 35 t/ha one year before raspberry planting, Plant Protection System: Grevit 200 SL; organic farm no. 2 Localization: Zaluski (52° 37″ N 20° 22″ E), Type of Soil: sandy middle soil, sandy-clay IV category (20% floatable particles), pH 5.5, Kind of Fertilizer:cow manure, Dose of Fertilizers and Time of Given: 30 t/ha one year before raspberry planting, Plant Protection System: no protection; organic farm no. 3 Localization: Radzanow(51° 33″ N 20° 51″ E), Type of Soil: sandy middle soil IVa and III category (10% floatable particles), pH 6.0, Kind of Fertilizer:sheep manure, green manure, Dose of Fertilizers and Time of Given: 10 t/ha and 15 t/ha one year before raspberry planting, Plant Protection System: Bioczos 33 SL, Grevit 200 SL; conventional farm no. 1 Localization: Czerwinsk nad Wisla (52° 23″ N 20° 20″ E), Type of Soil: sandy-loamy middle soil IV and III category (20% floatable particles), pH 5.5, Kind of Fertilizer: Hydrocomplex 12-11-18; Superba 8-11-36, Dose of Fertilizers and Time of Given: (200 kg/ha, 150 kg/ha) in autumn a year before raspberry planting; 3 doses in time of cultivation, Plant Protection System: Signum 33 WG, Miros 20 SP; conventional farm no. 2 Localization: Czerwinsk nad Wisla (52° 23″ N 20° 20″ E), Type of Soil: sandy-loamy middle soil IV and III category (25% floatable particles), pH 5.5, Kind of Fertilizer: amonium nitrate, polyphosphate, magnesium sulphate, Dose of Fertilizers and Time of Given: in autumn a year before raspberry planting; 3 doses in time of cultivation, Plant Protection System: Calypso 480 SC, Miros 20 SP, Zato 50 WG; conventional farm no. 3 Localization: Czerwinsk nad Wisla(52° 25″ N 20° 23″ E), Type of Soil: sandy-clay middle soil II and III category (20% floatable particles) pH 6.0, Kind of Fertilizer:Rosafert 5-12-24-3, Dose of Fertilizers and Time of Given: 250 kg/ha in autumn a year before raspberry planting; 4 doses in time of cultivation, Plant Protection System: Calypso 480 SC, Miros 20 SP, Zato 50 WG].
Click to Show/Hide
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor Function |
Compared with conventional raspberry leaves, organic raspberry leaves were characterized by a significantly higher content of dry matter, total polyphenols, total phenolic acids, chlorogenic acid, caffeic acid, salicylic acid and quercetin-3-O-rutinoside; moreover, the organic leaves were characterized by higher antioxidant activity. Among examined cultivars, 'Polka' c. was characterized by the highest antioxidant status. However, raspberry leaves from conventional farms contained more total carotenoids, violaxanthin, alpha-carotene, beta-carotene, total chlorophyll and individual forms of chlorophylls: a and b.
Click to Show/Hide
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor | Part | Location | NP Content | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Cultivation System: organic farm
|
Leaves | Poland |
NP Content: 31.31 ± 4.13 mg/100g fresh weight
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Cultivation System: conventional farm
|
Leaves | Poland |
NP Content: 21.49 ± 3.55 mg/100g fresh weight
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Rubus idaeus cv. Polana
|
Leaves | Poland |
NP Content: 14.80 ± 1.10 mg/100g fresh weight
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Rubus idaeus cv. Polka
|
Leaves | Poland |
NP Content: 36.43 ± 4.95 mg/100g fresh weight
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Rubus idaeus cv. Tulameen
|
Leaves | Poland |
NP Content: 46.88 ± 6.02 mg/100g fresh weight
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Rubus idaeus cv. Laszka
|
Leaves | Poland |
NP Content: 13.34 ± 1.51 mg/100g fresh weight
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Rubus idaeus cv. Glen Ample
|
Leaves | Poland |
NP Content: 13.09 ± 3.79 mg/100g fresh weight
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Species Name: Vitis vinifera cv. Pinot noir | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor Name: Drought Stress Treatment | [5] | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Experiment Detail |
3-year old single shoot V. vinifera plants (cultivar Pinot noir 18 Gm grafted on Kober 5BB, 51 plants) potted in 3L pots in a sandy loam soil were used. All plants were well watered (200 mL per day) at the beginning of the experiment (04.06.2010; DAY 0; 5 plants) and water was supplied to all control plants once every day (250 mL per day), whereas water supply of stressed plants was stopped. Physiological measurements and sampling of leaves took place on 07.06.2010 (DAY 3; 5 control, 5 stressed plants), 10.06.2010 (DAY 6; 5 control, 5 stressed plants) and 12.06.2010 (DAY 8; 5 control, 10 stressed plants). Due to very hot weather conditions in June 2010 the experiment was stopped after 8 days and 12 available control plants were used to restart the drought treatment with 6 control and 6 stressed plants on 11.06.2010 and all plants were measured on 15.06.2010 (DAY 5). The mean leaf temperatures at midday were: 25 ℃ (04.06.2010; DAY 0), 31.9 ℃ (07.06.2010; DAY 3), 30.8 ℃ (15.06.2010; DAY 5), 35.8 ℃ (10.06.2010; DAY 6) and 35.7 ℃ (12.06.2010; DAY 8). The mean PAR radiation per day (measured from 6:00 am till 7:00 pm) was 144.1 µmol m-2 s-1. Each plant was used only once for physiological measurements and sampling of leaves.On every day of the experiment (day 0, 3, 5, 6, 8) the pot weight and the volumetric soil moisture content (ThetaProbe ML2x and handheld data logger Moisture Meter HH2, Delta-T Devices, Cambridge, United Kingdom) was recorded. The water potential (PWSC Model 3000, Soilmoisture Equipment Corporation, Santa Barbara, USA) was determined for the 6th leaf (representing the insertion level of the shoot from the basis) of every plant and measurement day. Chlorophyll fluorescence and gas exchange parameters of light adapted leaves were determined with the 4th and 5th leaf, whereas dark adaptation was performed only with the 5th leaf. Immediately after these non-invasive measurements, the 5th leaf was harvested, frozen in liquid nitrogen and further used for the measurement of polyphenols, selected primary metabolites and volatiles (VOCs).
Click to Show/Hide
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor Function |
The content of different groups of primary and secondary metabolites is significantly influenced by severe drought stress in grapevine leaves. The content of the majority of the metabolites (around 60% of primary metabolites, around 85% of polyphenols and about 40% of the detected and identified VOCs) increased upon drought stress treatment. Among these especially the primary metabolites citric acid and glyceric acid were strongly influenced by the short as well as the prolonged drought stress treatment, whereas all polyphenols were only induced upon the prolonged drought stress treatment.
Click to Show/Hide
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Factor | Part | Location | NP Content | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Normal condition
|
Leaves | Vienna, Austria |
NP Content: 11.7 ± 3.1 µg/g dry weight
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Dry 3-5 days
|
Leaves | Vienna, Austria |
NP Content: 11.2 ± 4.0 µg/g dry weight
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Dry 6-8 days
|
Leaves | Vienna, Austria |
NP Content: 40.3 ± 20.1 µg/g dry weight
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||